Introduction to Biochemistry What is Biochemistry Biochemistry chemistry

Introduction to Biochemistry

What is Biochemistry? • Biochemistry = chemistry of life. • Biochemists use physical and chemical principles to explain biology at the molecular level. • Basic principles of biochemistry are common to all living organism

How does biochemistry impact you? • Medicine • Agriculture • Industrial applications • Environmental applications
Principle Areas of Biochemistry • Structure and function of biological macromolecules • Metabolism – anabolic and catabolic processes. • Molecular Genetics – How life is replicated. Regulation of protein synthesis
Life Before Biochemistry

Once upon a time, a long time ago…. . Vitalism: idea that substances and processes associated with living organisms did not behave according to the known laws of physics and chemistry Evidence: 1) Only living things have a high degree of complexity 2) Only living things extract, transform and utilize energy from their environment 3) Only living things are capable of self assembly and self replication

Origins of Biochemistry: A challenge to “Vitalism. ” Famous Dead Biochemist!

Fallacy #1: Biochemicals can only be produced by living organisms • Dead Biochemist #1 • 1828 Friedrich Wohler

Fallacy #2: Complex bioconversion of chemical substances require living matter Dead Biochemists #2 • 1897 Eduard Buchner Glucose + Dead Yeast = Alcohol

Fallacy #2: Complex bioconversion of chemical substances require living matter Dead Biochemists #3 • Emil Fischer

Fallacy #2: Complex bioconversion of chemical substances require living matter Dead Biochemists #4 1926 J. B. Sumner

Findings of other famous dead biochemist • 1944 Avery, Mac. Leod and Mc. Carty identified DNA as information molecules • 1953 Watson (still alive) and Crick proposed the structure of DNA • 1958 Crick proposed the central dogma of biology

Organization of Life • • elements simple organic compounds (monomers) macromolecules (polymers) supramolecular structures organelles cells tissues organisms
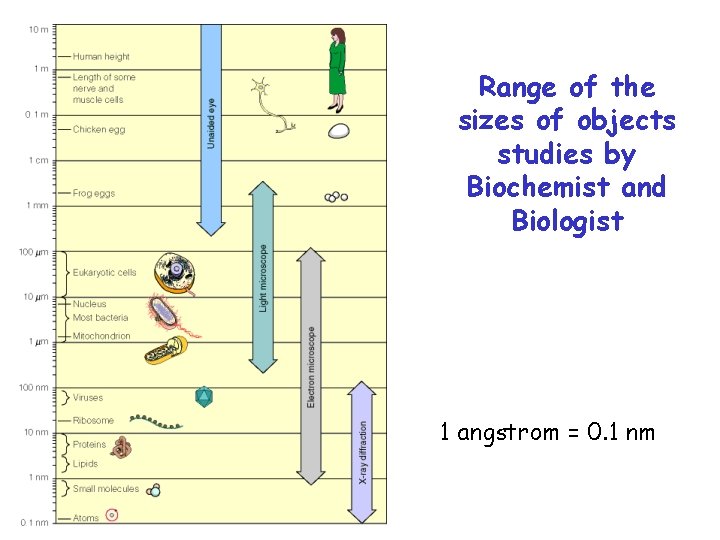
Range of the sizes of objects studies by Biochemist and Biologist 1 angstrom = 0. 1 nm
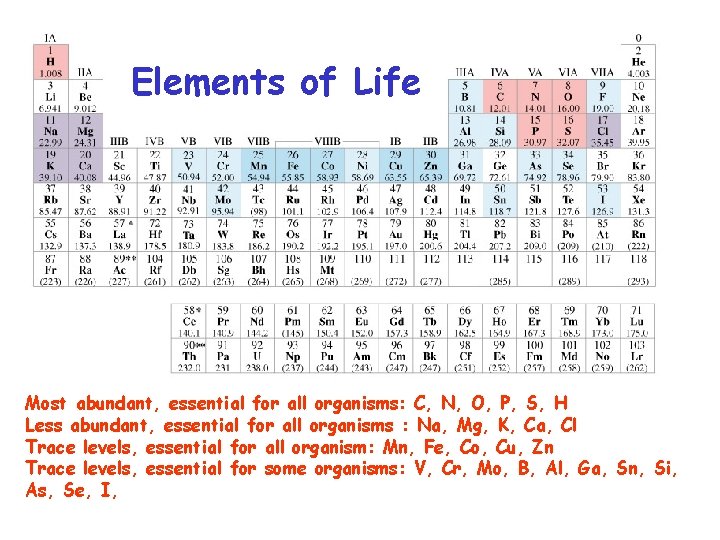
Elements of Life Most abundant, essential for all organisms: C, N, O, P, S, H Less abundant, essential for all organisms : Na, Mg, K, Ca, Cl Trace levels, essential for all organism: Mn, Fe, Co, Cu, Zn Trace levels, essential for some organisms: V, Cr, Mo, B, Al, Ga, Sn, Si, As, Se, I,
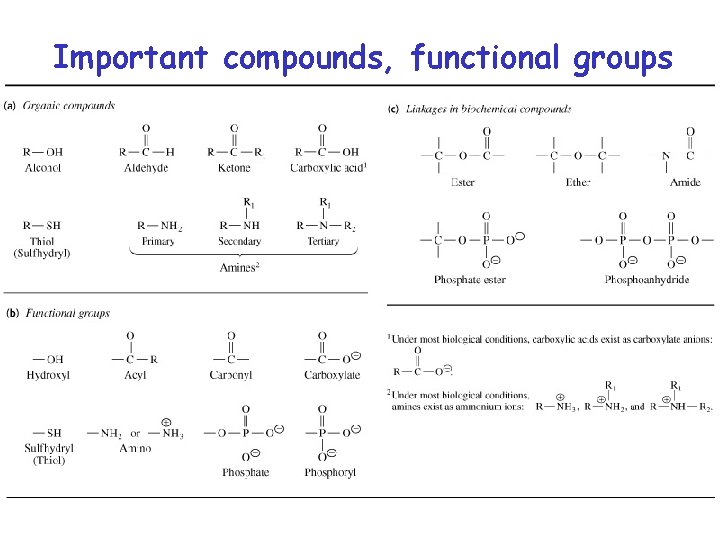
Important compounds, functional groups
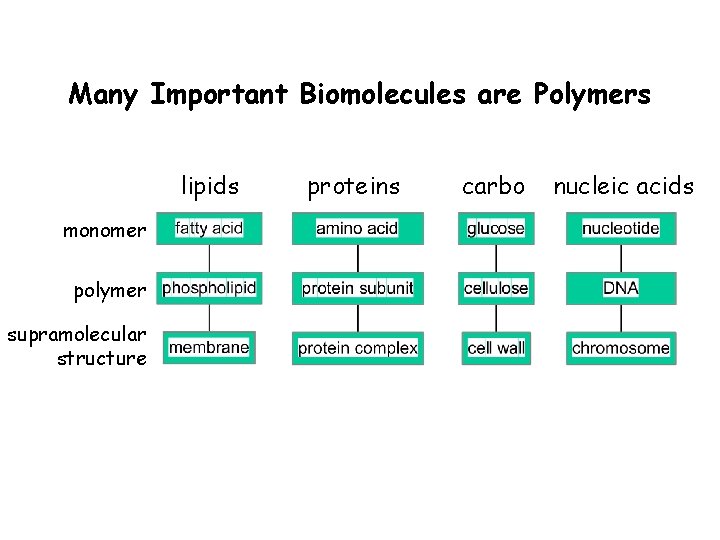
Many Important Biomolecules are Polymers lipids monomer polymer supramolecular structure proteins carbo nucleic acids
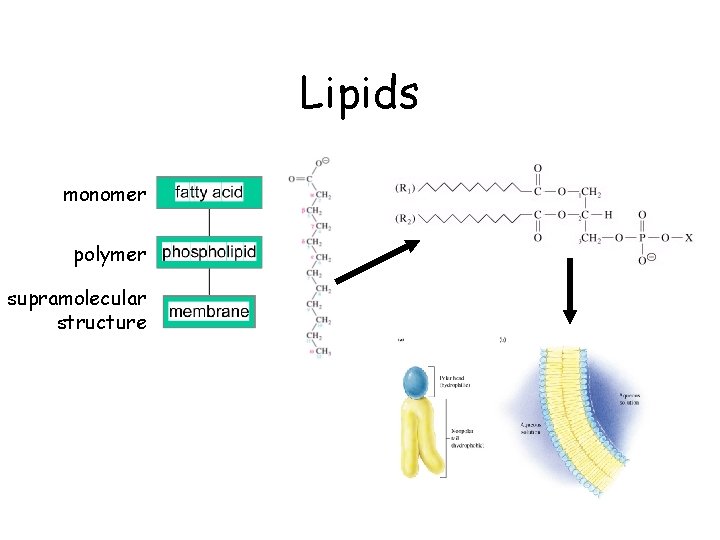
Lipids monomer polymer supramolecular structure
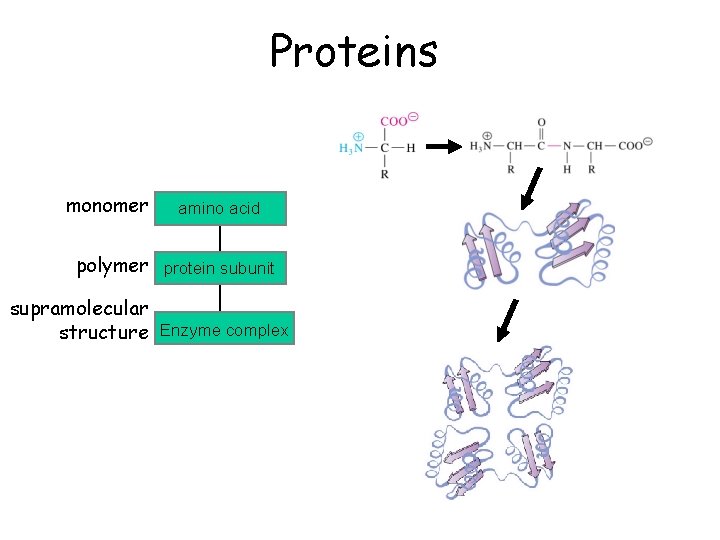
Proteins monomer amino acid polymer protein subunit supramolecular structure Enzyme complex
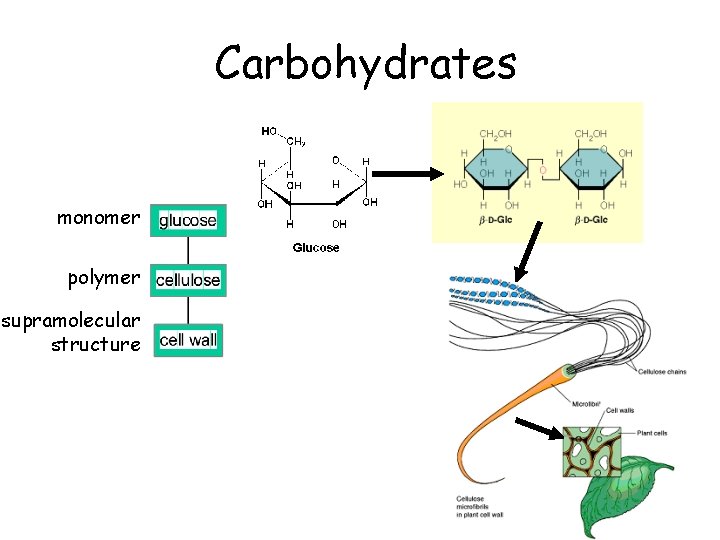
Carbohydrates monomer polymer supramolecular structure
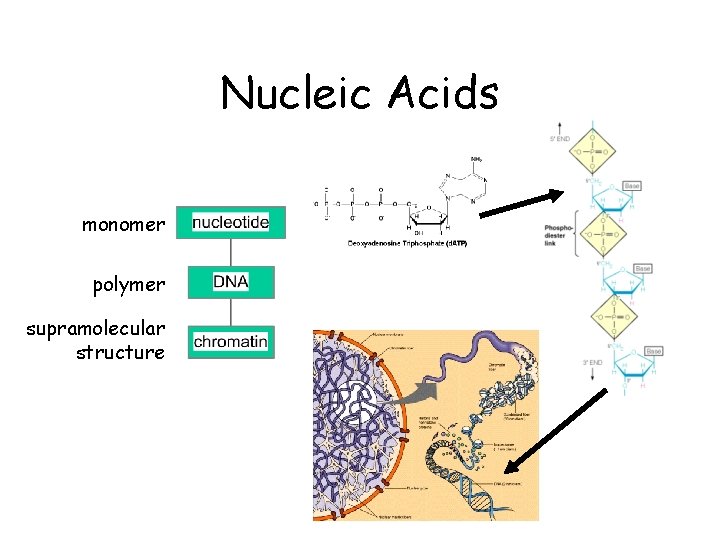
Nucleic Acids monomer polymer supramolecular structure
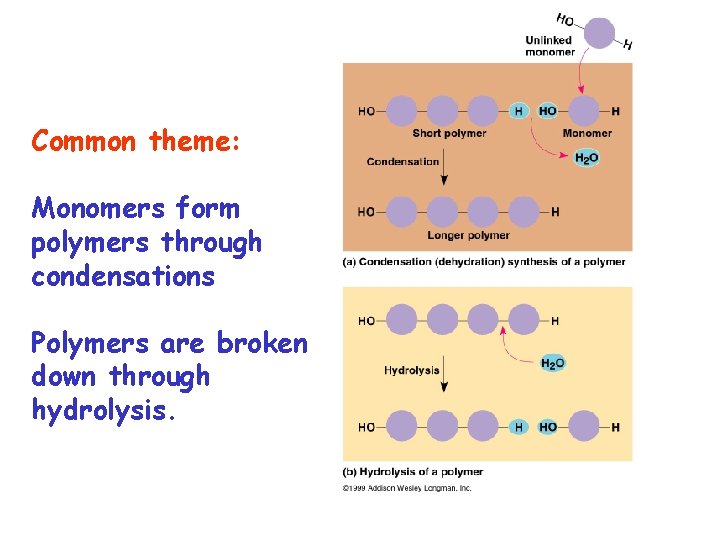
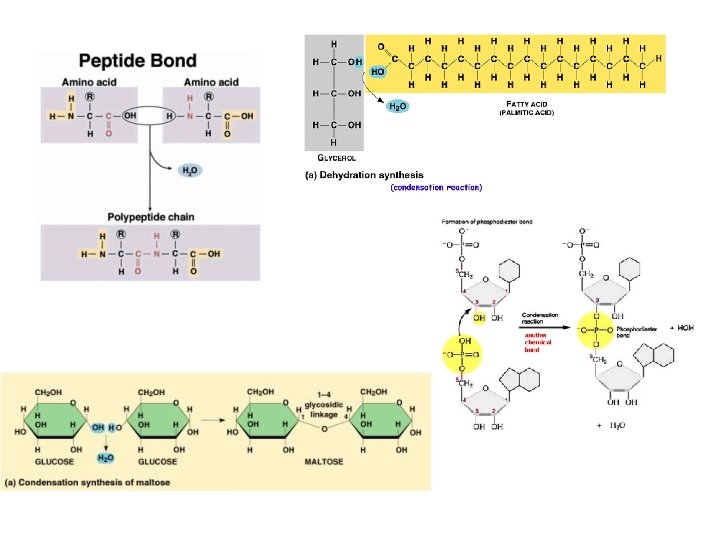
Common theme: Monomers form polymers through condensations Polymers are broken down through hydrolysis.
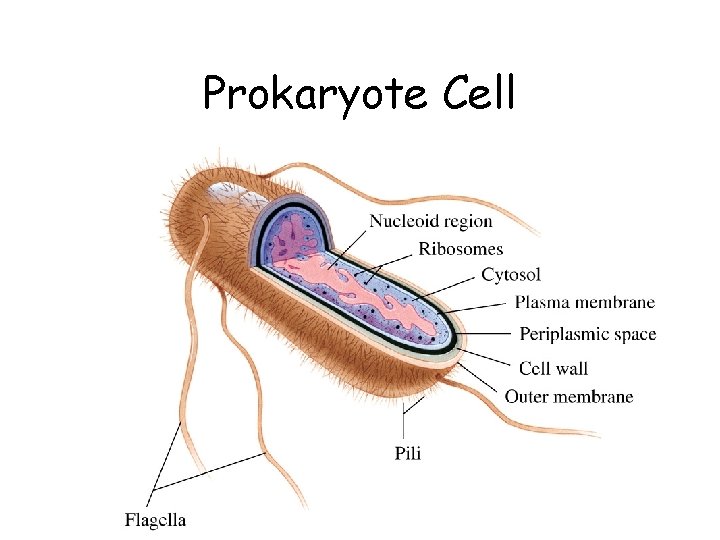
Prokaryote Cell
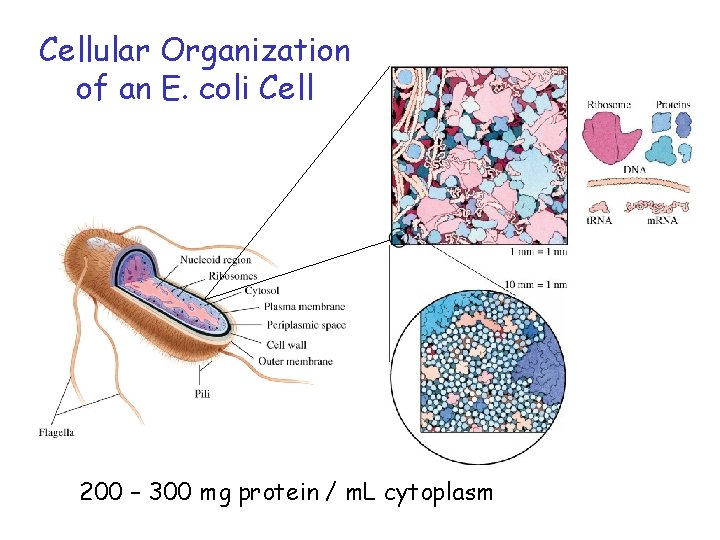
Cellular Organization of an E. coli Cell 200 – 300 mg protein / m. L cytoplasm
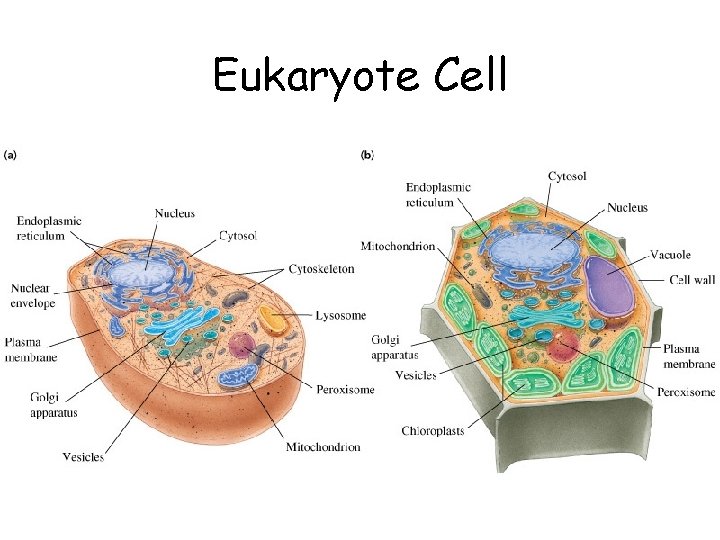
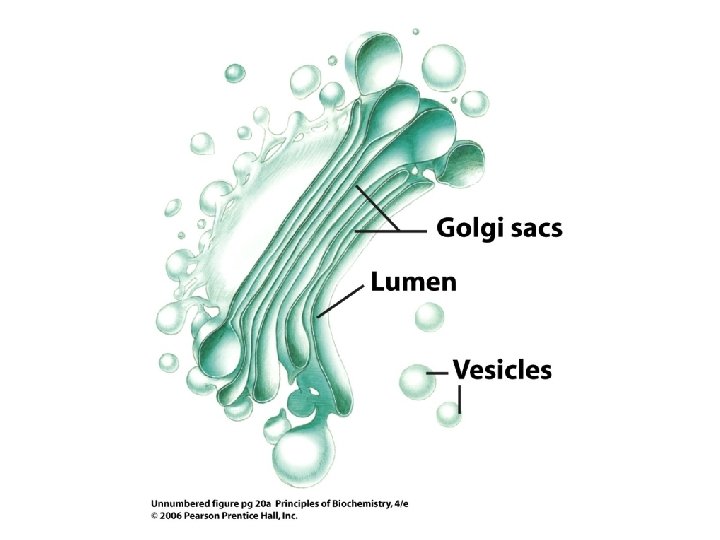
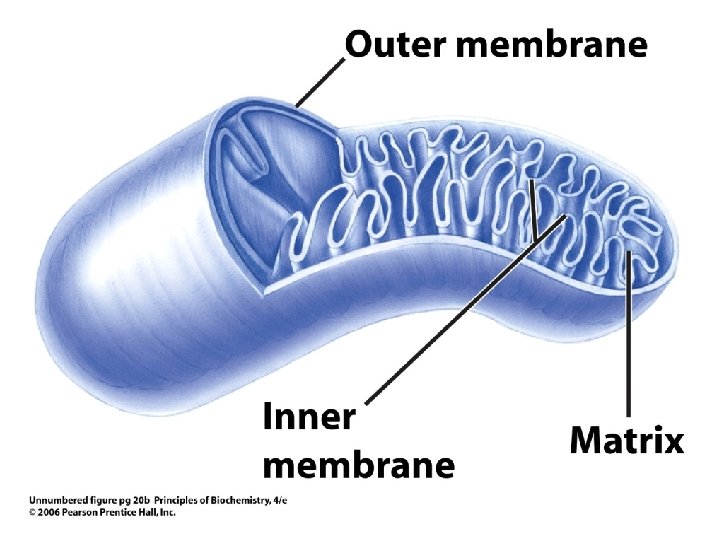
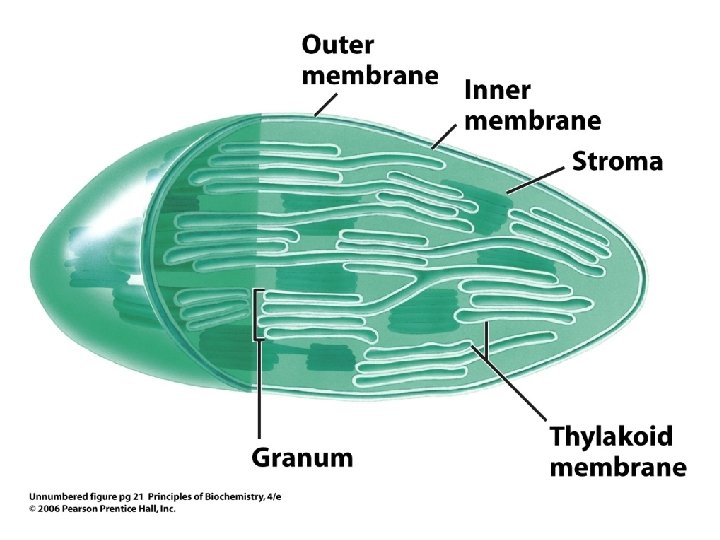
Eukaryote Cell
- Slides: 30