Introduction to Biochemistry Andy Howard Biochemistry Fall 2013

Introduction to Biochemistry Andy Howard Biochemistry, Fall 2013 IIT 08/20/2013 Biochemistry: Introduction 1

What is biochemistry? l By the end of this course you should be able to construct your own definition; but for now: l Biochemistry is the study of chemical reactions in living tissue. 08/20/2013 Biochemistry: Introduction p. 2 of 48

Plans l l l What is biochemistry? Cells Cell components Organic and biochemistry Concepts from organic chemistry to remember Small molecules and macromolecules 08/20/2013 Classes of small molecules l Classes of macromolecules l Course logistics l Biochemistry: Introduction – Homework – Exams – i. Clickers p. 3 of 48

What will we study? Biochemistry is the study of chemical reactions in living tissue, both within cells and in intercellular media. l As such, it concerns itself with a variety of specific topics: l 08/20/2013 Biochemistry: Introduction p. 4 of 48

Topics in biochemistry What reactions occur; l The equilibrium energetics and kinetics of those reactions; l How the reactions are controlled, at the chemical and cellular or organellar levels; l How the reactions are organized to enable biological function within the cell and in tissues and organisms. l 08/20/2013 Biochemistry: Introduction p. 5 of 48

Organic and biological chemistry Most molecules in living things (other than H 2 O, O 2, N 2, and CO 2) contain C-C or C-H bonds, so biochemistry depends heavily on organic chemistry l But the range of organic reactions that occur in biological systems is fairly limited compared to the full range of organic reactions: l 08/20/2013 Biochemistry: Introduction p. 6 of 48

Why we use only a subset of organic chemistry in biochemistry Biochemical reactions are almost always aqueous. Frederich l They occur within a narrow temperature and Wöhler pressure range. l They occur within narrowly buffered p. H ranges. l Many of the complex reaction mechanisms discovered and exploited by organic chemists since the 1860's have no counterparts in the biochemical universe. l 08/20/2013 Biochemistry: Introduction p. 7 of 48

Cells l Most biochemical reactions (but not all!) take place within semi-independent biological entities known as cells l Cells in general contain replicative and protein-synthetic machinery in order to reproduce and survive l They often exchange nutrients and information with other cells 08/20/2013 Biochemistry: Introduction p. 8 of 48
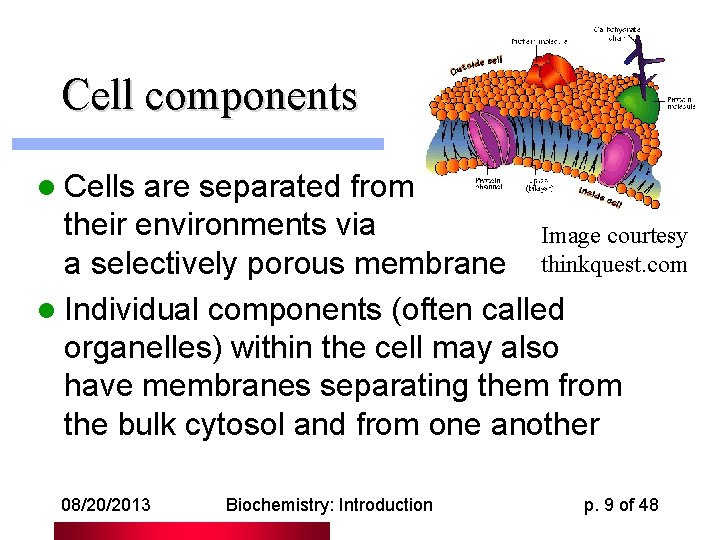
Cell components l Cells are separated from their environments via Image courtesy a selectively porous membrane thinkquest. com l Individual components (often called organelles) within the cell may also have membranes separating them from the bulk cytosol and from one another 08/20/2013 Biochemistry: Introduction p. 9 of 48
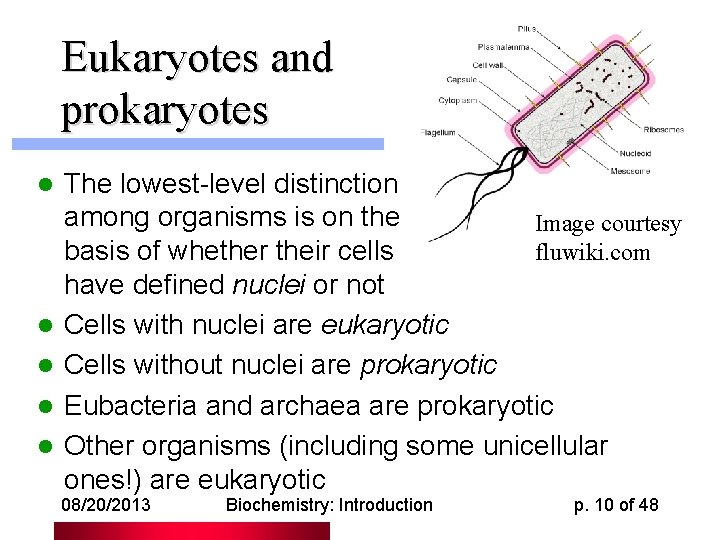
Eukaryotes and prokaryotes l l l The lowest-level distinction among organisms is on the Image courtesy fluwiki. com basis of whether their cells have defined nuclei or not Cells with nuclei are eukaryotic Cells without nuclei are prokaryotic Eubacteria and archaea are prokaryotic Other organisms (including some unicellular ones!) are eukaryotic 08/20/2013 Biochemistry: Introduction p. 10 of 48

Eukaryotic organelles I Nucleus: contains Image courtesy genetic information; Williamsclass. com site for replication and transcription l Endoplasmic reticulum: site for protein synthesis and protein processing l Ribosome: protein-synthetic machine l Golgi apparatus: site for packaging proteins for secretion and delivery l 08/20/2013 Biochemistry: Introduction p. 11 of 48

Eukaryotic organelles II Mitochondrion: site for most energyproducing reactions l Lysosome: digests materials during endocytosis and cellular degradation l Peroxisome: site for oxidation of some nutrients and detoxification of the H 2 O 2 created thereby l Cytoskeleton: network of filaments that define the shape and mobility of a cell l 08/20/2013 Biochemistry: Introduction p. 12 of 48

Eukaryotic organelles III Chloroplast: site for most photosynthetic reactions l Vacuoles: sacs for water or other nutrients l Cell wall: bacterial or plant component outside cell membrane that provides rigidity and protection against osmotic shock l 08/20/2013 Biochemistry: Introduction p. 13 of 48
Molecular machines Cells contain components featuring enzyme-driven molecular machines that accomplish specific tasks l Too small and too internalized within cells to be considered as organelles themselves, but still important l Examples: proteasome, spliceosomes, fatty acid synthases l Ribosomes are borderline between organelles and molecular machines l 08/20/2013 Biochemistry: Introduction p. 14 of 48

Concepts from organic chemistry There are some elements of organic chemistry that you should have clear in your minds. l All of these are concepts with significance outside of biochemistry, but they do play important roles in biochemistry. l If any of these concepts is less than thoroughly familiar, please review it: l 08/20/2013 Biochemistry: Introduction p. 15 of 48

Organic concepts I Image courtesy Michigan State U. l Covalent bond: A strong attractive interaction between neighboring atoms in which a pair of electrons is roughly equally shared between the two atoms. – Covalent bonds may be single bonds, in which one pair of electrons is shared; double bonds, which involve two pairs of electrons; or triple bonds, which involve three pairs (see above). – Single bonds do not restrict the rotation of other substituents around the bond; double and triple bonds do. 08/20/2013 Biochemistry: Introduction p. 16 of 48
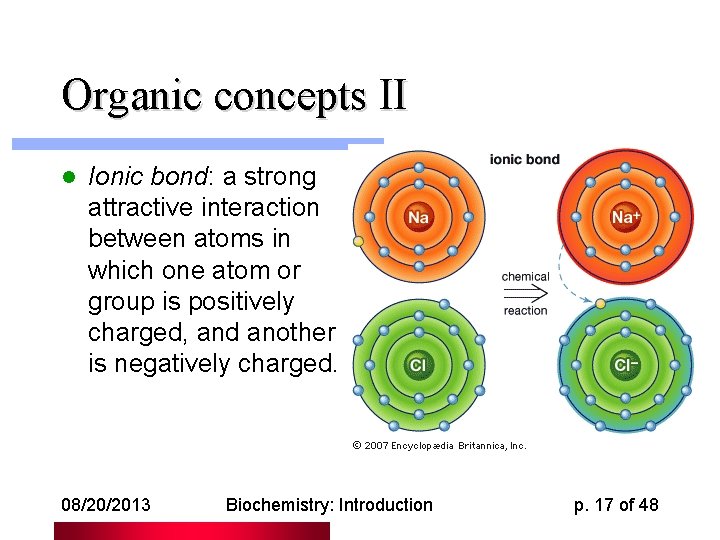
Organic concepts II l Ionic bond: a strong attractive interaction between atoms in which one atom or group is positively charged, and another is negatively charged. 08/20/2013 Biochemistry: Introduction p. 17 of 48

Organic concepts III l Hydrogen bond: A weak attractive interaction between neighboring atoms in which a hydrogen atom carrying a slight, partial positive charge shares that positive charge with a neighboring electronegative atom. Cartoon courtesy CUNY Brooklyn – The non-hydrogen atom to which the hydrogen is covalently bonded is called the hydrogenbond donor; – the neighboring atom that takes on a bit of the charge is called the hydrogen-bond acceptor 08/20/2013 Biochemistry: Introduction p. 18 of 48
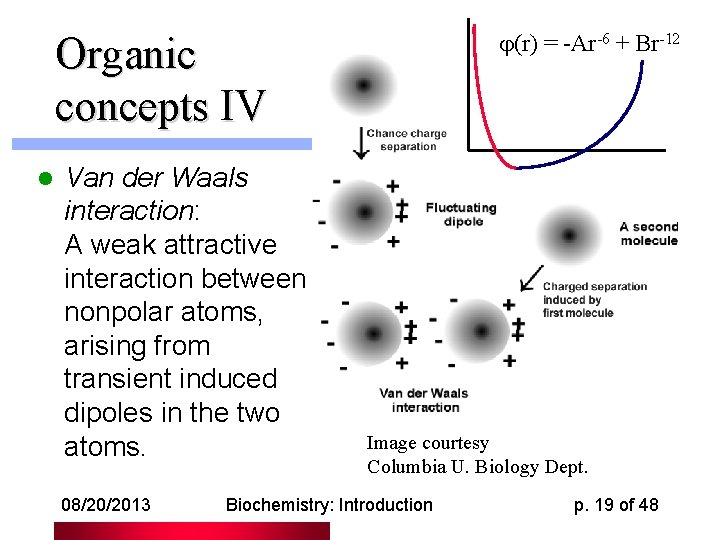
(r) = -Ar-6 + Br-12 Organic concepts IV l Van der Waals interaction: A weak attractive interaction between nonpolar atoms, arising from transient induced dipoles in the two atoms. 08/20/2013 Image courtesy Columbia U. Biology Dept. Biochemistry: Introduction p. 19 of 48

Organic Concepts V l Chirality: The property of a molecule under which it cannot be superimposed upon its mirror image. 08/20/2013 Image courtesy DRECAM, France Biochemistry: Introduction p. 20 of 48

Chirality, continued Chirality manifests itself at the locations of specific atoms l In principle many different atoms can be chiral; but l In biomolecules, chirality almost always appears at carbon atoms (rarely at phosphorus or nitrogen) l In general, a carbon will be chiral if and only if it has four non-identical substituents l 08/20/2013 Biochemistry: Introduction p. 21 of 48
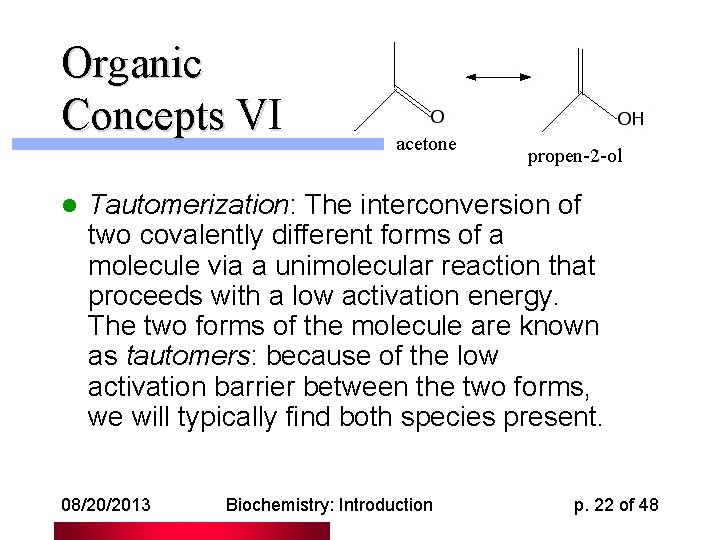
Organic Concepts VI l acetone propen-2 -ol Tautomerization: The interconversion of two covalently different forms of a molecule via a unimolecular reaction that proceeds with a low activation energy. The two forms of the molecule are known as tautomers: because of the low activation barrier between the two forms, we will typically find both species present. 08/20/2013 Biochemistry: Introduction p. 22 of 48

Organic Concepts VII l Nucleophilic substitution: a reaction in which an electron-rich (nucleophilic) molecule attacks an electron-poor (electrophilic) molecule and replaces group or atom within the attacked species. – The displaced group is known as a leaving group. – This is one of several types of substitution reactions, and it occurs constantly in biological systems. 08/20/2013 Biochemistry: Introduction p. 23 of 48

Organic Concepts VIII l Polymerization: creation of large molecules by sequential addition of simple building blocks – often by dehydration, i. e. , the elimination of water from two species to form a larger one: R 1 -O-H + HO-R 2 -X-H R 1 -X-R 2 -OH + H 2 O – The product here can then react with HO-R 3 -X-H to form R 1 -X-R 2 -X-R 3 -OH with elimination of another water molecule, and so on. 08/20/2013 Biochemistry: Introduction p. 24 of 48

Organic Concepts IX Equilibrium: in the context of a chemical reaction, the state in which the concentrations of reactants and products are no longer changing with time because the rate of reaction in one direction is equal to the rate in the opposite direction. l Kinetics: the study of the rates at which reactions proceed. Conventionally, we use the term thermodynamics to describe our understanding of the energetics of equilibrium systems l 08/20/2013 Biochemistry: Introduction p. 25 of 48

Organic Concepts X l Catalysis: the lowering of the energetic barrier between substrates and products in a reaction by the participation of a substance that ultimately is unchanged by the reaction – It is crucial to recognize that catalysts (chemical agents that perform catalysis) do not change the equilibrium position of the reactions in which they participate: – they only change the rates (the kinetics) of the reactions they catalyze. l Zwitterion: a compound containing both a positive and a negative charge 08/20/2013 Biochemistry: Introduction p. 26 of 48
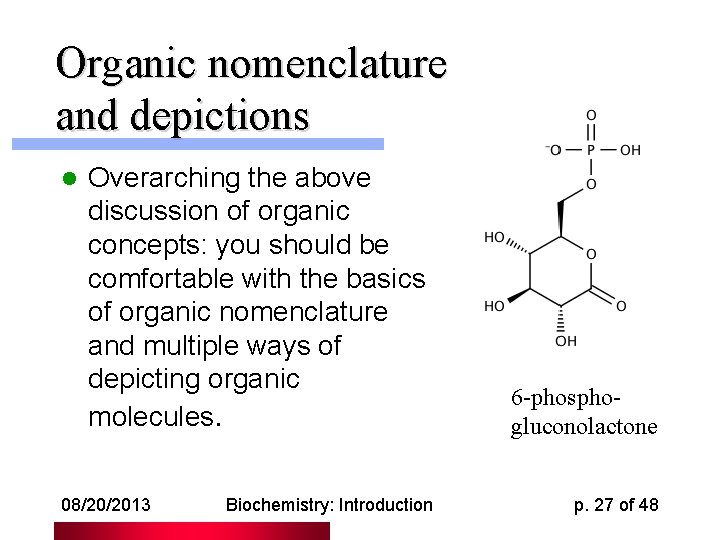
Organic nomenclature and depictions l Overarching the above discussion of organic concepts: you should be comfortable with the basics of organic nomenclature and multiple ways of depicting organic molecules. 08/20/2013 Biochemistry: Introduction 6 -phosphogluconolactone p. 27 of 48
Classes of small molecules l Small molecules other than water make up a small percentage of a cell's mass, but small molecules have significant roles in the cell, both on their own and as building blocks of macromolecules. The classes of small molecules that play significant roles in biology are listed below. In this list, "soluble" means "water-soluble". 08/20/2013 Biochemistry: Introduction p. 28 of 48

Biological small molecules I Water: Hydrogen hydroxide. In liquid form in biological systems. See below. l Lipids: Hydrophobic molecules, containing either alkyl chains or fused-ring structures. A biological lipid usually contains at least one highly hydrophilic moeity. l 08/20/2013 Biochemistry: Introduction p. 29 of 48

Biological small molecules II l Carbohydrates: Polyhydroxylated compounds for which the building blocks are highly soluble. – The typical molecular formula for the monomeric forms of these compounds is (CH 2 O)n, where 3 < n < 9, – but usually n = 3, 5 or 6. – In addition to the -OH groups, there’s a carbonyl at either position 1 or position 2 08/20/2013 Biochemistry: Introduction p. 30 of 48

Biological small molecules III Amino acids: Compounds containing an amine (NH 3+) group and a carboxyl (COO-) group. l The most important biological amino acids are a-amino acids, in which the amine group and the carboxyl group are separated by one carbon, and that intervening carbon has a hydrogen attached to it. Thus the general formula for an a-amino acid is l H 3 N+ - CHR - COOl 08/20/2013 Biochemistry: Introduction p. 31 of 48

Biological small molecules IV l Nucleic acid bases: Soluble compounds that include an aromatic nitrogen-containing ring system. – The ring systems are derived either from purine or pyrimidine. – The most important biological nucleic acids are those in which the ring system is covalently attached to a five-carbon sugar, ribose, usually with a phosphate group attached to the same ribose ring. 08/20/2013 Biochemistry: Introduction p. 32 of 48

Small molecules V l Inorganic ions: Ionic species containing no carbon but containing one or more atoms and at least one net charge. – Ions of biological significance include Cl-, Na+, K+, Mg+2, Mn+2, I-, Ca+2, PO 4 -3, SO 4 -2, NO 3 -, NO -, and NH +. 2 4 – Phosphate (PO 4 -3) is often found in partially protonated forms HPO 4 -2 and H 2 PO 4– Ammonium ions occasionally appear as neutral ammonia (NH 3), particularly at higher p. H values 08/20/2013 Biochemistry: Introduction p. 33 of 48

Biological Small Molecules VI Cofactors: This is a catchall category for small molecules that serve in some functional role in biological organisms. Many are vitamins or are derived from vitamins; a vitamin is defined as an organic molecule that is necessary in small quantities for metabolism but cannot be synthesized by the organism. Thus the same compound may be a vitamin for one organism and not for another. l Ascorbate (vitamin C) is a vitamin for humans and guinea pigs but not for most other mammals. l Cofactors often end up as prosthetic groups, covalently or noncovalently attached to proteins and involved in those proteins' functions. l 08/20/2013 Biochemistry: Introduction p. 34 of 48
Biological macromolecules l Most big biological molecules are polymers, i. e. molecules made up of large numbers of relatively simple building blocks. l Cobalamin is the biggest nonpolymeric biomolecule I can think of (MW 1356 Da) 08/20/2013 Biochemistry: Introduction Structure courtesy Wikimedia p. 35 of 48

Categories of biological polymers Biopolymers are assembled by adding one monomer at a time to the growing chain by dehydration l Proteins l Nucleic acids l Polysaccharides. . Only the polysaccharides are typically branched; the others are linear 08/20/2013 Biochemistry: Introduction p. 36 of 48

Polymers and oligomers l These are distinguished only by the number of building-blocks contained within the multimer l Oligomers: typically < 50 building blocks l Polymers 50 building blocks. 08/20/2013 Biochemistry: Introduction p. 37 of 48
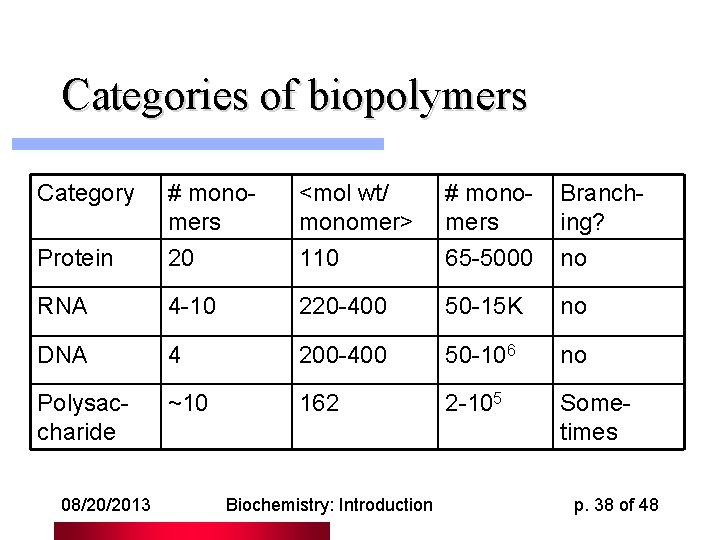
Categories of biopolymers Category # monomers <mol wt/ monomer> # monomers Branching? Protein 20 110 65 -5000 no RNA 4 -10 220 -400 50 -15 K no DNA 4 200 -400 50 -106 no Polysaccharide ~10 162 2 -105 Sometimes 08/20/2013 Biochemistry: Introduction p. 38 of 48

Course structure I teach biology 401 all semester; Prof. Nicholas Menhart teaches the followup course, biology 402, which focuses on specific metabolic systems l We will introduce general concepts of metabolism this semester without going into specific systems l We’ll spend a fair amount of time discussing techniques and analytical approaches, which will be instantiated in 402 l 08/20/2013 Biochemistry: Introduction p. 39 of 48

Examination plans l Two midterms (9/19, 10/24) plus a final on Thursday 5 December from 2 to 4 pm – My exams will be closed-book, closed-notes exams. Calculators are not allowed. – You’ll have a help-sheet for each of my exams – Gauge your memorization with the help-sheet before you: what’s on the help sheet doesn’t need to be memorized – My exams tend to be long but easy: budget your time carefully! 08/20/2013 Biochemistry: Introduction p. 40 of 48

Grading I’m a moderately tough grader, but I do curve this course l Curving is relative to students over several years of performance, not just this year l The cutoff for an A is likely to be around an 82, but it’s uncertain l Homework, literature assignments, i. Clicker quizzes, and discussion-board participation count; see Blackboard site for details l 08/20/2013 Biochemistry: Introduction p. 41 of 48

Textbook and Lecture Notes Required textbook: Garrett & Grisham, Biochemistry, either 4 th edition or 5 th edition. It’s a detail-rich text and is clearly written. l Be prepared for the lecture notes themselves to evolve during the course; they’re all posted, but I will generally revise them the day that I deliver the lecture. l 08/20/2013 Biochemistry: Introduction p. 42 of 48

Office Hours Life Sciences Room 174 l I should be available 3: 30 pm-6 pm Tuesdays and Thursdays and often from 11 am to 1 pm as well l If that doesn’t work, make an appointment: howard@iit. edu office 312 -567 -5881, cell 773 -368 -5067 l The discussion board is another good way to reach me and the rest of the class as well! l 08/20/2013 Biochemistry: Introduction p. 43 of 48

Assignments l Regular homeworks will be due weekly, generally on Fridays l But no assignment due this week l Literature assignments are due weekly on Tuesdays (except this one) l Specific readings already posted will be augmented but not deleted 08/20/2013 Biochemistry: Introduction p. 44 of 48

Lateness? l Regular homework: – No penalty if turned in by 24 h of deadline – Modest penalties if 1 -7 days late – After 7 days the answer key will be posted, so no credit given after that l Literature assignments: – No penalty if turned in by 24 h of deadline – Half credit if turned in 1 -7 days late – Limited credit later than that 08/20/2013 Biochemistry: Introduction p. 45 of 48

Arrangements for exams l Live students should take the exams on the stated dates l Internet & TV students should begin the midterms between 9 am on the statutory date and 5 pm on the day after l If you’re a non-local Internet student, you need to find a proctor well before 19 September! 08/20/2013 Biochemistry: Introduction p. 46 of 48

How exams work Combination of multiple-choice, short answer, paragraph-answer, and computational problems l No electronic devices of any kind allowed (you may need to review long division!) l Multiple-page help sheet will be available for each exam; in fact, the current drafts of the help-sheets are already on Blackboard l 08/20/2013 Biochemistry: Introduction p. 47 of 48

Other administrative stuff Feel free to watch the Internet lectures even if you’re in the live class l But if you’re in the live section, I expect you to attend class l If you miss a live class that includes an i. Clicker quiz, you’ll get a zero on that i. Clicker quiz unless you’ve made arrangements in advance to compensate l 08/20/2013 Biochemistry: Introduction p. 48 of 48
- Slides: 48