Introduction to Biophysics Lecture 18 Aggregation and bending

Introduction to Biophysics Lecture 18 Aggregation and bending
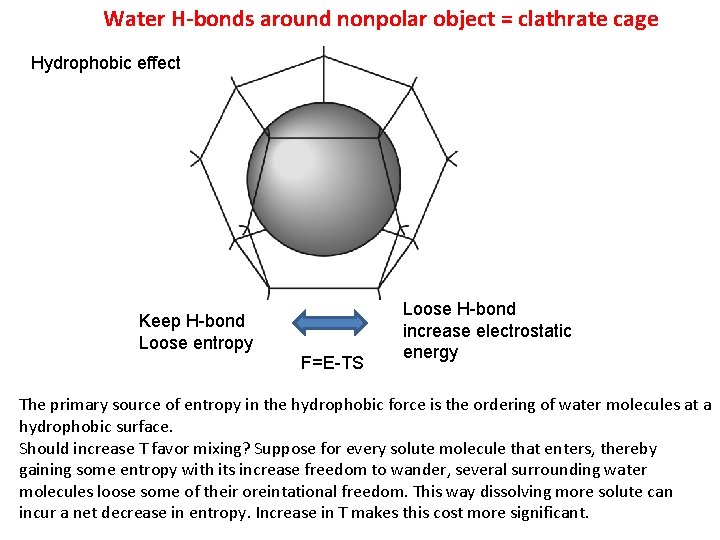
Water H-bonds around nonpolar object = clathrate cage Hydrophobic effect Keep H-bond Loose entropy F=E-TS Loose H-bond increase electrostatic energy The primary source of entropy in the hydrophobic force is the ordering of water molecules at a hydrophobic surface. Should increase T favor mixing? Suppose for every solute molecule that enters, thereby gaining some entropy with its increase freedom to wander, several surrounding water molecules loose some of their oreintational freedom. This way dissolving more solute can incur a net decrease in entropy. Increase in T makes this cost more significant.

Aggregation Change in p. H from 6. 5 to 5. 3 is enough to change charge of milk(casein) micelles and cause them to aggregate. © 2003, Miloslav Kalab; used by permission.

Examples of protein aggregation (often by exposing hydrophobic patch). 1. Substitution of glutamic acid for valine in sickle-cell hemoglobin 2. Fibrinogen to fibrin conversion (specific enzyme cuts part of fibrinogen) upon injury to blood vessel.
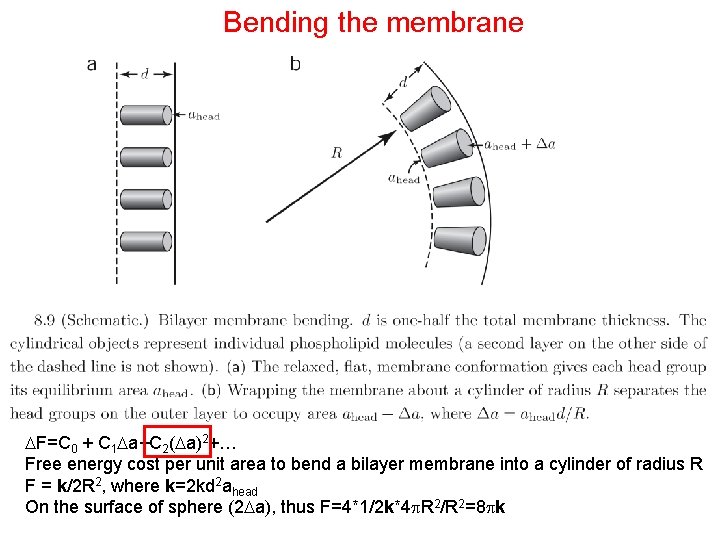
Bending the membrane F=C 0 + C 1 a+C 2( a)2+… Free energy cost per unit area to bend a bilayer membrane into a cylinder of radius R F = k/2 R 2, where k=2 kd 2 ahead On the surface of sphere (2 a), thus F=4*1/2 k*4 R 2/R 2=8 k

How to estimate k? Consider single layer at an oil-water interface. Bend patch of such layer into a spherical bulge with R ltail. Estimate energy cost at . Assume energy cost will be twice larger for bi-layer 2* = 2 k/(ltail)2 0. 05 J/m 2 ltail 1. 3 nm k 0. 8*10 -19 J

Reading: Finish reading Chapter 8. Problems: 8. 3 -8. 6
- Slides: 7