Introduction to Chemistry www unit 5 orgchemistry Problems

Introduction to Chemistry www. unit 5. org/chemistry

Problems call forth our courage and our wisdom; indeed, they create our courage and our wisdom. it is only because of problems that we grow mentally and spiritually. It is through the pain of confronting and resolving problems that we learn. M. Scott Peck, b. 1936 American Psychiatrist and Writer All through my life, the new sights of Nature made me rejoice like a ch Marie Curie, 1867 -1934 Polish-Born French C Believe that life is worth living, and your belief will help create the fact.

Chemistry is the study of matter and the transformations it can undergo…

…Matter is anything that occupies space. Chemistry Purpose with a
Interactive Periodic Table < e 1 H H He 1 2 3 Li Be B C N O F Ne 3 4 5 6 7 8 9 10 Al Si P S Cl Ar 13 14 15 16 17 18 Na Mg 11 4 K 19 5 7 Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 23 24 35 36 I Xe 53 54 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 12 38 Cs Ba 55 56 Fr Ra 87 88 * W 25 43 26 44 Re Os 75 76 27 28 29 47 30 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 Po At Rn 84 85 86 Rf Db Sg Bh Hs Mt 104 105 106 107 108 109 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 59 60 Ac Th Pa U 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103
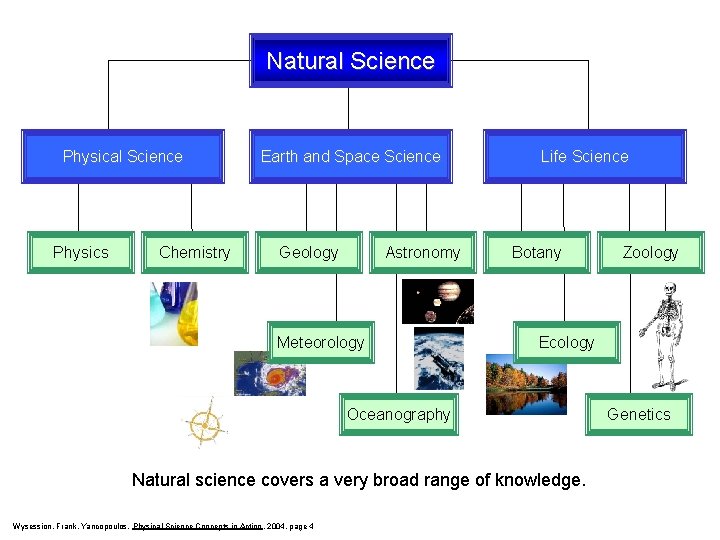
Natural Science Physics Chemistry Earth and Space Science Geology Astronomy Meteorology Life Science Botany Ecology Oceanography Natural science covers a very broad range of knowledge. Wysession, Frank, Yancopoulos, Physical Science Concepts in Action , 2004, page 4 Zoology Genetics

Table of Contents ‘Introduction to Chemistry’ Introduction / Perceptions (17 slides) Safety (21 slides) Pure vs. Applied Science (10 slides) Scientific Method (27 slides) Lab Equipment (6 slides) Alchemy vs. Chemistry (30 slides) Manipulating Numerical Data (11 slides) Conversion Factors and Unit Cancellation (6 slides) Simple Math with Conversion Factors (8 slides) Scientific Notation (18 slides) Using the Exponent Key (17 slides) Basic Concepts in Chemistry (7 slides) Metric System (10 slides) Measurement (23 slides) Essential Math (14 slides)

A Lost Child Keeping Warm Once upon a time a small child became lost. Because the weather was cold, he decided to gather material for a fire. As he brought objects back to his campfire, he discovered that some of them burned and some of them didn’t burn. To avoid collecting useless substances, the child began to keep track of those objects that burned and those that did not. He proposed a possible “generalization. ” Perhaps: “Cylindrical objects burn. ” This procedure if one of the elementary logical thought processes by which information is systematized. It is called inductive reasoning (a general rule is framed on the basis of a collection of individual observations (or facts)). Jaffe, New World of Chemistry, 1955, page 3 -4

“Cylindrical Objects Burn” WILL BURN WON’T BURN Tree limbs Rocks Broom handles Blackberries Pencils Marbles Chair legs Paperweights Flagpoles Jaffe, New World of Chemistry, 1955, page 3 -4

Using his generalization, the boy gathered more substances to burn. He collected three pieces of pipe, two ginger ale bottles, and the axle from an old car, while leaving a huge cardboard box full of newspapers. During the long cold night that followed he drew these conclusions: (1) The cylindrical shape of a burnable object may not be intimately associated with its flammability after all. (2) Even though the “cylindrical” rule is no longer useful, tree limbs, broom handles, pencils, and other burnables still burn. (3) He’d better bring the list along tomorrow. New idea: Perhaps “Wooden objects burn. ” Jaffe, New World of Chemistry, 1955, page 3 -4
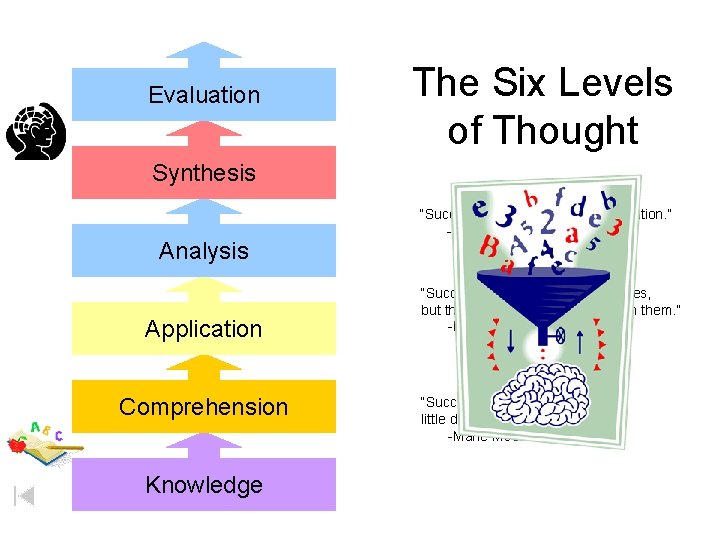
Evaluation The Six Levels of Thought Synthesis Analysis Application Comprehension Knowledge “Success is a journey, not a destination. ” -Ben Sweetland “Successful students make mistakes, but they don’t quit. They learn from them. ” -Ralph Burns “Success consist of a series of little daily efforts. ” -Marie Mc. Cuillough
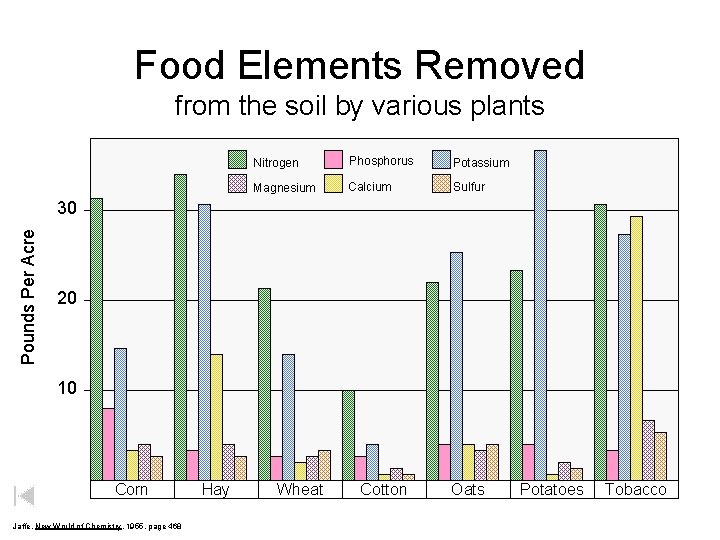
Food Elements Removed from the soil by various plants Nitrogen Phosphorus Potassium Magnesium Calcium Sulfur Pounds Per Acre 30 20 10 Corn Jaffe, New World of Chemistry, 1955, page 468 Hay Wheat Cotton Oats Potatoes Tobacco
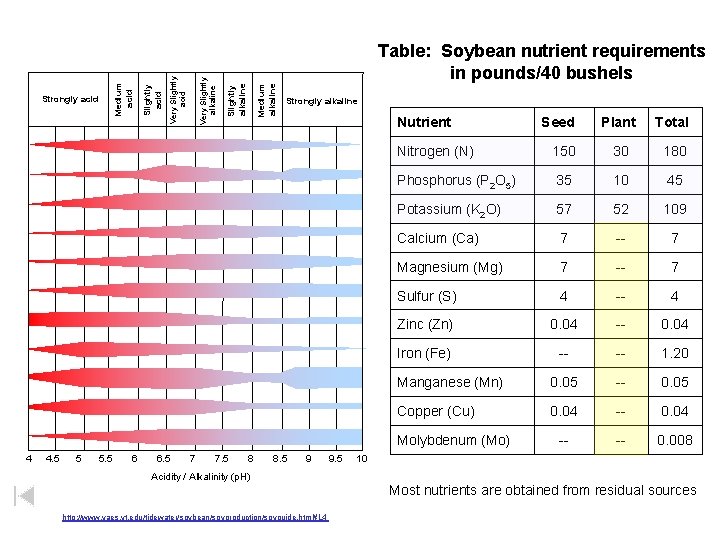
Medium alkaline Slightly alkaline Very Slightly acid Medium acid Strongly acid Table: Soybean nutrient requirements in pounds/40 bushels Strongly alkaline Nutrient 4. 5 5 5. 5 6 6. 5 7 7. 5 8 8. 5 9 Acidity / Alkalinity (p. H) http: //www. vaes. vt. edu/tidewater/soybean/soyproduction/soyguide. html#L 4 9. 5 Plant Total Nitrogen (N) 150 30 180 Phosphorus (P 2 O 5) 35 10 45 Potassium (K 2 O) 57 52 109 Calcium (Ca) 7 -- 7 Magnesium (Mg) 7 -- 7 Sulfur (S) 4 -- 4 Zinc (Zn) 0. 04 -- 0. 04 Iron (Fe) -- -- 1. 20 Manganese (Mn) 0. 05 -- 0. 05 Copper (Cu) 0. 04 -- -- 0. 008 Molybdenum (Mo) 4 Seed 10 Most nutrients are obtained from residual sources

Job Skills for the Future • • • Evaluate and Analyze Think Critically Solve Math Problems Organize and Use References Synthesize Ideas Apply Ideas to New Areas Be Creative Make Decisions with Incomplete Information Communicate in Many Modes Chemistry will develop ALL of these skills in YOU!

You’ve Finally Met Your Match

A Description of a Burning Candle A photograph of a burning candle is shown 1 in the upper right corner. The candle is cylindrical 2 and has a diameter 3 of about 3 cm. The length of the candle was initially about 16 centimeters 4, and it changed slowly 5 during observation, decreasing about 1 cm in one hour 6. The candle is made of a translucent 7, white 8 solid 9 which has a slight odor 10 and no taste 11. It is soft enough to be scratched with the fingernail 12. There is a wick 13 which extends from top to bottom 14 of the candle along its central axis 15 and protrudes about 5 mm above the top of the candle 16. The wick is made of three strands of string braided together 17. A candle is lit by holding a source of flame close to the wick for a few seconds. Thereafter the source of flame can be removed and the flame sustains itself at the wick 18. The burning candle makes no sound 19. While burning, the body of the candle remains cool to the touch 20 except near the top. Within about 1. 5 cm of the top the candle is warm 21 (but not hot) and sufficiently soft to mold easily 22. The flame flickers in response to air currents 23 and tends to become quite smoky while flickering 24. In the absence of air currents, the flame is of the form shown in the photograph, though it retains some movement at all times 25. The flame begins about 2 mm above the top of the candle 26, and at its base the flame has a blue tint 27. Immediately around the wick in a region about 2 mm wide and extending about 5 mm above the top of the wick 28 the flame is dark 29. This dark region is roughly conical in shape 30. Around this zone and extending about 1 cm above the dark zone is a region which emits yellow light 31, bright but not blinding 32. The flame has rather sharply defined sides 33 but a ragged top 34. The wick is white where it emerges from the candle 35, but from the base of the flame to the end of the wick 36 it is black, appearing burnt, except for the last 0, 5 cm, where it glows red 37. The wick curls over about 3 mm from its end 38. As the candle becomes shorter, the wick shortens too, so as to extend roughly a constant length above the top of the candle 39. Heat is emitted by the flame 40, enough so that it becomes uncomfortable in 10 to 20 seconds if one holds his finger 10 cm to the side of the quiet flame 41 or 10 – 12 cm above the flame 42. O’Connor Davis, Mac. Nab, Mc. Clellan, CHEMISTRY Experiments and Principles 1982, page 462,
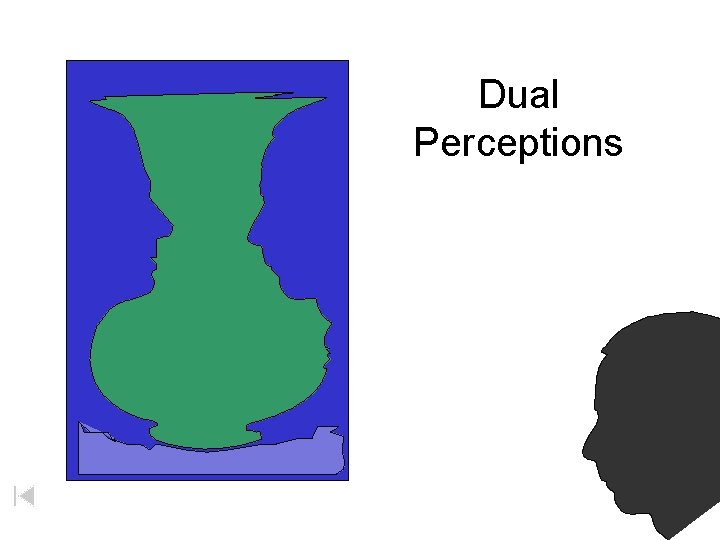
Dual Perceptions
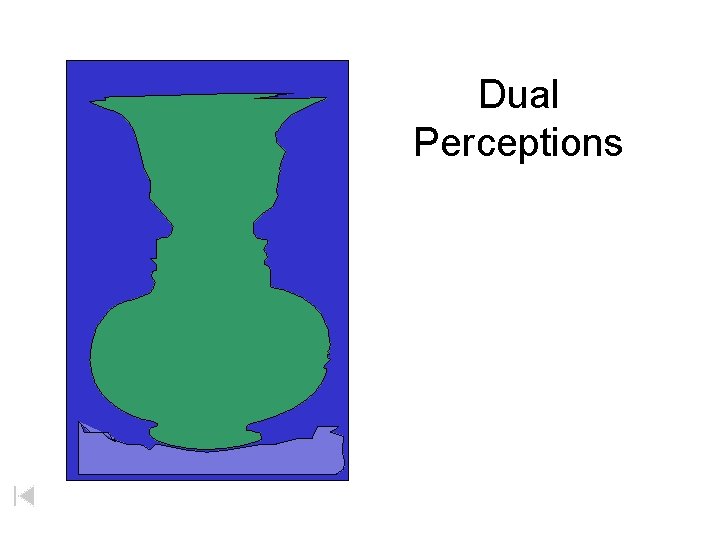
Dual Perceptions
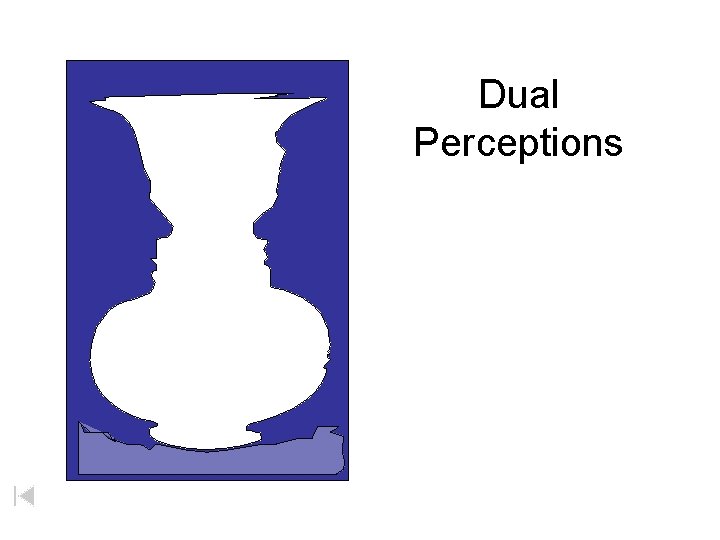
Dual Perceptions
Stack of Blocks




Click to see VIDEO A Colorful Demonstration: The Remsen Reaction
- Slides: 24