Introduction to Bonding Topic 13 Essential Question What

Introduction to Bonding Topic #13 Essential Question: What are all the differences and similarities between covalent bonds and ionic bonds?

What is a bond? • A bond can be thought of as a force that holds groups of two or more atoms together and makes them function as a unit • Example : water O H H Bonds require energy to break and release energy when made

Types of bonds • Ionic bonds - typically formed between metals and nonmetals • Covalent bonds - typically formed between nonmetals • Metallic bonds - formed between metals

Ionic Bonds • Ionic bonding results from the transfer of electrons. Then, the opposite charges attract each other. • Ionic bonds are strong

Ionic Bonds • Na and Cl – Na is a metal and likes to lose one electron and form a +1 ion. – Cl is a nonmetal and likes to gain one electron and form a -1 ion. – the final ionic compounds is Na. Cl Na+ + Cl- Na. Cl The electrostatic interaction keeps them together!
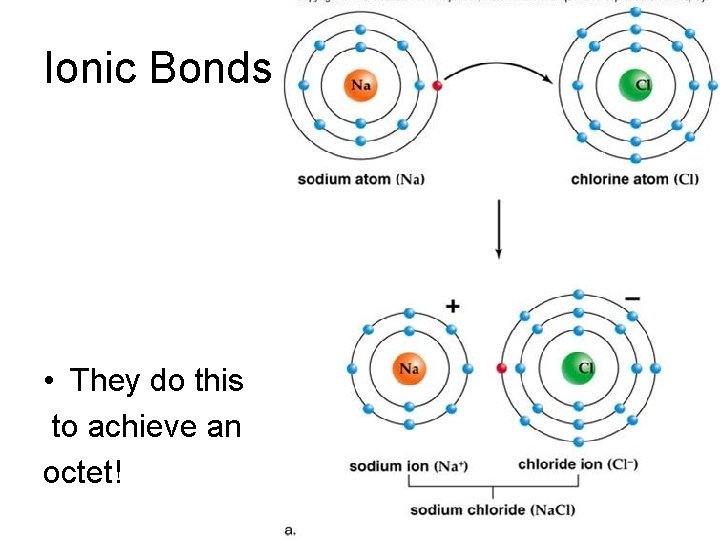
Ionic Bonds • They do this to achieve an octet!

Covalent Bonds • Covalent Bonds – exist between nonmetals bonded together – form when atoms of nonmetals share electrons – electrons can be shared equally or unequally – The unequal sharing results in polar molecules

Metallic Bonds • Metallic bonds exist between metals • Occur when two metals, usually the same metal, are bonded together • “sea of electrons” • “delocalized electrons” • ttp: //www. youtube. com/watch? v=XAn. TCYZP Js. E&feature=bf_next&list=PLBFE 28832 E 57 7 A 62 B

Regents Chemistry • Electronegativity

How can we tell really tell which type of bond we have? • Electronegativity – is the relative ability of an atom in a molecule to attract shared electrons to itself • This tells us what type of bond we have; – Covalent, polar covalent or ionic • Electronegativity values are determined by measuring the polarities of bonds between various elements to determine a specific value for each element

Electronegativity • Electronegativity values for each element are obtained by using the Periodic Table • In fact, there is a general trend in electronegativity we observe in the Periodic Table • Electronegativity values increase across and up the Periodic Table – See table on pg. 332

Electronegativity • We take the difference between the electronegativity values to determine exactly what type of bond exists, in essence the polarity of the bond See table 12. 1

Determining Bond Polarity • If the difference between the electronegativity values is: – 0. 0 – 0. 5: covalent bond (equal sharing) – 0. 6 – 1. 6: polar covalent bond (unequal sharing) – 1. 7 – up: ionic bond (transferring electrons)

Examples • Use your Reference Tables to determine the difference in electronegativity values and the type of bond for each of the following: – H-H – H-Cl – H-O – H-S • • H-F Na. Cl O 2 KBr Worksheet

Regents Chemistry • Intro to valence electrons

Electrons in an atom • Electrons surround the nucleus of an atom in specific energy levels or shells • Each level can hold only a certain amount of electrons • It is an atoms ability to the lose, gain or share electrons from its outer shell that determine its reactivity

The outer shell • The outer shell in an atom contains the valence electrons • Valence electrons can be lost, gained or shared to have eight electrons in the outer shell • Each group on the table tells the number of valence electrons

Periodic Table • Groups 1, 2, 13, 14, 15, 16, 17, 18 have 1, 2, 3, 4, 5, 6, 7, 8 valence electrons, respectively • We will not consider the transition metals • See periodic table

Sharing to reach the Octet Rule • The octet rule states that an atom cannot have more than 8 electrons in its outer shell • Valence electrons are lost, gained or shared with other atoms to attain 8 electrons in the outer shell • Eight valence electrons means a filled and happy shell - like the noble gases

Nonmetals share • Nonmetals share electrons to reach eight valence electrons • Single, double and triple bonds can be formed by sharing electrons

Metals + non-metals = lose/gain e • metals and nonmetals interact by losing and gaining electrons to reach 8 electrons (filled outer shell) • The oxidation states on the periodic table represent this desire to move electrons • ex: K+ want to lose 1 electron to reach noble gas configuration of eight electrons

Lewis structures: your assignment • The reading and problems focus on drawing Lewis structures • Lewis structures are a means to represent bond formation between atoms • Covalent bonded compounds have different Lewis structures than Ionic bonded compounds
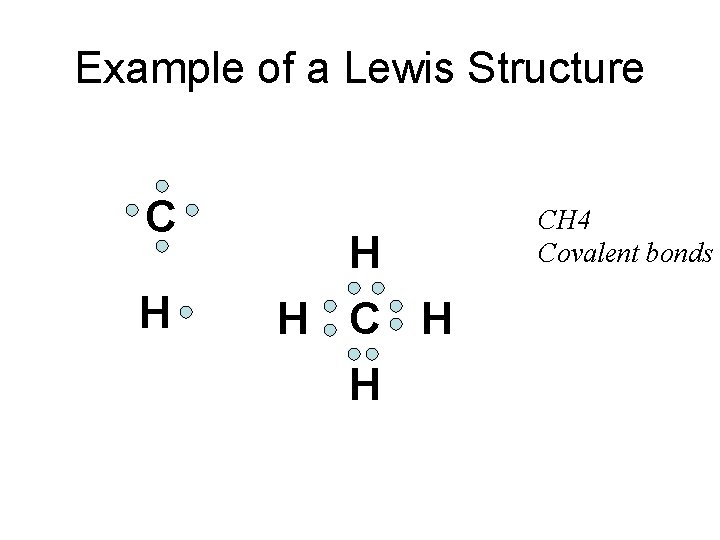
Example of a Lewis Structure C H H H CH 4 Covalent bonds

Regents Chemistry • Lewis Structures

Lewis Structures b The Lewis Structure is a representation of a molecule that shows how the valence electrons are arranged among the atoms in a molecule b We used dots around the elemental symbol to represent the valence electrons C

Single Lewis Structure - Practice b Draw four lone electrons first (if necessary) them pair them up b Draw Lewis Structures for the following atoms Na Be Al Br

Lewis Structures for Ionic Compounds b For Lewis Structures of ionic bonds the atoms are not joined but draw next to each other example: KBr + K [ Potassium loses an electron to achieve the noble gas configuration of Argon Br ] Bromine gains an electron to achieve the noble gas configuration of Krypton

Lewis Structures – Covalent Bonds b Hydrogen forms stable molecules when it shares two electrons b Two electrons fill Hydrogen’s valence shell b Helium does not form bonds because its valence shell is already filled; it is a noble gas b Second row non-metals Carbon through Fluorine from stable molecules when surrounded by eight electrons – the Octet Rule

Lewis Structures – Covalent Bonds b Valence electrons in covalent bonds can either be bonding pairs, if involved directly in the bond or lone pairs if not involved in the bond

Writing Lewis Structures - Rules b Obtain the total sum of the valence electrons from all of the atoms b Use one pair of electrons to form a bond between each pair of bound atoms. For convenience, a line (instead of a pair of dots) can be used to indicate each pair of bonding electrons b Arrange the electrons to satisfy the duet rule for hydrogen and the octet rule for second row non metals

Lewis Structures – Covalent Bonds b Examples PH 3 H l H– P –H • • Step 1) 8 total valence e- total Step 2) Draw one pair of electrons per bond 8 -6 = 2 left Step 3) Arrange the remaining electrons according to octet rule H H P H

Lewis Structures – Covalent Bond Practice Examples HBr . . H: Br: ·· CF 4 Worksheet

Regents Chemistry – Ionic Lewis Structures – Multiple bonds in Lewis Structures – Polyatomic ion Lewis Structures and Resonance

Lewis Structures for Ionic Compounds b For Lewis Structures of ionic bonds the atoms are not joined but draw next to each other example: KBr + K [ Br ] Potassium loses an electron to achieve the noble gas configuration of Argon Bromine gains an electron to achieve the noble gas configuration of Krypton

Examples of Ionic Lewis Structures b Draw Lewis Structures for the following: Na. Cl Li. Br KI

Multiple Bonds and Lewis Structures…review first b We have seen how to draw Lewis Structures for molecules with single bonds • For example NH 3 1. Sum the total valence e 2. Subtract number of bonding e 3. Place remaining valence e- 8 total valence e 3 bonds x 2 e- = 6 bonding 2 e- left over H N H H
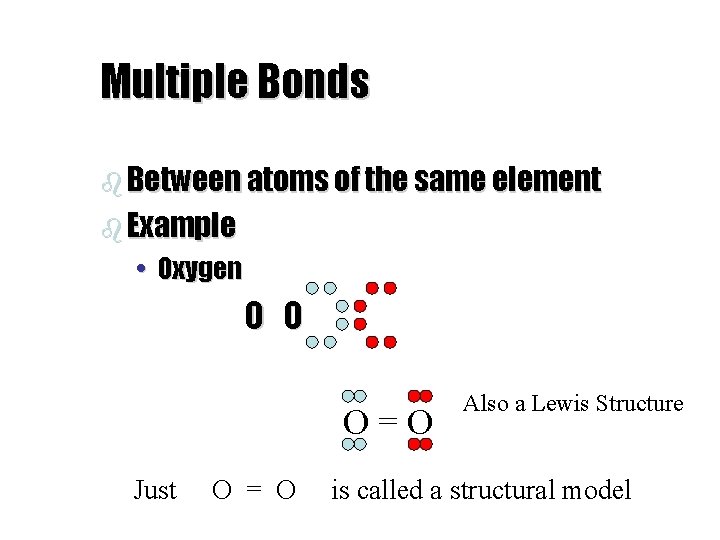
Multiple Bonds b Between atoms of the same element b Example • Oxygen O O O=O Just O = O Also a Lewis Structure is called a structural model

Example of Multiple Bonds Nitrogen N N We now meet the octet rule!
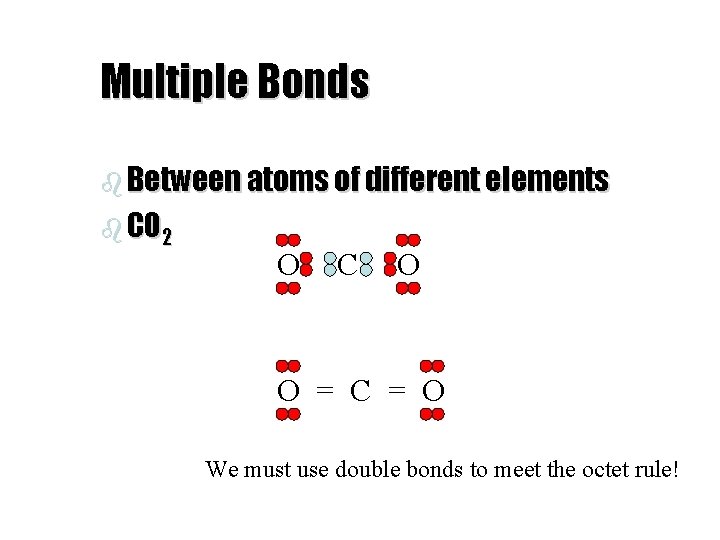
Multiple Bonds b Between atoms of different elements b CO 2 O C O O = C = O We must use double bonds to meet the octet rule!

Lewis Structures for Polyatomic Ions and Resonance Structures b Read pg. 344 (bottom) to 349 and answer questions a-g in example 12. 4 (pg. 347) and a-i in the Self Check exercise 12. 4 (pg. 348)
- Slides: 40