Introduction to Chemistry Rena F Abbasova Origins of

Introduction to Chemistry Rena F. Abbasova

Origins of the term Chemistry came from the following: • Arab Latinized word “Keme” meaning “ value ” • Egyptian word “Khemia” meaning “transmutation of earth” • Greek word “Khymeia” meaning “Art of alloying metal” and “Khumeia” meaning “casting together” or “putting together” Jabir Ibn Hayyan /Geber/, considered the "father of chemistry ", introduced a scientific and experimental approach to alchemy.

What is chemistry? • the study of matter and energy and the interactions between them. • It tends to focus on the properties of substances and the interactions between different types of matter, particularly reactions that involve electrons. • the study of substances and their transformation
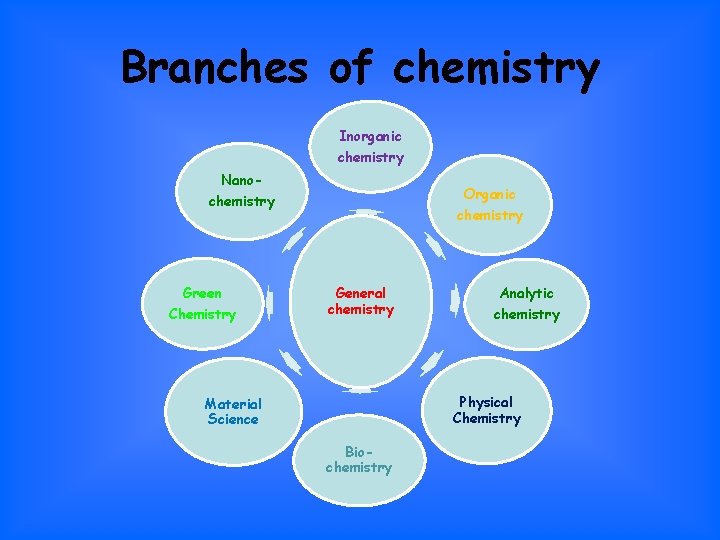
Branches of chemistry Inorganic chemistry Nano- Organic chemistry Green Chemistry chemistry General chemistry Analytic chemistry Physical Chemistry Material Science Biochemistry

Green Chemistry • Use non-toxic substances in inexpensive processes • Create environmentally friendly products ü Catalysts ü Alternative solvents ü Renewable raw materials
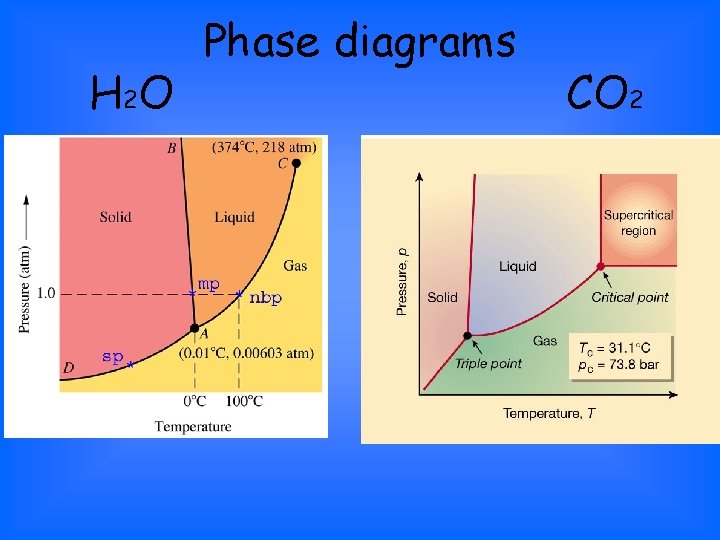
H 2 O Phase diagrams CO 2

Renewable energy alternative energy that comes from resources which are continually replenished such as sunlight, wind, rain, tides, waves and geothermal heat. The wind, Sun, and biomass are three renewable energy sources. Nellis Solar Power Plant, 14 MW power plant installed 2007 in Nevada, USA.

How big is "nano"? Atom: ~0. 1 nanometers. Atoms in a molecule: ~0. 15 nanometers apart. DNA double-helix: ~2 nanometers in diameter. Typical protein: ~10 nanometers long. Typical bacteria: ~200 nanometers long. Human hair: 50, 000– 100, 000 nanometers in diameter. One piece of paper: ~100, 000 nanometers thick. Can you figure out how tall you are in nanometers? Graphene is the basic structural element of some carbon allotropes including graphite, Charcoal , carbon nanotubes and fullerenes. The Nobel Prize in Physics for 2010 was awarded to Andre Geym and Konstantin Novoselov at the University of Manchester.

What is matter? Anything that has mass and occupies space Mixture Substance Matter with constant composition Element Substance made up of only one type of atom Matter with variable composition Compound Heterogeneous Two or more Mixture elements that are Mixtures that are chemically combined made up of more than one phase Homogeneous Mixtures Also called solutions. Mixtures that are made up of only one phase gold, silver, carbon, water, carbon sand, soil, salt water, pure oxygen and dioxide, sodium chicken soup, air, metal alloys, hydrogen bicarbonate, pizza, chocolate seltzer water. carbon monoxide chip cookies.

Physical and chemical changes. What is the difference between chemical and physical change? • Physical change rearranges molecules but doesn't affect their internal structures. • Chemical change is any change that results in the formation of new chemical substances. At the molecular level, chemical change involves making or breaking of bonds between atoms.
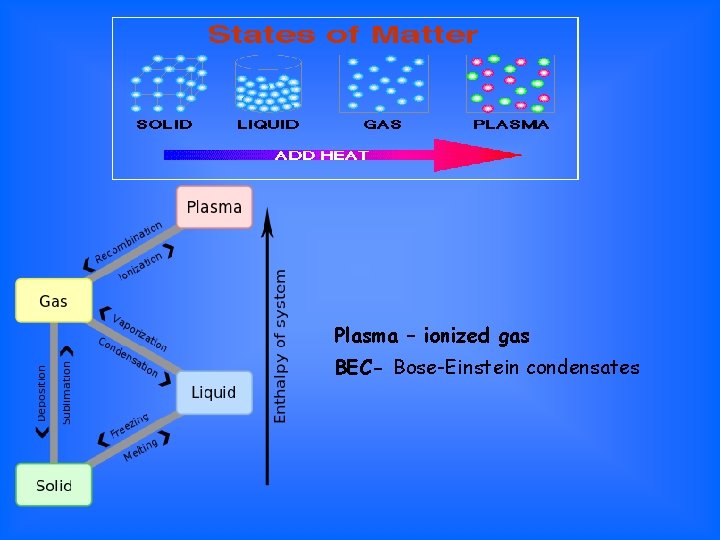
Plasma – ionized gas BEC- Bose-Einstein condensates
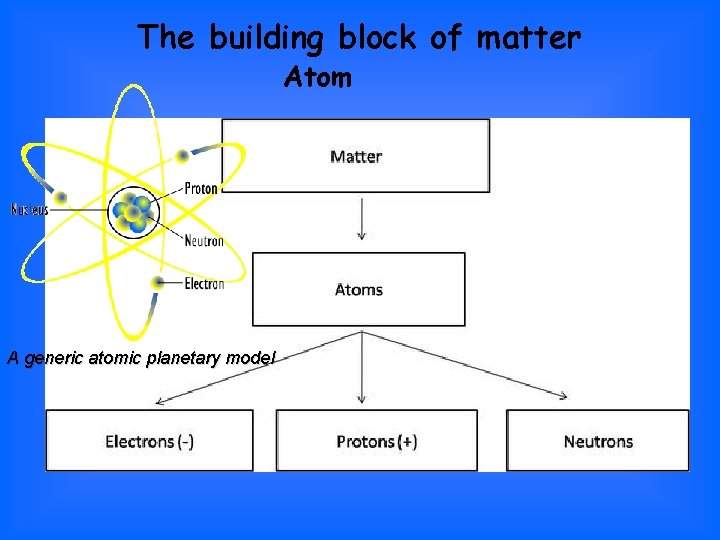
The building block of matter Atom A generic atomic planetary model
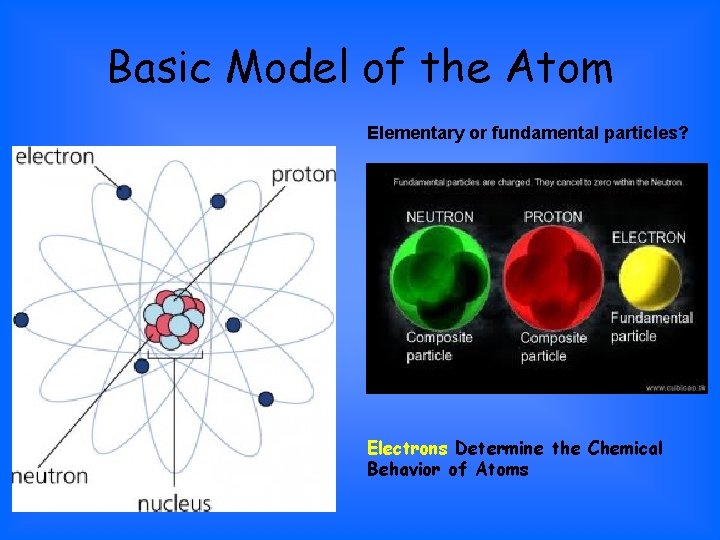
Basic Model of the Atom Elementary or fundamental particles? Electrons Determine the Chemical Behavior of Atoms
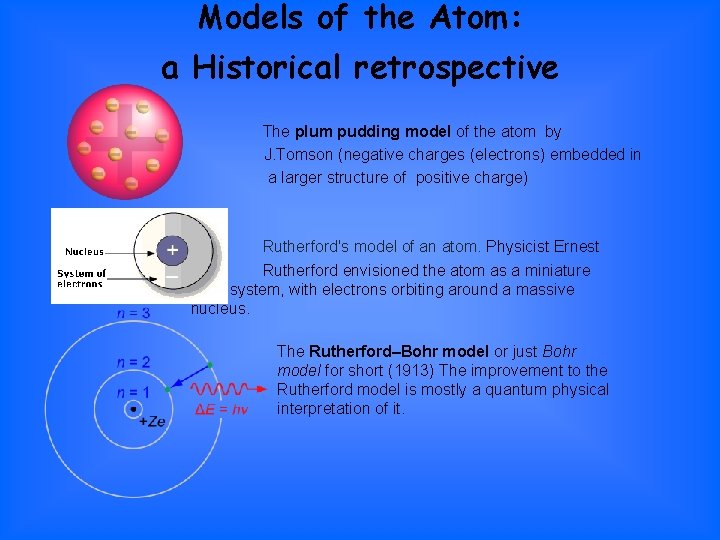
Models of the Atom: a Historical retrospective The plum pudding model of the atom by J. Tomson (negative charges (electrons) embedded in a larger structure of positive charge) Rutherford's model of an atom. Physicist Ernest Rutherford envisioned the atom as a miniature solar system, with electrons orbiting around a massive nucleus. The Rutherford–Bohr model or just Bohr model for short (1913) The improvement to the Rutherford model is mostly a quantum physical interpretation of it.

Chemical Elements Each elements is made of the same kind of atoms Currently, there about 118 elements discovered; some are found in nature and some are man-made. Elements carry the names and symbols. • • Names of the elements are given by its inventor(s). Symbols are the short-hand notations for elements consisting of one or two letters. The first letter is always in upper case and the second one in lower case. A compound is the substance formed by the chemical union of two or more elements in a definite ratio, which is fixed by the nature. Chemical nomenclature is the systematic naming of chemical compounds.
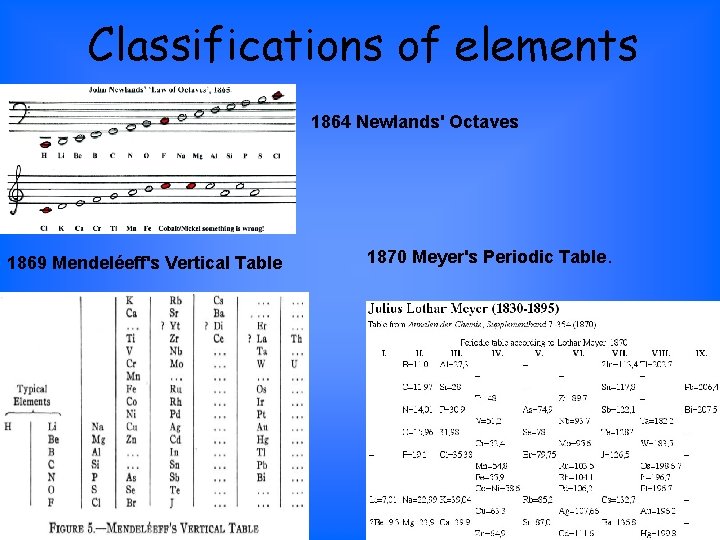
Classifications of elements 1864 Newlands' Octaves 1869 Mendeléeff's Vertical Table 1870 Meyer's Periodic Table.

Chemical laboratory Study of Chemistry includes not only theoretical but also experimental part. Having experimental skills is very important for chemists. Introduction to the laboratory SAFETY PROCEDURES Rules to be followed in labs laboratory equipment - is an important part of chemistry and science in general

Why do we study chemistry? Chemistry helps us to understand the world around us. A basic understanding of chemistry helps you to read and understand product labels. Chemistry can help you make informed decisions. If you understand how chemistry works you’ll be able to separate reasonable expectations from pure fiction. A command of chemistry can help keep you safe! You’ll know which household chemicals are dangerous to keep together or mix and which can be used safely.

ROLE OF CHEMISTRY IN SOCIETY AND INDUSTRY • Industry depends on chemistry. • Many professions and occupations need some knowledge of chemistry. • The application of theories in chemistry produces new and better materials. • Everyday experiences at homes and the community are all explained by chemistry.

Careers in Chemistry can help explain the natural world, prepare people for career opportunities, and produce informed citizens. . • Careers in Industry Many industries are chemical-based like food, petroleum, cosmetic, pharmaceutical, biotechnology, etc. • Career in Commerce A chemistry degree is a good training to acquire skills in mathematics, problem solving, communication, research, self-manage, etc.
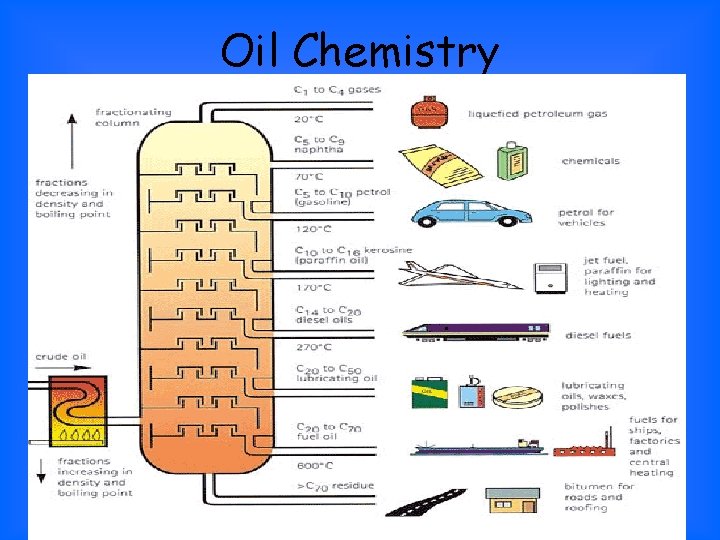
Oil Chemistry

Thank you for attention
- Slides: 23