Introduction to Biochemistry What is Biochemistry n n

Introduction to Biochemistry

What is Biochemistry? n n n Biochemistry is the chemistry of life. Basic principles of biochemistry are common to all living organism Biochemists use physical and chemical principles to explain biology at the molecular level.

Biochemistry Biochemical molecules Macromolecules contain carbon Valance of carbon = 4 Bonds with oxygen, hydrogen and nitrogen

How does biochemistry impact you? n n n Veterinary Medicine Agriculture Industrial applications Environmental applications

Principle Areas of Biochemistry • Structure and function of biological macromolecules • Metabolism – anabolic and catabolic reactions. • Molecular Genetics • Regulation of protein synthesis

Organization of Life n n n n Elements (Minerals) Simple organic compounds (monomers) Macromolecules (polymers) Supramolecular structure Organelles Cells Tissues Organisms
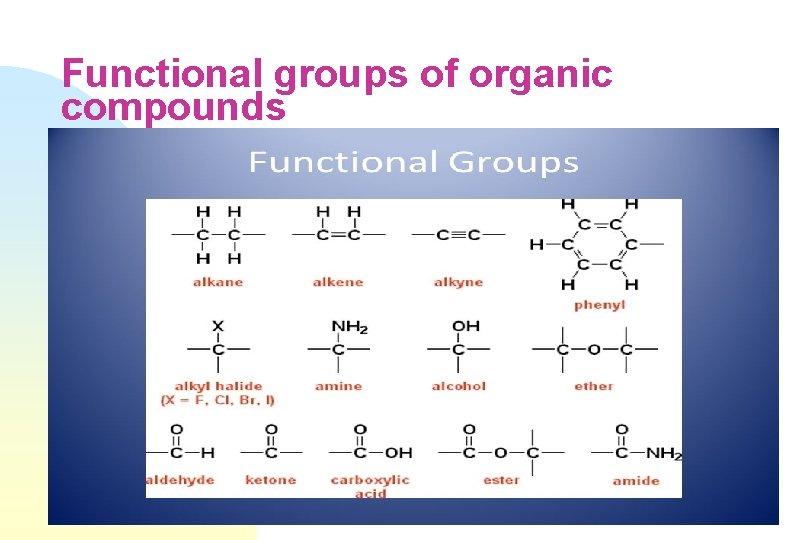
Functional groups of organic compounds
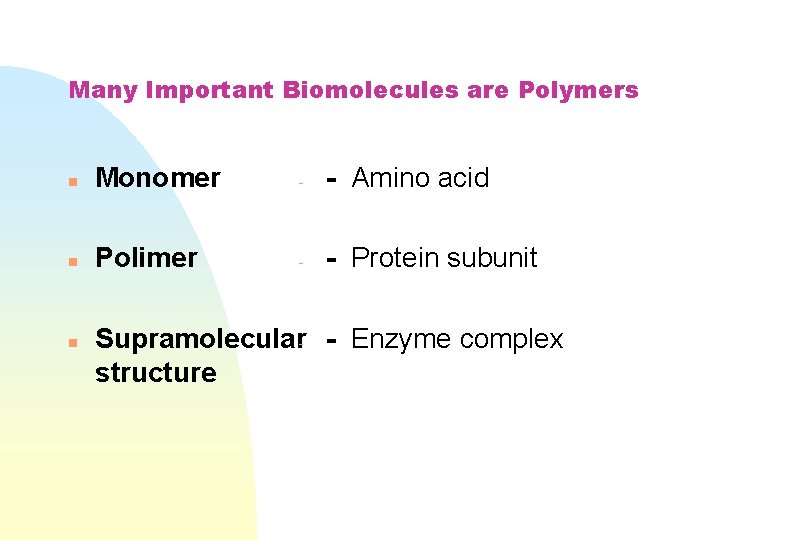
Many Important Biomolecules are Polymers n Monomer - - Amino acid n Polimer - - Protein subunit n Supramolecular - Enzyme complex structure

Cells Prokaryotes are organisms made up of cells that lack a cell nucleus or any membrane-encased organelles. Eukaryotes are organisms made up of cells that possess a membrane-bound nucleus (includes genetic material) as well as membrane-bound organelles.

Carbohydra tes • General characteristics • Contains carbon, hydrogen and oxygen • Soluble in water • Sizes – small single units to larger compounds • Function • Cell membrane – cell to cell recognition • Structure • Monomer – monosaccharide • Polymer – polysaccharide

Carbohydra tes • Monosaccharides (simple sugars) • C 6 H 12 O 6 Hexoses • 3 isomers (glucose, fructose and galactose) • Glucose, fructose and galactose are 6 -carbon sugars • Disaccharides • Formed by dehydration synthesis • Broken-up by hydrolysis Polysaccharides Glycogen, Starch, Cellulose

Polysaccharides n Glycogen – Stored in the liver and muscle. n Starches – plants n Cellulose - roughage

Lipids General Characteristics Contains carbon, hydrogen and oxygen Insoluble in water – soluble in other nonpolar substances (alcohol) Non-polar structure Function Cell membrane, steroids, fat-soluble vitamins Structure Most – fatty acid tail (H-C chain) that is non-polar

CHOLESTEROL IS CLASSIFIED AS A STEROL – LIPID. • MADE UP OF ‘FUSED’ CARBON RINGS AND SIDE CHAINS OF MOLECULES • A ‘‘PRECURSOR’’ TO OTHER STEROIDS – TESTOSTERONE, VITAMIN D • Side-chains made each steroid different
- Slides: 14