Introduction to Biophysics Lecture 1 Careers in Biophysics

Introduction to Biophysics Lecture 1 Careers in Biophysics History of Biophysics

What is Biophysics? It is neither “physics for biologists”, nor “physical methods applied to biology” It is a modern, interdisciplinary field of science leading to new approaches for our understanding of biological functions. Mathematics +Physics +Biology + Chemistry Paradigm: “Biological system is not simply the sum of its molecular components but is rather their functional integration” –example biological membrane. Scale from organism to single molecule Time scales from years to femtoseconds (10 -15), 1/1000 ps
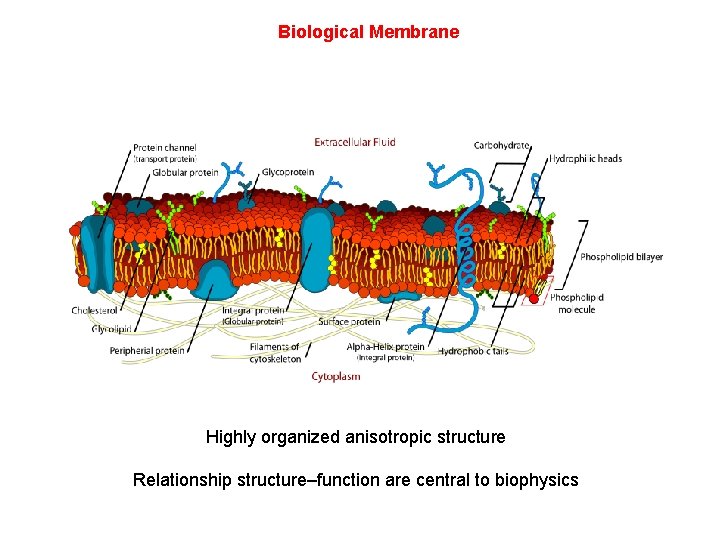
Biological Membrane Highly organized anisotropic structure Relationship structure–function are central to biophysics

Examples of problems to solve: How brain processes and stores information? How the heart pumps blood? How muscles contract? How plants use light for grow in photosynthesis? How genes are switched on and off?

Biophysics discovers how atoms are arranged to work in DNA and proteins. Protein molecules perform the body’s chemical reactions. Proteins make the parts of your eyes, ears, nose, and skin that sense your environment. They turn food into energy and light into vision. They provide immunity to illness. Proteins repair what is broken inside of cells, and regulate growth. They fire the electrical signals in your brain. They read the DNA blueprints in your body and copy the DNA for future generations.

Question biophysicists ask: How do protein Fold? How three-dimensional structure determines function? Why do molecules and parts of molecules assume the shapes they do? How do they fold into these shapes, and how do they change their structure under changing conditions? The shapes molecules take depend on the physical and chemical forces acting upon them and within them. X-ray crystallography, nuclear magnetic resonance spectroscopy and scanning probe microscopy, recombinant DNA, computation.

Job Market: Divisions of Biophysics: Universities Molecular biophysics Industry Biomechanics Medical Centers Membrane Biophysics Research Institutes Bio-electrochemistry Government Environmental Biophysics Impact on biotechnology and medicine Theoretical Biophysics

First Biophysicists Heraclitus 5 th century B. C. – earliest mechanistic theories of life processes, insight into dynamic. “Change is central to Universe”. “Logos is the fundamental order of all “on Nature” changes of objects with the flow of time” “You can not step twice into the same river”

First Biophysicists Epicurus 3 rd century B. C. – atom. Living organisms follow the same laws as non-living objects. Galen 2 th century AD – physician, most accomplished medical researcher of the Roman period. His theories dominated Western medicine for over millennium. Better anatomy only by Vesalius in 1543 Better understanding of blood and heart in 1628 Leonardo da Vinci 16 th century – mechanical principles of bird flight (to use for engineering design) - bionics

Borelli farther of biomechanics Giovanni Alfonso Borelli 17 th century- related animals to machines and utilized mathematics to prove his theories. De Motu Animalium – comprehensive biomechanical description of limb’s mobility, bird’s flight, swimming movement, heart function.

Luigi Galvani / Alessandro Volta Bio-electrochemistry 18 th (1771) Galvani touched frog nerve with charged scalpel. Signal transduction in neurons and communication between neurons and muscle has electrical nature.
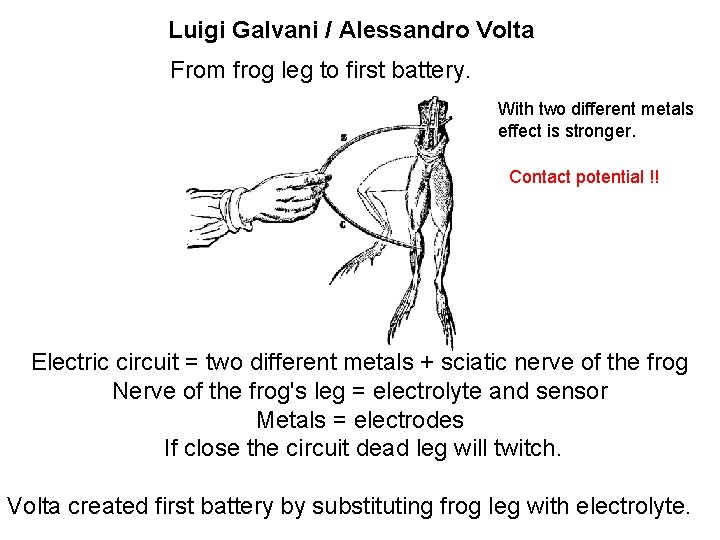
Luigi Galvani / Alessandro Volta From frog leg to first battery. With two different metals effect is stronger. Contact potential !! Electric circuit = two different metals + sciatic nerve of the frog Nerve of the frog's leg = electrolyte and sensor Metals = electrodes If close the circuit dead leg will twitch. Volta created first battery by substituting frog leg with electrolyte.
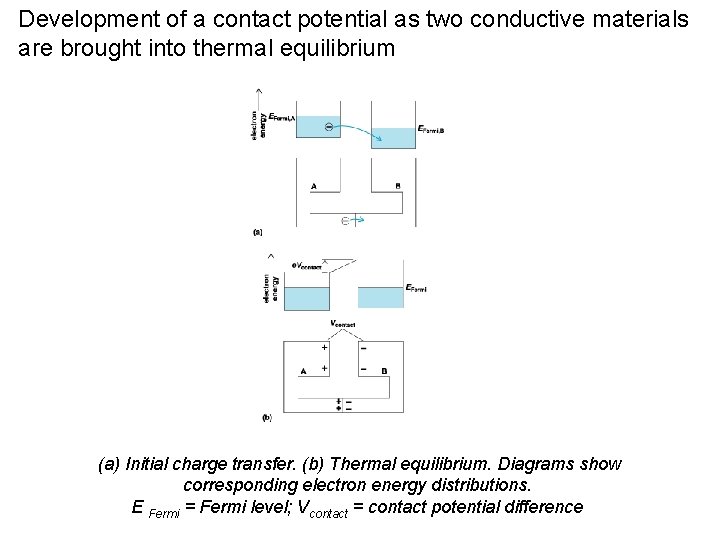
Development of a contact potential as two conductive materials are brought into thermal equilibrium (a) Initial charge transfer. (b) Thermal equilibrium. Diagrams show corresponding electron energy distributions. E Fermi = Fermi level; Vcontact = contact potential difference

History of discoveries in Biophysics: First law of thermodynamic Optical aspects of the human eye Theory of hearing Brown’s motion Osmotic process Nonequilibrium thermodynamics Discovery of X-rays – emergence of radiation biophysics

Transmission of order from one organism to it’s descendants The major advance in understanding the nature of gene mutation and gene structure. The work was a keystone in the formation of molecular genetics.

History of discoveries in Biophysics: Discovery of DNA structure Information theory Statistical physics of biopolymers

Grading: Midterm (take-home problem-solving exam) 20% Oral presentation of paper from research journal. 20% (Selection of papers on modern aspects of Biophysics will be provided by instructor. ) In class short quizzes 20% Final exam (problemsolving) 40%

PHYS 570 A (PHYS 57000) Computational (Bio)molecular Physics Sem. 1, 2, 3. Class 3, Cr. 3 Current Session: Not in session Prereq: Recommended background is Quantum Mechanics as taught in graduate (PHYS 660 or equivalent) or senior undergraduate (PHYS 460 & 461 or equivalent) courses and some familiarity with computer programming. The course is appropriate for students from physics, chemistry, biology, materials/computer science and related disciplines. Description: First principle (ab initio) methods provide significant insight about the electronic structure (ES) and physical properties of structures of interest in physics, chemistry, biology and materials science. These methods are particularly useful for understanding the electronic structure of molecular systems, such as active sites in metalloproteins or molecular nanostructures, and for interpreting experiments that probe their ground or excited states. This course will offer an introduction to the underlying theory and practical applications of some computational methods of electronic structure: * Hartree-Fock Theory * Kohn-Sham Density Functional Theory * Car-Parinello Molecular Dynamics For more information, please contact Prof. Jorge H. Rodriguez (270 Physics). PHYS 570 J (PHYS 57000) Biophysics II Summer, Fall, Spring Current Session: Not in session Prereq: NA Description: Biophysics is a dynamic and broad research area that applies physical principles and methods to studies of biologically interesting phenomenon. This course will introduce biophysics through developing an understanding of forces and energetics in biological materials. Topics covered will include: life in a low Reynolds number world (Brownian motion and diffusion), entropic forces, biochemical bonding, molecular motors (natural nanotechnology) and understanding nerve impulses (collective behavior). Biophysical methods such as atomic force microscopy, optical tweezers, single molecule microscopy and advanced fluorescence microscopy techniques (FRAP, FRET, FCS, two-photon, SHG, etc. ) will also be discussed. This course is designed for students at the senior undergraduate or entering graduate student level. The course will be self-contained (i. e. no prior biology courses are required)

BIOL 60000 Bioenergetics Offering: Fall, Credit 2. 0 Prerequisites: Pre-requisite: BCHM 56100 and 56200 and CHM 37300. Description: Energy transduction in biological membranes: physical chemical foundations; electron transfer, proton translocation; and active transport. Atomic structures of integral membrane protein complexes responsible for respiratory, photosynthetic generation of electrochemical potential; ATPase motor, and structure-based mechanisms. Offered in alternate years. BIOL 61100 Crystallography of Macromolecules Offering: Fall, Credit 3. 0 Prerequisites: Authorized equivalent courses or consent of instructor may be used in satisfying course pre- and co-requisites Description: The special techniques required in the structure determination of biological macromolecules. Symmetry of macromolecules. Data collection and processing. The isomorphous replacement technique. The molecular replacement technique. Use of anomalous dispersion. Restraint and constraint refinement. Computational techniques. Instructor approval is required

STAT 598 C. F 09 Statistical Methods For Bioinformatics and Computational Biology (Banner Course Number: 59800) Semester: Fall Prerequisites: At least one course from the list of STAT 514, STAT 524, and STAT 525. A prior experience with R and/or knowledge of basic biological concepts is desirable, but not required. Credits: 3 Primary Audience: The target audience are graduate students in statistics, as well as graduate students in life sciences who had previously taken a statistics class. Description: The course discusses statistical methods and algorithms for analysis of high-throughput experiments in molecular biology, using analysis of gene expression microarrays as a leading example. The objectives of the course are: Introduce relevant biological concepts, and describe the existing high-throughput technologies and biological questions that these technologies can help answer. Discuss statistical methods that have become standard when analyzing these data, as well as open research problems in this field. Discuss data structures and implementation of the methods in the R-based open source project Bioconductor. Although prior exposure to R is desirable, the course is self-contained. Life sciences students who have previous exposure to statistical methods but never used R will be able to learn the necessary concepts during the course. The course is project-driven and provides hands-on experience with data analysis, critical review of literature and communication of the results. At the end of the course the students will be able to perform independent analysis of biological data in an interdisciplinary environment such as a pharmaceutical company, or a computational biology research lab.


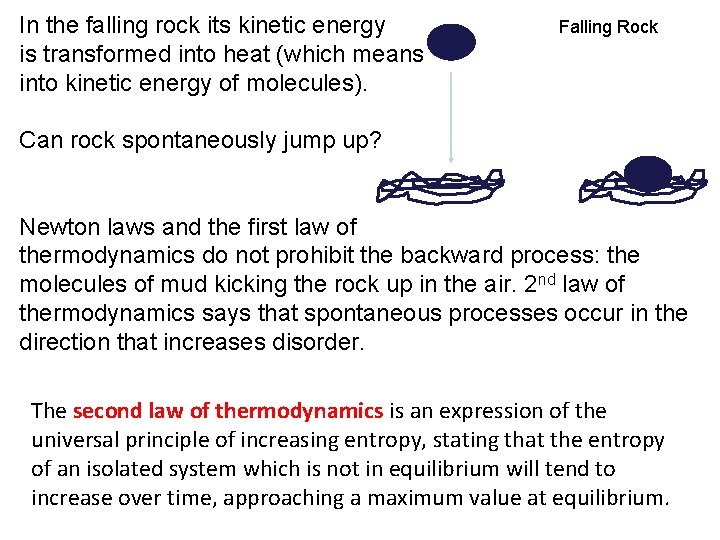
In the falling rock its kinetic energy is transformed into heat (which means into kinetic energy of molecules). Falling Rock Can rock spontaneously jump up? Newton laws and the first law of thermodynamics do not prohibit the backward process: the molecules of mud kicking the rock up in the air. 2 nd law of thermodynamics says that spontaneous processes occur in the direction that increases disorder. The second law of thermodynamics is an expression of the universal principle of increasing entropy, stating that the entropy of an isolated system which is not in equilibrium will tend to increase over time, approaching a maximum value at equilibrium.

1854 Rudolf Clausius – introduced entropy as a parameter of phenomenological thermodynamics

Question biophysicists ask: How life generates order? 2 nd law of thermodynamic - in isolated system molecular disorder never decreases spontaneously. Question: why Earth is full of life which is highly organized? Vitalism?

The Concept of Free Energy: -“useful” energy of a system - the part of total energy that can be harnessed to do “useful” work F=E-TS (total energy – randomness (or disorder)) If F<0 – process is spontaneous, T=constant F can decrease if E decreases (exmp. - heat loss) S increases (disorder tents to increase) Life doesn’t create order from nowhere. Life captures order, ultimately from the Sun. Prosesses of free energy transduction then transmit order through the biosphere.

Suggested Reading: P. Nelson Biological Physics Chapter 1 Schrodenger “What is life? The Physical Aspects of the living cell. ” Biophysics Society http: //www. biophysics. org/ – careers in Biophysics Careers in biophysics brochure was posted on course web site http: //www. physics. purdue. edu/academic_programs/courses/phys 570 G/

Home work: 1. Name people and dates for those discoveries: First law of thermodynamic Optical aspects of the human eye Theory of hearing Brown’s motion Osmotic process Nonequilibrium thermodynamics Discovery of X-rays Discovery of DNA structure Information theory 2. Nelson page 33 – problem 1. 7 Tour de France
- Slides: 29