Introduction to Biophysics Lecture 24 Donna Equilibrium Human

Introduction to Biophysics Lecture 24 Donna Equilibrium
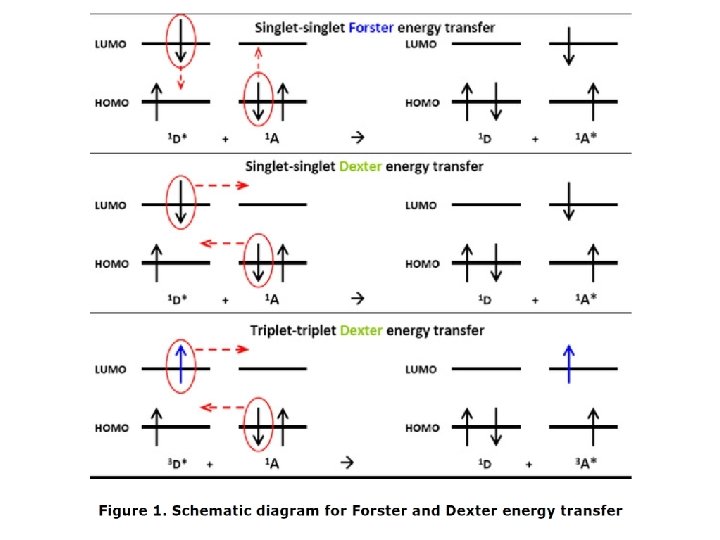
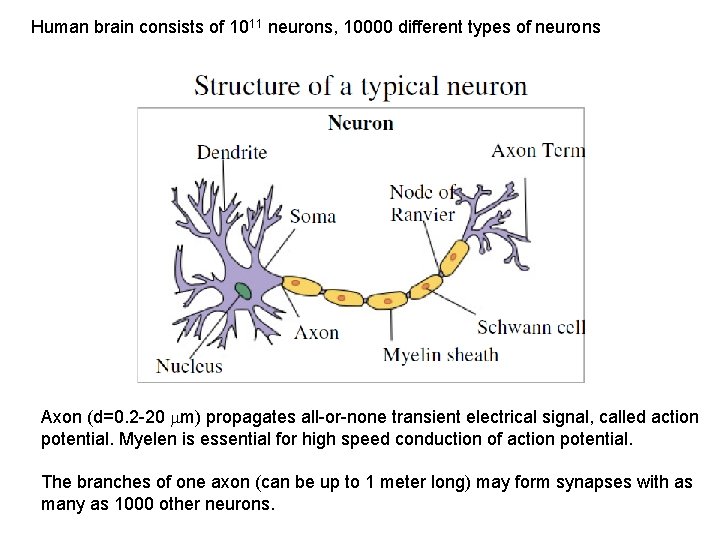
Human brain consists of 1011 neurons, 10000 different types of neurons Axon (d=0. 2 -20 m) propagates all-or-none transient electrical signal, called action potential. Myelen is essential for high speed conduction of action potential. The branches of one axon (can be up to 1 meter long) may form synapses with as many as 1000 other neurons.
Purkinje cell in cerebellum makes about 150000 contacts.

Introducing membrane as electrical structure Membrane: electrically isolating leaflet containing a mosaic of various passive an active electrical devices, controlling membrane potential. Cell membrane has High electrical resistance Low dielectric constant Very thin hydrophobic electrically isolating interface behaving like a capacitor with certain capacity (C) and resistance (R). Thus, it is possible to consider passive electric behavior of the cell as RC-element. Specific capacity: Csp = 0 / x = 10 [m. F/m 2] (1923, H. Fricke) x – membrane thickness, 80Å lipids =3. 5 membrane =9 Csp defines the relation between the amount of charge ( , C/m 2) which is required to generate the membrane potential difference Csp = /
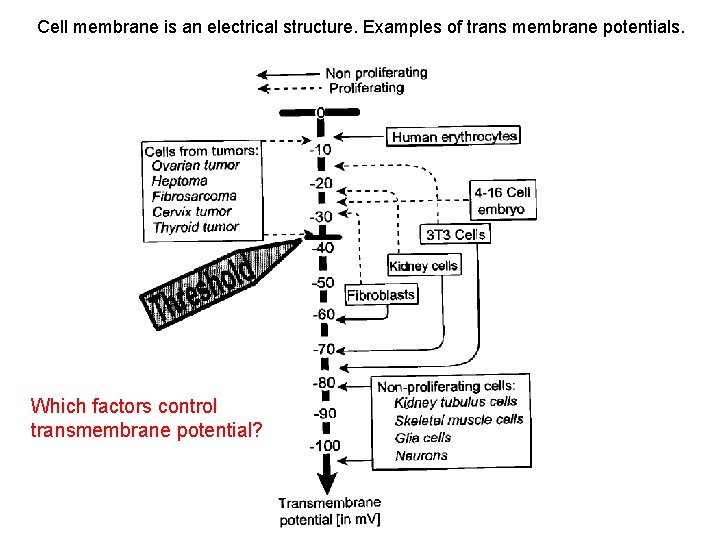
Cell membrane is an electrical structure. Examples of trans membrane potentials. Which factors control transmembrane potential?

Which factors control transmembrane potential? 1. Nernst potential 2. Donnan equilibrium 3. Active ion pumping
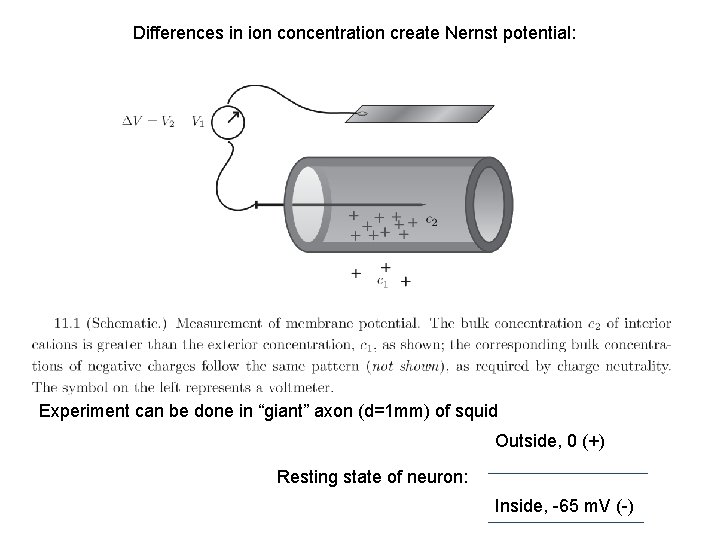
Differences in ion concentration create Nernst potential: Experiment can be done in “giant” axon (d=1 mm) of squid Outside, 0 (+) Resting state of neuron: Inside, -65 m. V (-)

KCl on both sides of the membrane. What if one ion (K+) is more permeable than the other (Cl-)? The layer of net positive charge (excess of K+) will form just outside of the membrane. V = KNernst = -(k. BT/ez)*ln(c 2/c 1) gives the potential arising when permeant species reach equilibrium By analogy with chemical potential (which should be everywhere the same for molecular species equilibrium), the electrochemical potential of permeable ion should be everywhere the same in the equilibrium µi (electrochem) = µi 0 + zi. F F- Faraday constant k -Debye screening length Does Cl- ion obey Nernst eq. in this example?

Real cell: K+ Na+, Cl- and charged macromolecules (125 m. M, electrons) Outside: Na (140 m. M), K (10 m. M), Cl (150 m. M) 1. Electro neutrality outside the cell: C 1, Na + C 1, K – C 1, Cl = 0 C 2 2. Electro neutrality inside the cell: C 2, Na + C 2, K – C 2, Cl + micro/e= 0 3. V – the same electrostatic potential affects every ion species: -(k. BT/ez)*ln(c 2, Na/c 1, Na) = -(k. BT/ez)*ln(c 2, K/c 1, K) =-(k. BT/(-)ez)*ln(c 2, Cl/c 1, Cl) c 1, Na/c 2, Na= c 1, K/c 2, K= c 2, Cl/c 1, Cl Gibbs- Donnan relation Solving: Na (210 m. M), K (15 m. M), Cl (100 m. M) Simply the presence of charged macromolecules results in the permanent (resting) electrostatic potential (about -10 m. V). No energy is required to maintain the Donnan potential Calculate osmotic pressure in Donnan equilibrium
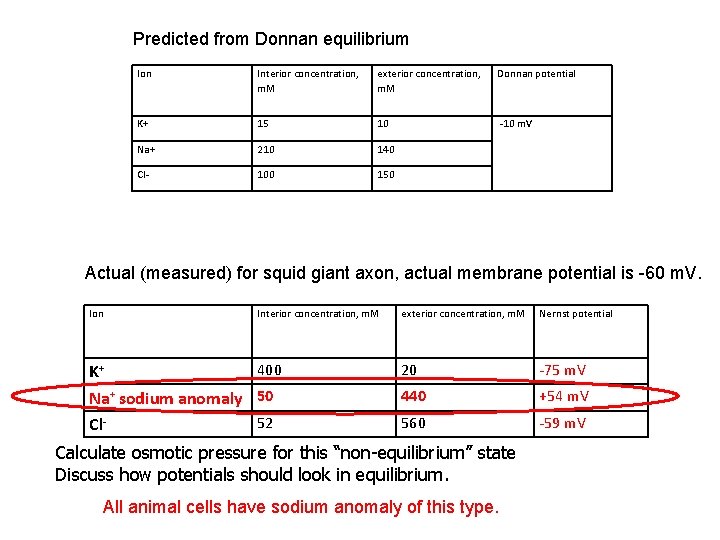
Predicted from Donnan equilibrium Ion Interior concentration, m. M exterior concentration, m. M Donnan potential K+ 15 10 -10 m. V Na+ 210 140 Cl- 100 150 Actual (measured) for squid giant axon, actual membrane potential is -60 m. V. Ion Interior concentration, m. M 400 K+ Na+ sodium anomaly 50 52 Cl- exterior concentration, m. M Nernst potential 20 -75 m. V 440 +54 m. V 560 -59 m. V Calculate osmotic pressure for this “non-equilibrium” state Discuss how potentials should look in equilibrium. All animal cells have sodium anomaly of this type.

Conclusion: ions in living cells are not in equilibrium. What are molecular mechanisms/machines maintaining nonequilibrium ion concentration? What is the associated metabolic cost? How can we shut down metabolism to test this proposal? What happens when Nernst potential of particular ion is far from trans-membrane potential?

Ohmic conductance hypothesis (explores nonequilibrium steady state when Nernst potential is not equal transmembrane potential, good only close to resting conditions): j q, i = zi e ji = ( V - i. Nernst )gi j - (number of ions per unit area) is positive if the net flux is outward gi - conductance per area, 1/m 2 , note it will be different for different ions. How we can find it experimentally?
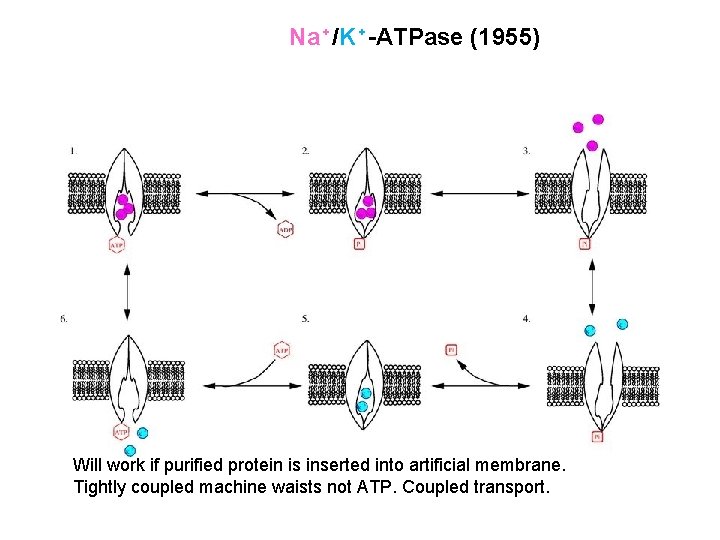
Na⁺/K⁺-ATPase (1955) Will work if purified protein is inserted into artificial membrane. Tightly coupled machine waists not ATP. Coupled transport.

Suggested reading: Glaser 2. 5. 4. 2. 5. 5 Nelson 7. 4, 11. 1 Homework: your turn 11 A, page 473
- Slides: 15