INTRODUCTION TO BIOCHEMISTRY 1 AISHA OMER ORGANIZATION OF

INTRODUCTION TO BIOCHEMISTRY 1 ----AISHA OMER

ORGANIZATION OF LIFE

Elements of life q. Elements of Life: Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, Sulfur. ØThe elements of life are the element which make up 99% of the biomolecules which are found in living organism. ØThis means 99% of your body, a tree, a mushroom, and even bacteria are made up of CHNOPS.

Atoms form compounds in two ways: 1. Ionic bonds – consists of ions and forms through the electrical force between oppositely charged ions An ion is an atom that has lost or gained electrons Cation – an ion that loses electrons so becomes positively charged Anion – an ion that gains electrons so becomes negatively charged 2. Covalent bonds – forms when atoms share one or more pairs of electrons A molecule consists of two or more atoms held together by covalent bonds

2/25/2021 4: 45 PM 5
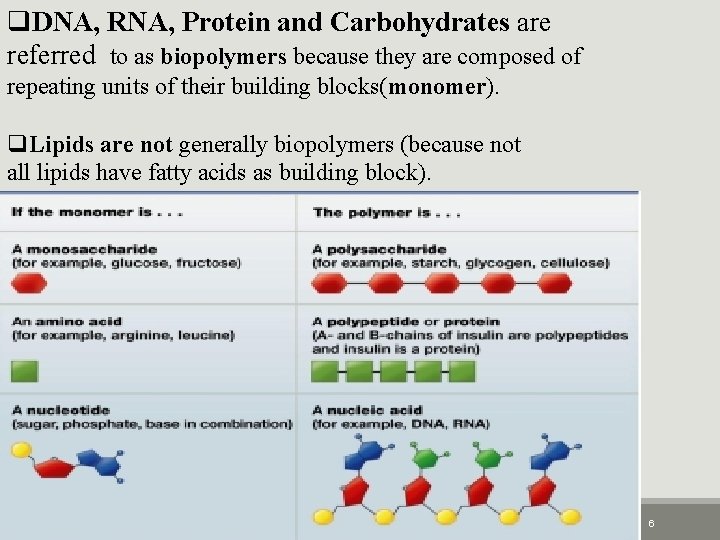
q. DNA, RNA, Protein and Carbohydrates are referred to as biopolymers because they are composed of repeating units of their building blocks(monomer). q. Lipids are not generally biopolymers (because not all lipids have fatty acids as building block). 2/25/2021 4: 45 PM 6
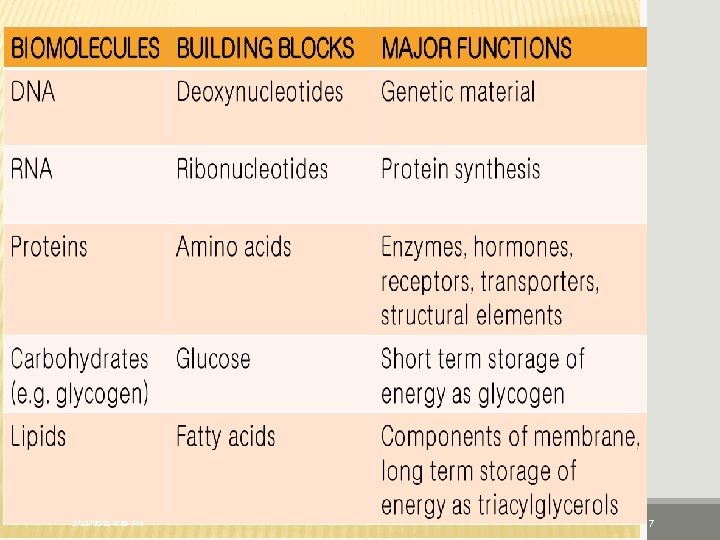
2/25/2021 4: 45 PM 7
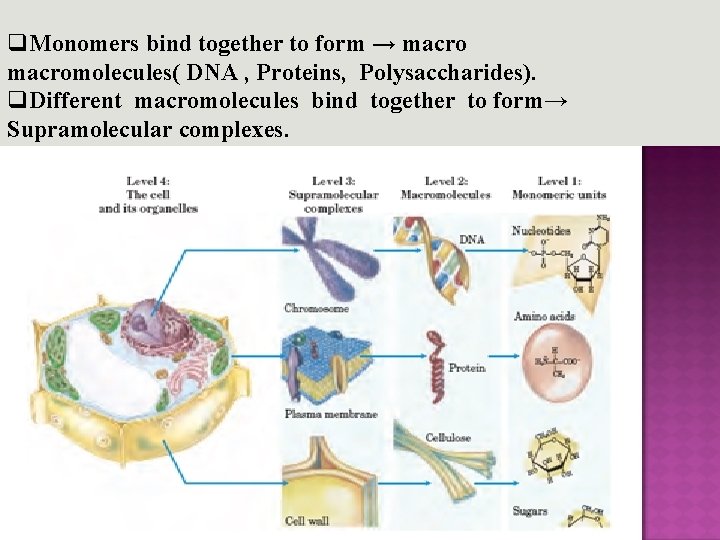
q. Monomers bind together to form → macromolecules( DNA , Proteins, Polysaccharides). q. Different macromolecules bind together to form→ Supramolecular complexes.
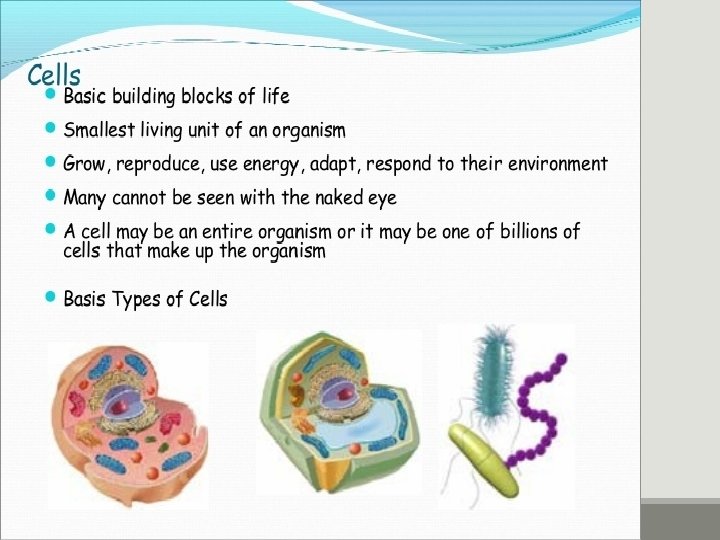

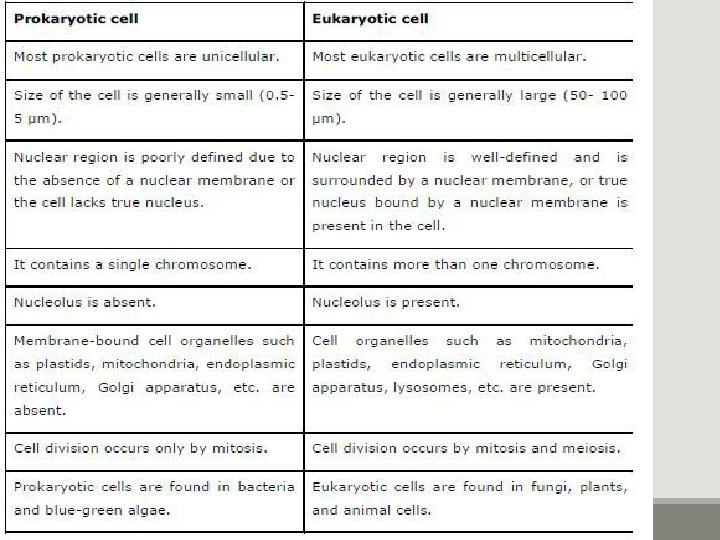
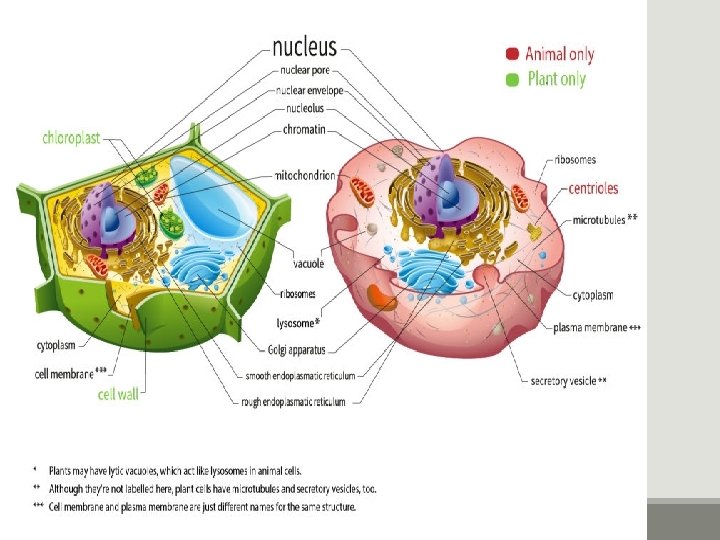
Major types of cell PROKARYOTIC & EUKARYOTIC CELLS PRO=Before, KARYON=Nucleus Prokaryotic cells lack a nucleus EU=True, KARYON=Nucleus Eukaryotic cells have a nucleus NUCLEUS: A complex membrane bound structure that contains genetic information in the form of chromosomes, whose principle constituent is DNA.





Thank you 2/25/2021 4: 45 PM 18
- Slides: 18