Introduction to Chemistry 1 What is Chemistry The

















- Slides: 20

Introduction to Chemistry 1

What is Chemistry? • The study of matter – Composition (makeup) – what kinds and what numbers of particles are present – Properties (characteristics) – those features of matter that are used to identify a substance – The ways in which properties are related to composition – How one kind of matter interacts with another kind of matter (chemical reactions) 2

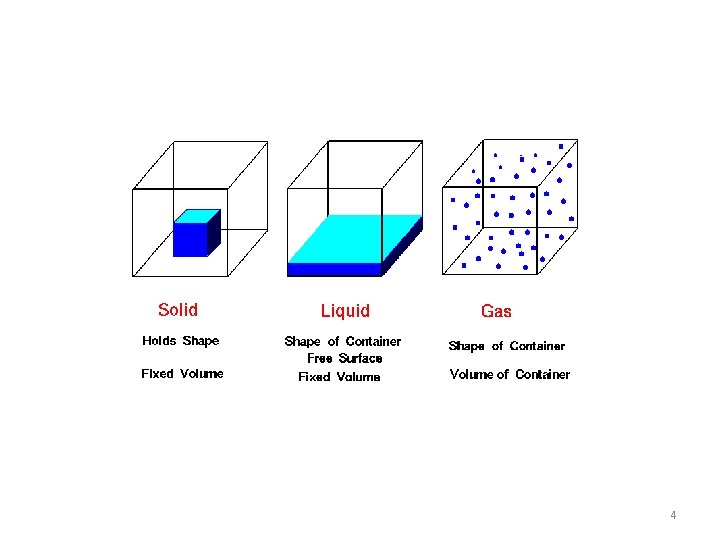
What is Matter? • Anything that has mass and takes up space (volume) PHASES OR STATES OF MATTER 3 most common states at room temperature are: • Solids – definite shape and volume Particles tightly packed together • Liquids – definite volume, no definite shape Particles can change position (slide past each other) • Gases – no definite shape or volume Particles are much farther apart than in solids or liquids 3

4

Properties of Substances • PHYSICAL PROPERTIES Observed without changing the chemical identity of a substance – Examples: color, smell, crystalline or geometric shape, density, melting point, boiling point, conductivity, solubility • CHEMICAL PROPERTIES Observed when the substance takes part in a chemical reaction (a change that converts it to a new substance) – Examples: toxicity, flammability, chemical stability, reactivity 5

Changes in Matter • PHYSICAL CHANGE Does not produce new substances – Examples: crumpling a sheet of paper, melting an ice cube, casting silver in a mold, breaking a bottle, cutting an apple in half • CHEMICAL CHANGE Produces new substances – Examples: fireworks exploding, food rotting, autumn leaves changing color, wood burning, metal rusting 6

How Do I Know Which Change Is Happening? A physical change can be reversed to recover the original matter; a chemical change cannot. 7

Some evidence of a physical change: – change in shape – change in state (solid, liquid, or gas) – change in size Some evidence of a chemical change: – change in odor – change in color – change in temperature (production or loss of heat) – light, heat, or sound given off – formation of gas (bubbles in liquid) 8

Example Physical vs. Chemical Change Physical Change: dissolving sugar in water If you evaporate the water (reaction reversed), you will get the sugar back Chemical Change: heating sugar to make caramel When the mixture cools down (reaction reversed), you get cold caramel but you do not get the sugar back 9

Questions? Comments? Concerns? 10

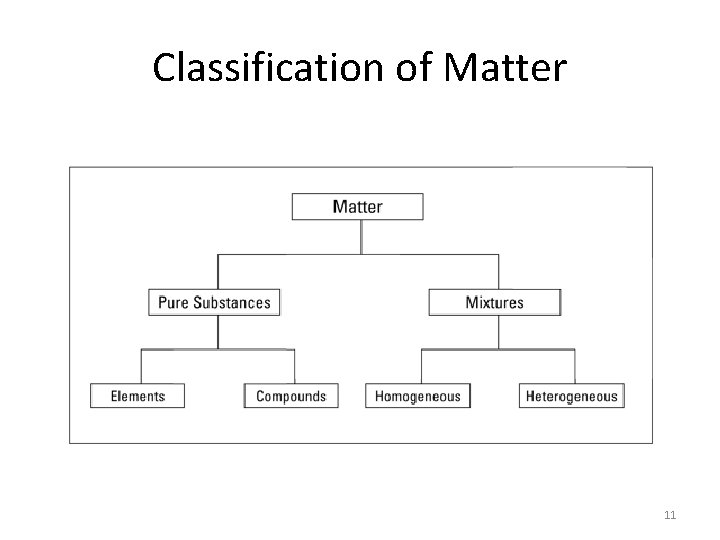
Classification of Matter 11

Element • A pure material that cannot be broken down or changed into a simpler substance by physical or chemical means • The simplest kind of matter • The basic building block of matter • There are > 100 elements (118) Examples: carbon, hydrogen, oxygen, gold, mercury, plutonium, etc, etc … 12

Element Facto-Bonanza! • The rarest naturally-occurring element in the earth's crust may be astatine. The entire crust appears to contain about 28 g of the element. • Although oxygen gas is colorless, the liquid and solid forms of oxygen are blue. • Approximately 20% of the oxygen in the atmosphere was produced by the Amazon rainforest. • The human body contains enough carbon to provide 'lead' (which is really graphite) for about 9, 000 pencils. • Although you may consider gold to be rare, there is enough gold in the Earth's crust to cover the land surface of the planet knee-deep. • Californium is often called the most expensive substance in the world (as much as $68 million for one gram). • Aluminum is the most common metal in the Earth's crust (8 percent of its weight). • Gallium is a metal that melts on palm of the hand. • The only elements that are liquid at room temperature are bromine and mercury. 13

More to come on elements (classification, Periodic Table, nomenclature) 14

Compound • A substance made up of 2 or more elements chemically combined • Has its own properties that are very different from the elements that make it up Example: water vs. hydrogen and oxygen gases • Formed as a result of a chemical change • Can be broken down into its components by chemical means • Made up of a fixed ratio of atoms 15
• There approximately 10 million known compounds • Approximately 100, 000 new compounds are developed and discovered every year • Examples: water (H 2 O), table salt (Na. Cl), carbon dioxide (CO 2), ammonia (NH 3), sugar (C 12 H 22 O 11), etc, etc … 16

Mixture • Contains 2 or more substances that have been combined or blended but not chemically changed • Substances in a mixture keep their own properties • Substances in a mixture can be present in any amount • Mixture can be separated by physical means (evaporating, filtering, centrifuging, etc. ) 17

Homogeneous Mixture • The mixture has the same composition throughout • Involves a single phase • Also referred to as a solution • Examples: glass, filtered sea water, air, tea, steel (carbon & iron) 18

Heterogeneous Mixture • The mixture does not blend smoothly throughout • Individual substances remain distinct • Often involves 2 phases (solid & liquid, etc. ) • Examples: chocolate chip ice cream, some salad dressings, pulpy orange juice, beach sand, unfiltered sea water 19

Still OK? 20