Introduction to Chem II Instructors Course Objectives Course

Introduction to Chem II • • Instructors Course Objectives Course Topics Laboratory Exercises Course Website Today’s Agenda Syllabus

Course Objectives • Review some familiar topics • Investigate some of these topics at a more in-depth level • Model sound pedagogy • Obtain hand-on practice with Venier Data Collection • Show some effective demonstrations

Course Topics • • Stoichiometry Calorimetry Equilibrium Solubility Acid-base chemistry Redox chemistry Thermochemistry

5 Lab Exercises • A calorimetry experiment using a temperature probe • Solubility using a Ca ion selective electrode • Equilibrium constant using a Colorimeter • Acid-base titration • Ag Ion Indicator electrode

Course website http: //alpha. chem. umb. edu/chemistry/bpschem. II/ Syllabus Lab experiments Course notes Homework solutions

Today’s Agenda • • Take a 2 hr exam Paperwork, surveys Lunch Lecture; g/mol, Classification of reactions, Stoichiometry, LR, Energetics of Reactions • Lab Lecture; Calorimetry • Lab Experiment 1 • Early start on HW

Investigating Stoichiometry using Calorimetry Experiment 1

Heat of Reaction - DH • At constant pressure – most lab experiments • a. A + b. B → products DH/mol A • . DH = q (heat produced or absorbed)

Calorimetry • Method of measuring the heat of reaction • Calorimeter-coffee cup • q = cm. DT – c is the specific heat [J/(g ºC)] of solution – m = mass of solution –. DT is change in temperature • . DT is directly proportional to the heat of reaction

The experiment • Mix reactants in different molar ratios • Predict the stoichiometry of the reaction from the ratio that gives the maximum temperature increase
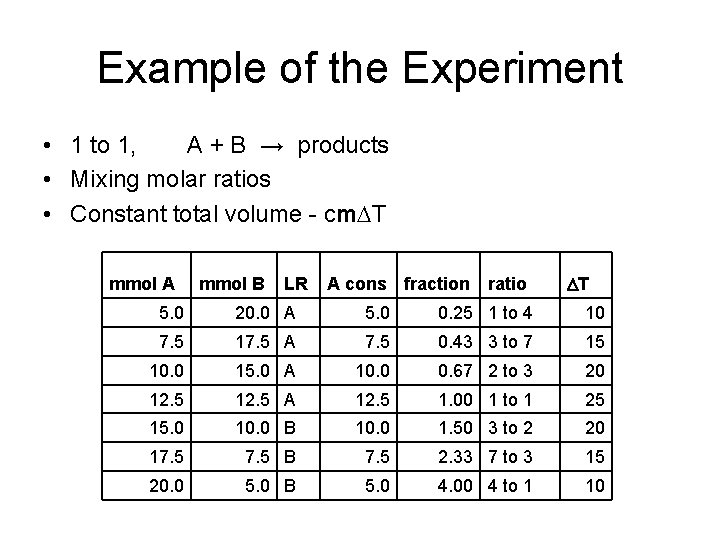
Example of the Experiment • 1 to 1, A + B → products • Mixing molar ratios • Constant total volume - cm. DT mmol A mmol B LR A cons fraction ratio DT 5. 0 20. 0 A 5. 0 0. 25 1 to 4 10 7. 5 17. 5 A 7. 5 0. 43 3 to 7 15 10. 0 15. 0 A 10. 0 0. 67 2 to 3 20 12. 5 A 12. 5 1. 00 1 to 1 25 15. 0 10. 0 B 10. 0 1. 50 3 to 2 20 17. 5 B 7. 5 2. 33 7 to 3 15 20. 0 5. 0 B 5. 0 4. 00 4 to 1 10
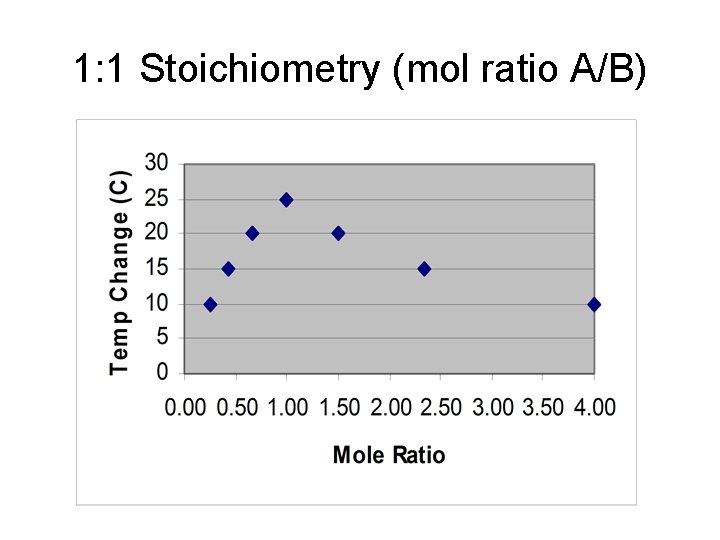
1: 1 Stoichiometry (mol ratio A/B)
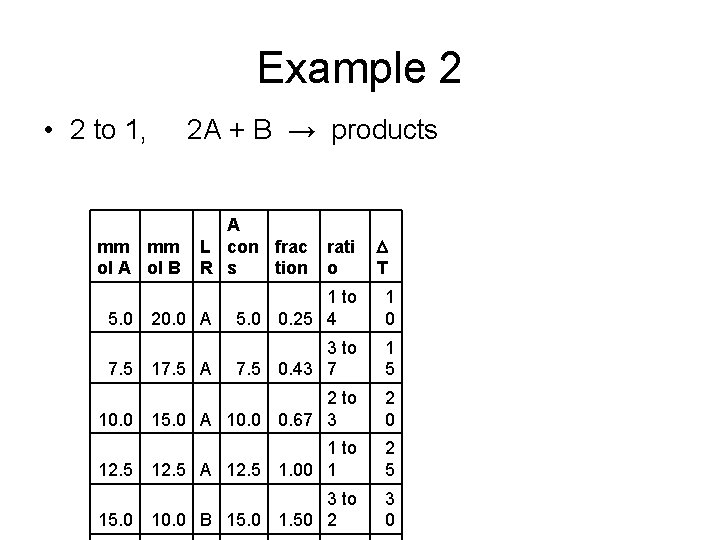
Example 2 • 2 to 1, 2 A + B → products mm mm ol A ol B A L con frac R s tion rati o D T 20. 0 A 1 to 5. 0 0. 25 4 1 0 17. 5 A 3 to 7. 5 0. 43 7 1 5 10. 0 2 to 15. 0 A 10. 0 0. 67 3 2 0 12. 5 1 to 12. 5 A 12. 5 1. 00 1 2 5 15. 0 3 to 10. 0 B 15. 0 1. 50 2 3 0 5. 0 7. 5
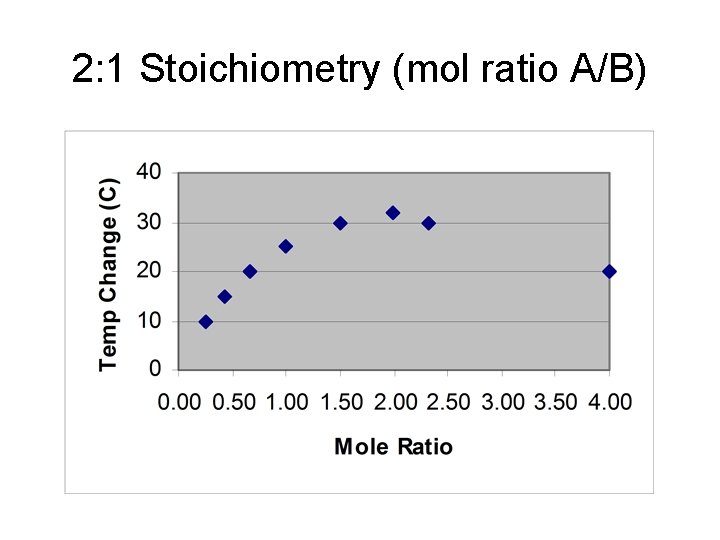
2: 1 Stoichiometry (mol ratio A/B)
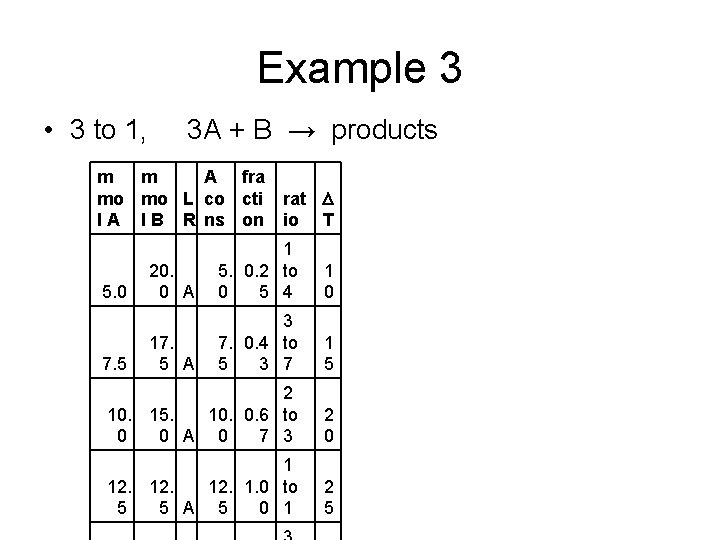
Example 3 • 3 to 1, 3 A + B → products m m A fra mo mo L co cti rat D l A l B R ns on io T 20. 0 A 1 5. 0. 2 to 0 5 4 1 0 17. 5 A 3 7. 0. 4 to 5 3 7 1 5 2 10. 15. 10. 0. 6 to 0 0 A 0 7 3 2 0 1 12. 12. 1. 0 to 5 5 A 5 0 1 2 5 5. 0 7. 5
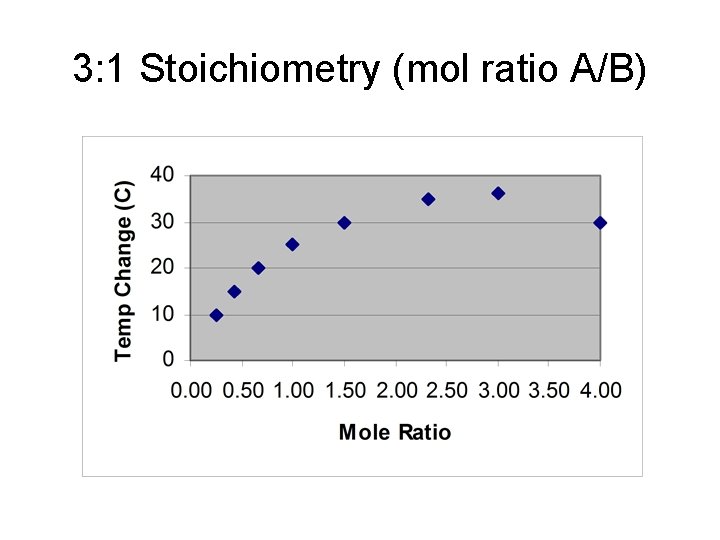
3: 1 Stoichiometry (mol ratio A/B)

Determining the DHm • . DH = cm. DT = (4. 4 J/g. C)*(50 g)*(36) = 7920 J • mol A reacted = 18. 8 mmol A • . DHm = DH/(mol A reacted) = (7920)/(. 0188 mol) = 421276 J/mol = 421 k. J/mol

Products • • Thiosulfate is a classic reducing agent 2 S 2 O 32 - ↔ S 4 O 62 - + 2 e. Cl- is the product of the reduction of OCl. Write a balanced redox equation – Step 1: determine half reactions. – Step 2 Make the reduction half reaction and oxidation half reaction have the same number of electrons by multiply reactions by common denominator – Step 3: Add reactions
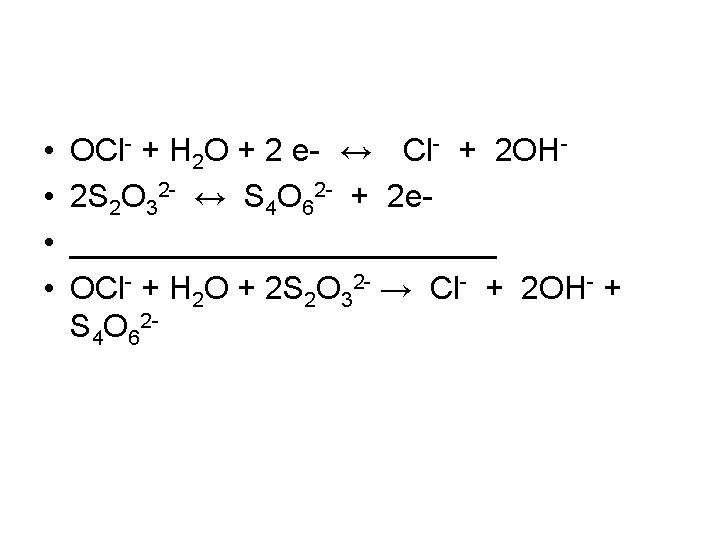
• • OCl- + H 2 O + 2 e- ↔ Cl- + 2 OH 2 S 2 O 32 - ↔ S 4 O 62 - + 2 e____________ OCl- + H 2 O + 2 S 2 O 32 - → Cl- + 2 OH- + S 4 O 62 -

Solubility of Ca. SO 4 Experiment 2

Goals • Determine the solubility of Ca. SO 4 in three different solution – Saturated Ca. SO 4 in H 2 O – Saturated Ca. SO 4 in 0. 10 M KNO 3 – Saturated Ca. SO 4 in 0. 10 M Na 2 SO 4 • Compare and rationalize the results

Major concepts • Solubility Product Constants and saturated solution • Le. Chatlier’s principle and the common ion effect • Effect of ionic strength and ion activities on Ksp • Ion Selective Electrodes

Ksp of Ca. SO 4 • Ca. SO 4(s) ↔ Ca 2+ + SO 42 • Ksp(Ca. SO 4) = [Ca 2+][SO 42 -] = 2. 4∙ 10 -5

Saturated solution in water • Add several grams of Ca. SO 4 to 1 L of water • Shake and mix for weeks • Allow Ca. SO 4 that did not dissociate to settle to bottom • Ksp(Ca. SO 4) = [Ca 2+][SO 42 -] = 2. 4∙ 10 -5 = x 2 [Ca 2+] = 5. 0∙ 10 -3 M

Saturated solution in 0. 10 M Na 2 SO 4 • Add several grams of Ca. SO 4 to 1 L of 0. 10 M Na 2 SO 4 • Common Ion effect • Ksp(Ca. SO 4) = [Ca 2+][SO 42 -] = 2. 4∙ 10 -5 = x(x+0. 10) Assume x <<< 0. 10 x = 2. 4∙ 10 -4 M [Ca 2+] = 2. 4∙ 10 -4 M

Saturated solution in 0. 10 M KNO 3 • Activities • Ksp(Ca. SO 4) = ACa 2+ASO 42 - = [Ca 2+]g. Ca 2+[SO 42 -]g. SO 42 - = 2. 4∙ 10 -5 • Activity coefficient (g) is dependent on the ionic strength of the solution, and the size and charge of the ion. It is a number between 0 and 1. At very low ionic strength, g approaches 1
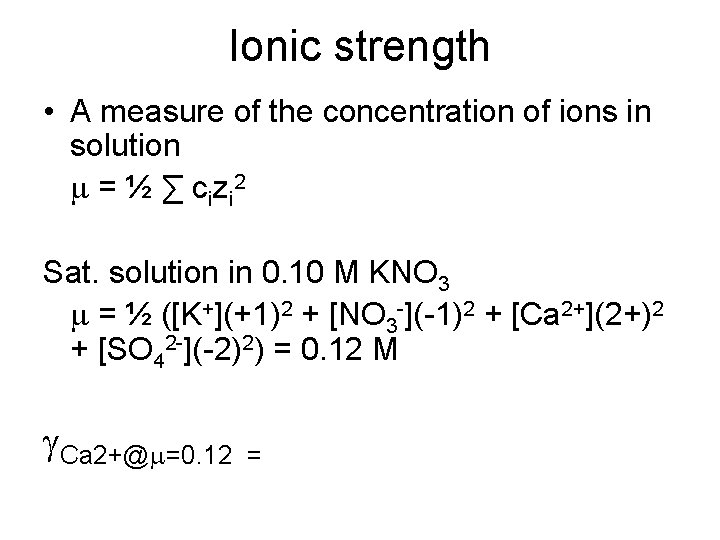
Ionic strength • A measure of the concentration of ions in solution m = ½ ∑ cizi 2 Sat. solution in 0. 10 M KNO 3 m = ½ ([K+](+1)2 + [NO 3 -](-1)2 + [Ca 2+](2+)2 + [SO 42 -](-2)2) = 0. 12 M g. Ca 2+@m=0. 12 =

Take home message • The common ion effect decreases the solubility by over an order of magnitude • At high ionic strengths, solubility increases slightly ( by a factor of 1. 5 -5).

Ion Selective Electrode • A probe that consists of two reference electrodes connected electrically through a specific type of salt bridge through the solution being measured. • The salt bridge is a membrane that specifically binds the ion of interest • A junction potential develops at this membrane that is proportional to the concentration of the ion of interest
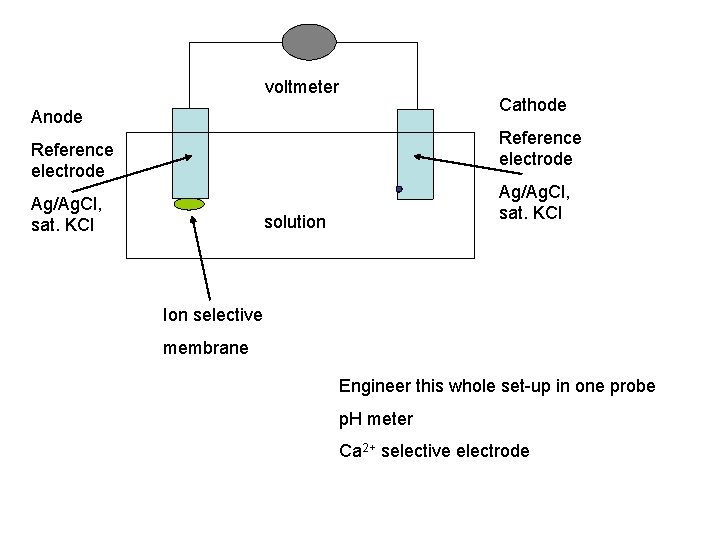
voltmeter Anode Cathode Reference electrode Ag/Ag. Cl, sat. KCl solution Ion selective membrane Engineer this whole set-up in one probe p. H meter Ca 2+ selective electrode
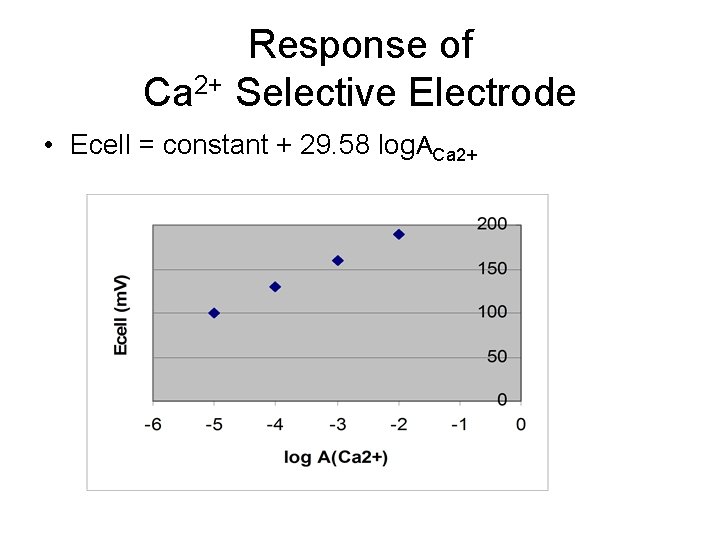
Response of Ca 2+ Selective Electrode • Ecell = constant + 29. 58 log. ACa 2+
- Slides: 31