Introduction to Biophysics Lecture 22 Electrostatics Nernst equation

Introduction to Biophysics Lecture 22 Electrostatics, Nernst equation
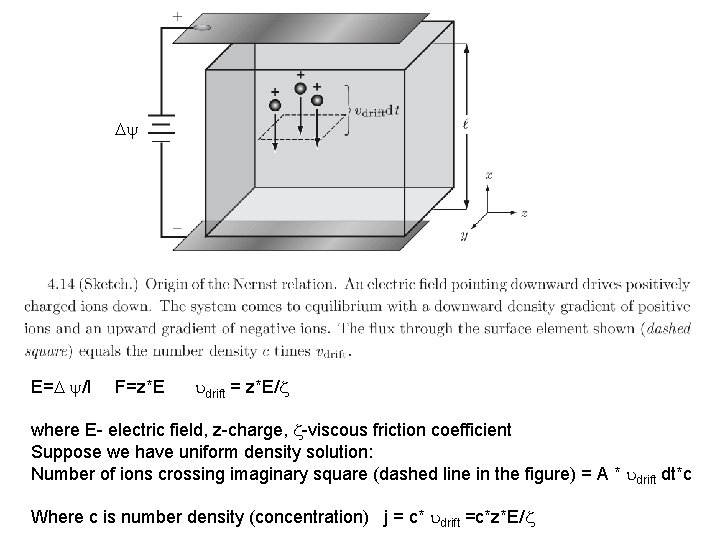
E= /l F=z*E drift = z*E/ where E- electric field, z-charge, -viscous friction coefficient Suppose we have uniform density solution: Number of ions crossing imaginary square (dashed line in the figure) = A * drift dt*c Where c is number density (concentration) j = c* drift =c*z*E/

Electric field creates non-uniform density of ions j(x) = c*z*E/ - D*dc/dx = D (z. E*c/ k. BT -dc/dx) Using Einstein relation D* =k. BT What electric field is needed to get zero net flux (to cancel diffusive tendency to erase non-uniformity)? 1/c*(dc/dx) = z. E/ k. BT d(lnc)/dx = z. E/ k. BT Integrate from l =0 to l (bottom to top) and use (-d. U/dx = z. E) thus U=-q. Ex potential energy per unit charge: = bot – top = -E*l ln(cbot) – ln(c top) = -z* eq/ k. BT (ln(c)) = -z* eq/ k. BT – Nernst relation Discuss why D drops out.

Calculate eq caused by difference in concentration of Na+ ion 10 times.
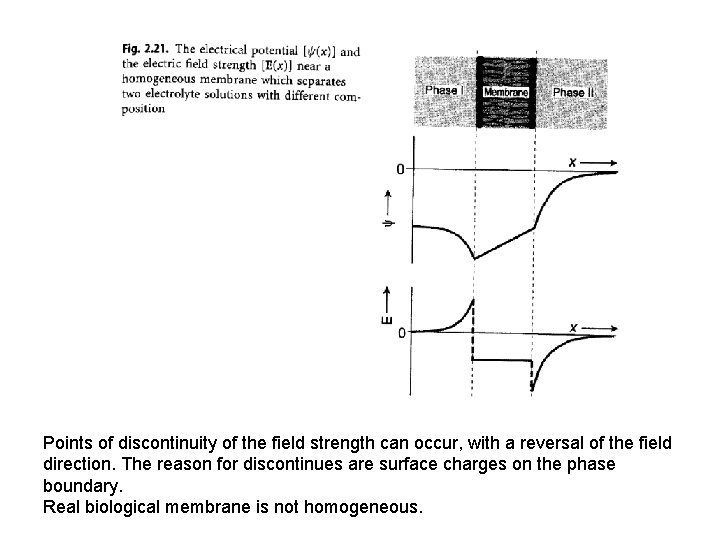
Points of discontinuity of the field strength can occur, with a reversal of the field direction. The reason for discontinues are surface charges on the phase boundary. Real biological membrane is not homogeneous.

Homework: Read Nelson 4. 6. 3

Suggested reading: Nelson 7. 4 Glaser 2. 4. 1. and 2. 4. 3 Calculate the Debye-Hückel radius (1/ ) for Ringer’s physiological solution which has following composition: 105 m. M Na. Cl, 5 m. M KCl, 25 m. M Na 2 HPO 4, 2 m. M Ca. Cl 2)
- Slides: 7