Introduction to Atoms Mrs Brostrom Integrated Science C

Introduction to Atoms Mrs. Brostrom Integrated Science C

http: //www. youtube. com/watch? v= jq 37 UQ-kr. W 4&feature=related 0: 50 • Atom- the smallest part of an element that has all the properties of that element – Extremely small – Aluminum foil 50, 000 atoms thick

What is an atom made of? • Nucleus: – An atom’s central region made up of protons and neutrons (small and dense) – A subatomic particle that has a positive charge is a proton – A subatomic particle that has no charge is a neutron

Subatomic Particles Protons: • A subatomic particle with a positive charge • Found in the nucleus • 1. 7 x 10 -24 g • 0. 00000000000017 g • About 1 amu The SI unit used to express the masses of atoms is called the atomic mass unit (amu)
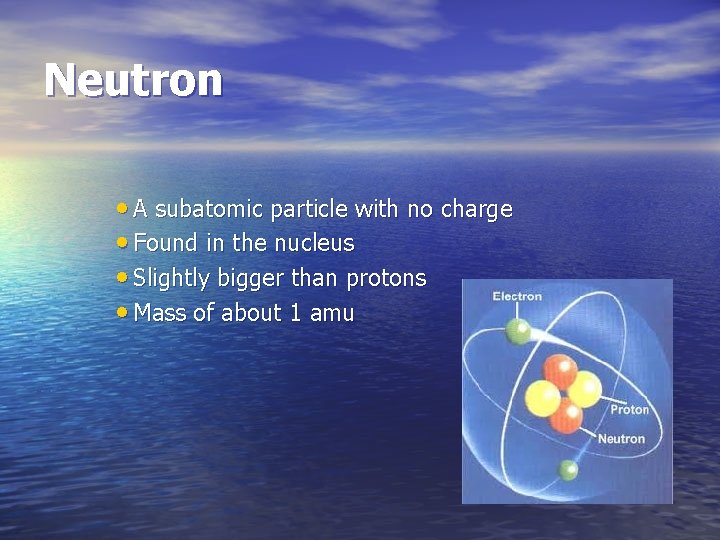
Neutron • A subatomic particle with no charge • Found in the nucleus • Slightly bigger than protons • Mass of about 1 amu

Electron • a very light subatomic particle that has a negative charge • Found around the nucleus in an electron cloud. • 1800 electrons = 1 proton mass • Mass is so small it is negligible (or zero)

Atomic number – # of protons in nucleus (also the # of electrons in a neutral atom) – Each element has a different atomic # because it has a specific # of protons

Mass number – Equals the # of protons + neutrons (in the nucleus) – Mass # - atomic # = # of neutrons

• An electron has a certain energy that is determined by its path around the nucleus (energy level). Electrons with different amounts of energy exist in different energy levels The higher the energy level the more energy the electrons have

• First energy level: 2 electrons • Second energy level: 8 electrons • Third energy level: 18 electrons • Fourth energy level: 32 electrons

Rules for drawing atoms: 1. 2. 3. 4. 5. Atomic # = # protons mass number –atomic number = # neutrons # of electrons = # of protons The row (period) the element is on determines how many energy level the atom will need The group the element is in determines how many electrons will be in the outermost energy level

Guided Practice 16 S Sulfur 32. 066 Protons: _____ Neutrons: _____ Electrons: ______ How many energy shells will this have? ____ How many valence (outer) electrons does this element have? ____ Bohr Model:

Guided Practice 3 Li Lithium 6. 941 Protons: _____ Neutrons: _____ Electrons: ______ How many energy shells will this have? ____ How many valence (outer) electrons does this element have? ____ Bohr Model:

Guided Practice 10 Ne Neon 20. 180 Protons: _____ Neutrons: _____ Electrons: ______ How many energy shells will this have? ____ How many valence (outer) electrons does this element have? ____ Bohr Model:

The Atom Song http: //www. youtube. com/watch? v= v. Uz. TQWn-wf. E 3: 28

White Board Practice • Protons and neutrons are found in the ______ of an atom

• Electrons have a _____ charge

• The atomic number = The number of _______

• The number of protons = the number of ____

• The mass number equals The number of ____ + the number of _______

• Electrons exist in _______ around the nucleus
- Slides: 21