INTRODUCTION TO CHEMISTRY LAB SAFETY MATTER Chapters 1

INTRODUCTION TO CHEMISTRY LAB SAFETY MATTER Chapters 1 -2

SAFETY MATTERS!!!

Questions: 1. List 3 unsafe activities shown in the illustration and explain why each is unsafe. 2. List 3 correct lab procedures depicted in the illustration. 3. What should Bob do after the accident? 4. What should Sue have done to avoid an accident? 5. Compare Luke and Duke's lab techniques. Who is following the rules? 6. What are three things shown in the lab that should not be there? 7. Compare Joe and Carl's lab techniques. Who is doing it the correct way? 8. What will happen to Ray and Tim when the teacher catches them? 9. List three items in the illustration that are there for the safety of the students in the lab.

What is wrong in this picture?


The point is to follow the safety rules in the lab or this could be you! A disregard for safety rules will not be tolerated!

What is Chemistry? Chemistry: study of the composition of matter & the changes that matter undergoes. Matter: anything that has volume and mass. What is NOT matter? Light, Heat, Radio waves, Magnetic fields

Scientific Method – A systematic approach for learning about the world 1) Observe – an event or process. State the problem and gather background info. 2) Form a Hypothesis – suggested explanation for observed events. 3) Perform Experiments - to test the hypothesis.

The Experiment Variables: factors that may change. Independent Variable (Manipulated Variable) Change only ONE variable! Ex. Amount of Fertilizer Dependent Variable (Responding Variable) What you measure during the experiment. Ex. How high the plant grows Controlled Variables: everything else that must be held constant (so they don’t influence the results). Ex. amount of water, amount of sunlight, type of soil

The Experiment (cont’d) Control Setup – Exactly like the experimental setup except it DOES NOT contain the variable of interest. Used for comparison Multiple Trials – To ensure accuracy

Scientific Method (cont’d) 4) Record & Analyze Data - Data – Recorded observations & measurements - Create a data table and/or graph - Analyze – make calculations, use statistics 5) Draw Conclusions - Create a theory to explain your results

Hypotheses, Theories, Laws Hypothesis: testable prediction to explain observations If I drop 2 objects with different weights, will they hit the ground at the same time or different times? Scientific Law: a fact of nature that is observed so often it becomes truth. Makes predictions about what will happen. The sun will rise every morning. But does not explain why something happens… Theory: an explanation based on many observations & experiments. They answer the question “why”. Why does the sun rise? (Heliocentric Theory – Copernicus)

Scientific Measurements Base Units: Mass – kilogram (gram) Length – meter Temperature – Kelvin (Celsius) Time – second q Derived Units: Consist of a combination of base units q Volume – l x w x h liter q Density – mass/vol g/m. L

Density = mass volume Units: g/m. L or g/cm 3 Why is it important? It can help to identify an unknown substance See reference tables.

Measurement Prefixes – Allow scientists to easily convert and represent different magnitudes of a number. (Based on “ 10”. ) kilo, hecto, deka, base, deci, centi, milli k h D d c m “King Henry died by drinking chocolate milk. ”

Conversions to MEMORIZE 100 centi = 1 base 1000 milli = 1 base 1000 base = 1 kilo

Measurements All measurements MUST HAVE: A number (magnitude) A unit (label) Ex. 25 mg Note: Read a graduated cylinder from the BOTTOM of the meniscus!

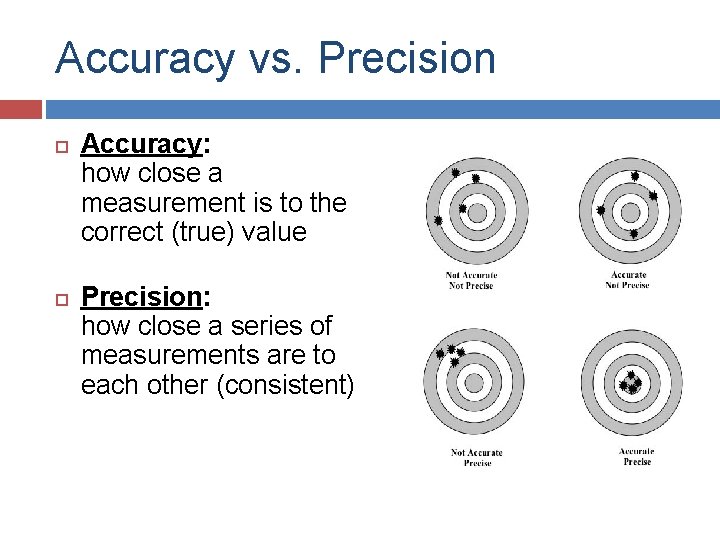
Accuracy vs. Precision Accuracy: how close a measurement is to the correct (true) value Precision: how close a series of measurements are to each other (consistent)

Percent Error – A way of expressing accuracy = | theoretical – experimental | x 100% theoretical * Theoretical Value – correct value based on reliable references * Experimental value – value measured in the lab

Percent Error Example In lab, you measure the mass of your sample to be 6. 2 g. Reliable sources indicate that the actual mass of the sample should have been 8. 3 g. What was your percent error? |6. 2 – 8. 3| x 100 = 25. 3% error 8. 3

Scientific Notation Scientific Notation – used to express very large or very small numbers. Format (# btwn 1 & 9. 9) x 10 exponent Example: 2. 6 x 1023 A positive exponent represents a number greater than one. A negative exponent represents a number less than one.

Scientific Notation Practice Ans. Convert each of the following numbers to standard notation. a. 3. 4 x 105 340, 000 b. 2. 8 x 103 2800 c. 6. 1 x 10 -2 0. 061 d. 7. 8 x 10 -4 0. 00078 Convert each of the following numbers to scientific notation. a. 5600 5. 6 x 103 b. 0. 00921 9. 21 x 10 -3 c. 980, 000 9. 8 x 105 d. 0. 0000073 7. 3 x 10 -6
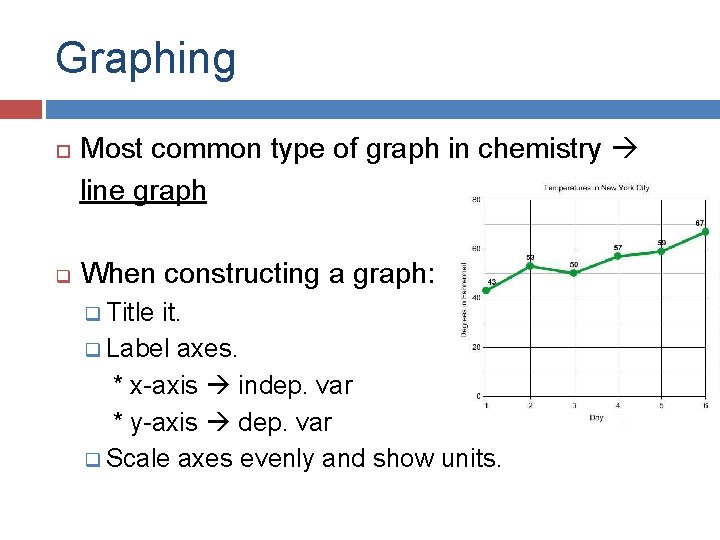
Graphing q Most common type of graph in chemistry line graph When constructing a graph: q Title it. q Label axes. * x-axis indep. var * y-axis dep. var q Scale axes evenly and show units.

Graphing (cont’d) Interpreting relationships from graphs: Direct Proportions – both variables increase or both decrease Indirect Proportions – one variable increases as the other decreases
- Slides: 25