Introduction to Antibacterial Therapy Clinically Relevant Microbiology and

Introduction to Antibacterial Therapy: Clinically Relevant Microbiology and Antibiotic Use Edward L. Goodman, MD Hospital Epidemiologist Core Faculty July 11, 2013

Outline l Basic Clinical Bacteriology l Antibiotics – Categories – Pharmacology – Mechanisms of Resistance l Antibiotic – “Pearls” Stewardship

Scheme for the Four Major Classes of Bacterial Pathogens in Hospitalized Patients l Gram Positive Cocci l Gram Negative Rods l Fastidious Gram Negative Organisms l Anaerobes

Gram Positive Cocci Gram stain: clusters l Catalase pos = Staph l Coag pos = S aureus l Coag neg = variety of species l Chains and pairs l Catalase neg = streptococci l Classify by hemolysis l Type by specific CHO l

Staphylococcus aureus >95% produce penicillinase (beta lactamase) = penicillin resistant l At PHD ~53% of SA are hetero (methicillin) resistant = MRSA (less than national average) l Glycopeptide (vancomycin) intermediate (GISA) l – MIC 8 -16 – Eight nationwide l First VRSA reported July 5, 2002 MMWR – Seven isolates reported (5/7 from Michigan) – MICs 32 - >128 – No evidence of spread w/in families or hospital

Coagulase Negative Staph l Many species – S. epidermidis most common l Mostly methicillin resistant (65 -85%) l Often contaminants or colonizers – use specific criteria to distinguish – Major cause of overuse of vancomycin l S. lugdunensis is rarely a contaminant – Causes destructive endocarditis

Streptococci l Beta hemolysis: Group A, B, C etc. l Invasive – mimic staph in virulence l S. pyogenes (Group A) – Pharyngitis, – Soft tissue Invasive l TSS l – Non suppurative sequellae: ARF, AGN

Other Beta hemolytic l S. agalactiae (Group B) – Peripartum/Neonatal – Diabetic foot – Bacteremia/endocarditis/metastatic foci l Group C/G Streptococcus – large colony variants: similar clinical illness as GAS plus bacteremia, endocarditis, septic arthritis – Small colony variants = Strept milleri

Viridans group l Anginosus sp. l Bovis sp. : Group D l Mutans sp. l Salivarius sp. l Mitis sp.

Enterococci l Formerly considered Group D Streptococci now a separate genus – Bacteremia without IE does not need cidal/syngergistic therapy – Endocarditis does need cidal/syngergistic – Bacteriuria in elderly, obstructed – Part of mixed abdominal/pelvic infections l Role in mixed flora intra-abdominal infection trivial- therapy for 2° peritonitis need not cover it Intrinsically resistant to cephalosporins l No bactericidal single agent l – For endocarditis need pen/amp/vanc plus AG – Daptomycin is cidal in vitro l l Little experience in endocarditis Resistance develops (NEJM Aug 25, 2011)

Gram Negative Rods l l l Fermentors Oxidase negative Facultative anaerobes Enteric flora Numerous genera – Escherischia – Enterobacter – Serratia, etc l UTI, IAI, LRTI, 2°B Non-fermentors l Pure aerobes l Pseudomonas (oxidase +) and Acinetobacter (oxidase -) l – Nosocomial LRTI, bacteremia, UTI – Opportunistic – Inherently resistant l New mechanisms of MDR emerging

Fastidious Gram Negatives Neisseria, Hemophilus, Moraxella, HACEK l Growth requirements l – CO² and enrichment l Culture for Neisseria must be plated at bedside – Chocolate agar with CO 2 – Ligase chain reaction (like PCR) has reduced number of GU cultures for N. gonorrhea l l Can’t do MIC without culture (at reference lab only) FQ resistance 13% in 2011 – FQ not recommended for empiric Rx since 2007

Anaerobes Gram negative rods – Bacteroides (gut/gu flora) – Fusobacteria (oral and gut) – Prevotella (mostly oral) l Gram positive rods – Clostridia (gut) – Proprionobacteria (skin) l Gram positive cocci – Peptostreptococci and peptococci (oral, gut, gu) l

Anaerobic Gram Negative Rods l Fastidious l Produce beta lactamase l Endogenous flora l When to consider – Part of mixed infections – Confer foul odor – Heterogeneous morphology – Gram stain shows GNR but routine cults negative

(My) Antibiotic Classification l Narrow Spectrum – Active against only one of the four classes of bacteria l Broad Spectrum – Active against more than one of the classes

Narrow Spectrum l Active mostly against only one of the classes of bacteria – gram positive: glycopeptides, linezolid, daptomycin, telavancin – aerobic gram negative: aminoglycosides, aztreonam – anaerobes: metronidazole
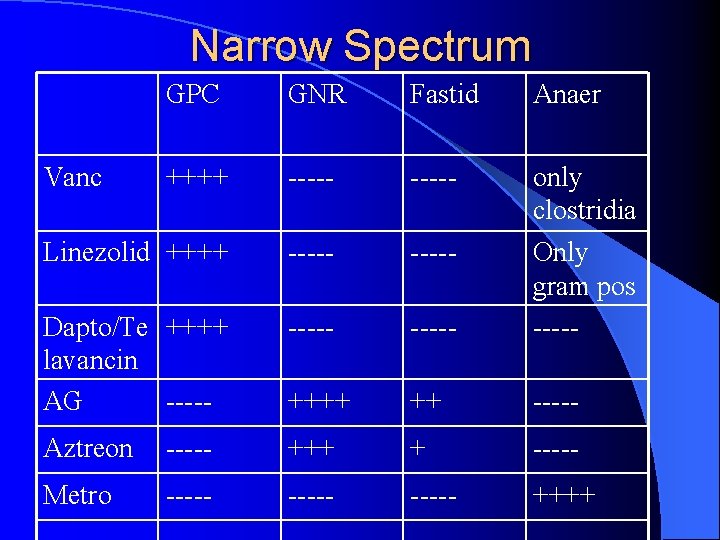
Narrow Spectrum GPC GNR Fastid Anaer ++++ ----- Linezolid ++++ ----- Dapto/Te ++++ lavancin AG ----- only clostridia Only gram pos ----- ++++ ++ ----- Aztreon ----- +++ + ----- Metro ----- ++++ Vanc
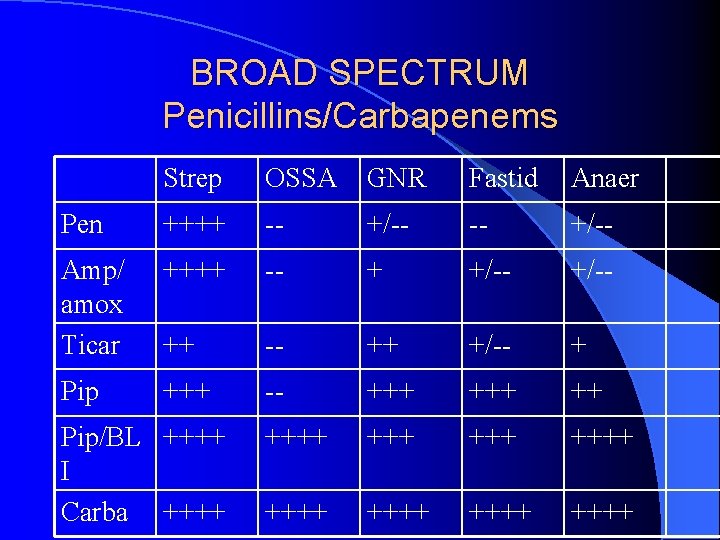
BROAD SPECTRUM Penicillins/Carbapenems Strep OSSA GNR Fastid Anaer Pen ++++ -- +/-- Amp/ amox Ticar ++++ -- + +/-- ++ +/-- + Pip +++ -- +++ ++++ ++++ Pip/BL ++++ I Carba ++++
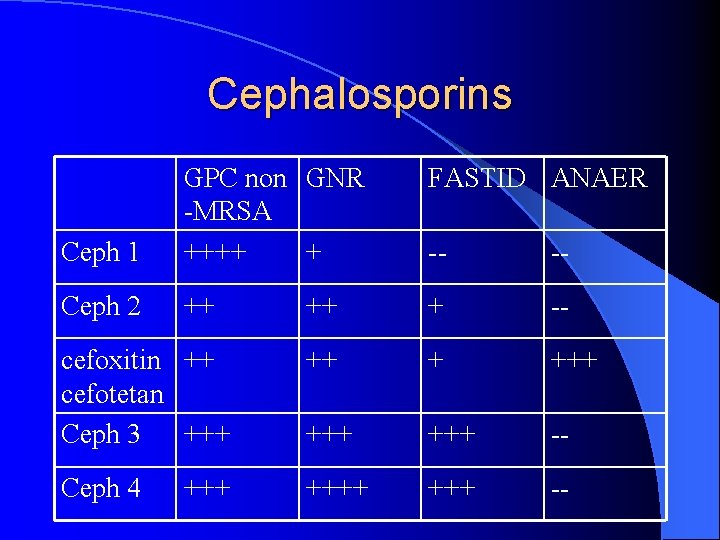
Cephalosporins FASTID ANAER Ceph 1 GPC non GNR -MRSA ++++ + -- -- Ceph 2 ++ ++ + -- cefoxitin ++ cefotetan Ceph 3 +++ +++ +++ -- Ceph 4 ++++ -- +++

Pharmacodynamics l MIC=lowest concentration to inhibit growth l MBC=the lowest concentration to kill l Peak=highest serum level after a dose l AUC=area under the concentration time curve l PAE=persistent suppression of growth following exposure to antimicrobial
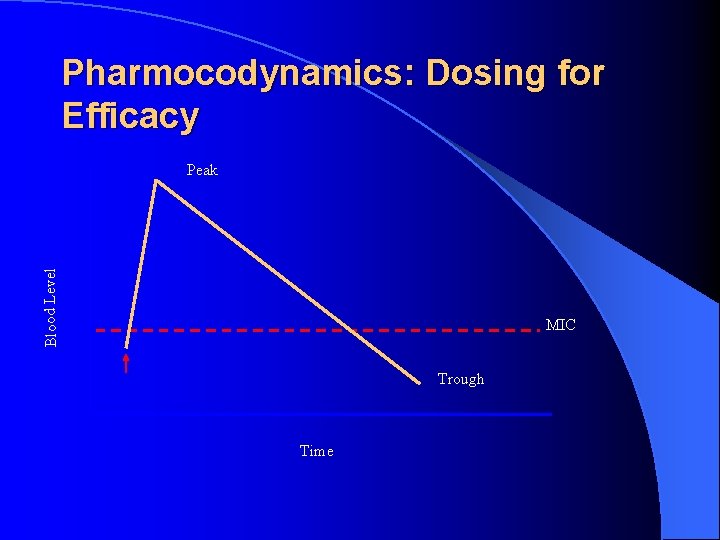
Pharmocodynamics: Dosing for Efficacy Blood Level Peak MIC Trough Time

Parameters of antibacterial efficacy Time above MIC (non concentration killing) - beta lactams, macrolides, clindamycin, glycopeptides l 24 hour AUC/MIC - aminoglycosides, fluoroquinolones, azalides, tetracyclines, glycopeptides, quinupristin/dalfopristin l Peak/MIC (concentration dependent killing) aminoglycosides, fluoroquinolones, daptomycin, l

l l Time over MIC For beta lactams, should exceed MIC > 50% of dose interval Higher doses may allow adequate time over MIC For most beta lactams, optimal time over MIC can be achieved by continuous infusion (except temperature labile drugs such as imipenem, ampicillin) For Vancomycin, evolving consensus that troughs should be >15 for most serious MRSA infections, especially pneumonia and bacteremia – If MRSA MIC >= 2 and patient responding slowly or poorly, should change vancomycin to daptomycin, linezolid or tigecycline – Few THD MRSA have MIC >1

Higher Serum/tissue levels are associated with faster killing l Aminoglycosides – Peak/MIC ratio of >10 -12 optimal – Achieved by “Once Daily Dosing” – PAE helps l Fluoroquinolones – 10 -12 ratio achieved for enteric GNR l PAE helps – not achieved for Pseudomonas – Not always achieved for Streptococcus pneumoniae l Daptomycin – Dose on actual body weight

FQ AUC/MIC = AUIC l For Streptococcus pneumoniae, FQ should have AUIC >= 30 l For gram negative rods where Peak/MIC ratio of 10 -12 not possible, then FQ AUIC should >= 125 l For MRSA, vancomycin AUIC needs to be >=400. Not easily achieved when MIC >=2.

A Brief Overview of Antimicrobial Resistance

ESKAPE Organisms (mechanism) l Enterococcus faecium VRE (Van A) l Staphylococcus aureus MRSA (MEC A) l Klebsiella pneumoniae (ESBL – KPC) l Acinetobacter baumanii (KPC – NDM 1) l Pseudomonas aeruginosa(Amp. C, KPC, NDM-1) l Enterobacter species (Amp. C)

Mechanisms of Antimicrobial Resistance in Bacteria FC Tenover Amer J Med 2006; 119: S 3 -10
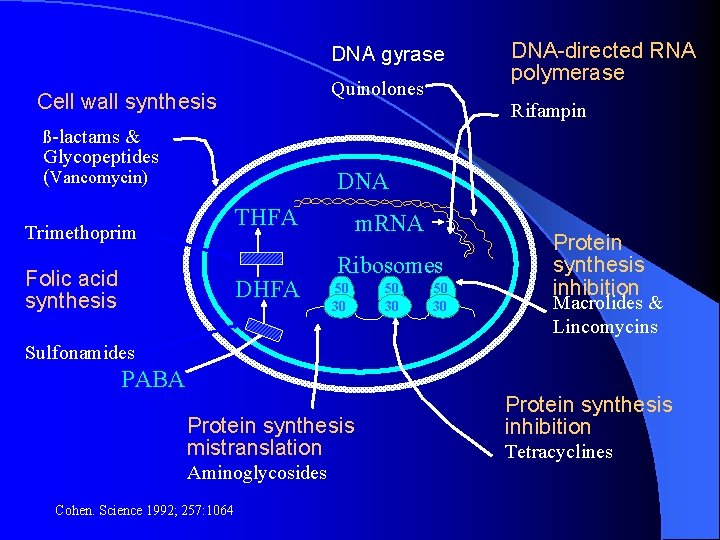
DNA gyrase Quinolones Cell wall synthesis DNA-directed RNA polymerase Rifampin ß-lactams & Glycopeptides (Vancomycin) DNA THFA Trimethoprim Folic acid synthesis DHFA m. RNA Ribosomes 50 30 Protein synthesis inhibition Macrolides & Lincomycins Sulfonamides PABA Protein synthesis mistranslation Aminoglycosides Cohen. Science 1992; 257: 1064 Protein synthesis inhibition Tetracyclines
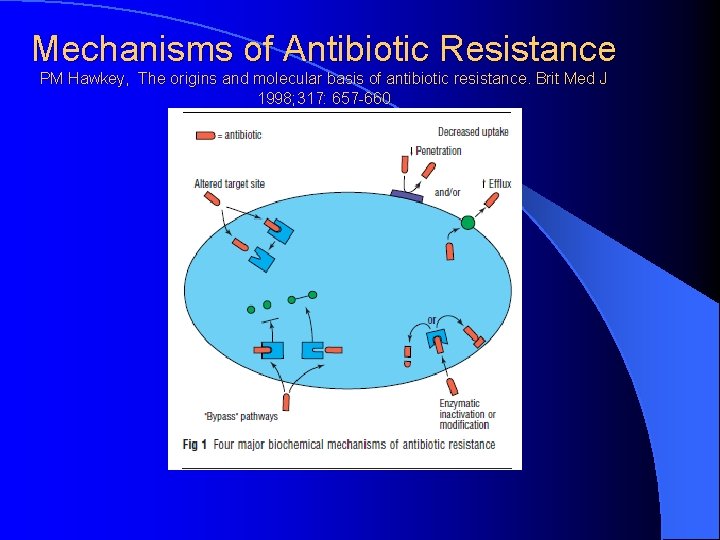
Mechanisms of Antibiotic Resistance PM Hawkey, The origins and molecular basis of antibiotic resistance. Brit Med J 1998; 317: 657 -660

Interplay of β lactam antibiotics and bacteria PM Hawkey, The origins and molecular basis of antibiotic resistance. Brit Med J 1998; 317: 657 -660

Bad Beta Lactamases (for dummies like me) ESBL – Klebsiella and E coli – Require carbapenems although for UTI Pip/tazo might work – Not clear how transmissible but use Contact Isolation l AMP C – SPICE organisms l Inducible/derepressed chromosomal beta lactamases – Requires carbapenems when AMP C expressed – Do not require Contact Isolation unless associated plasmid transmits MDR l

Really Bad Beta Lactamases l Carbapenem Resistant Enterobacteraciae (CRE) – Resistant to everything but colistin and sometimes tigecycline l New Delhi Metalloproteinases (NDM) – Pseudomonas and enterobacteraciae – Resistant to all but colistin l These patients require Contact Isolation and Cohorting

Antibiotic Use and Resistance l Strong epidemiological evidence that antibiotic use in humans and animals associated with increasing resistance l Subtherapeutic dosing encourages resistant mutants to emerge; conversely, rapid bactericidal activity discourages l Hospital antibiotic control programs have been demonstrated to reduce resistance

Antibiotic Armageddon “There is only a thin red line of ID practitioners who have dedicated themselves to rational therapy and control of hospital infections” Kunin CID 1997; 25: 240

When to Cover for MRSA l Severe purulent SSTI l Necrotizing pneumonia/empyema l Central line associated l (Known MRSA carriers? ) l Go To Drug = Vancomycin

Is Vancomycin Needed for every patient with SSTI? CID 2011: 1 -38

When to Cover for Pseudomonas l Severe COBPD/bronchiectasis – Frequent ABX – Steroid dependent – Known airway colonization l Neutropenic septic leukemic l (Burn patients)

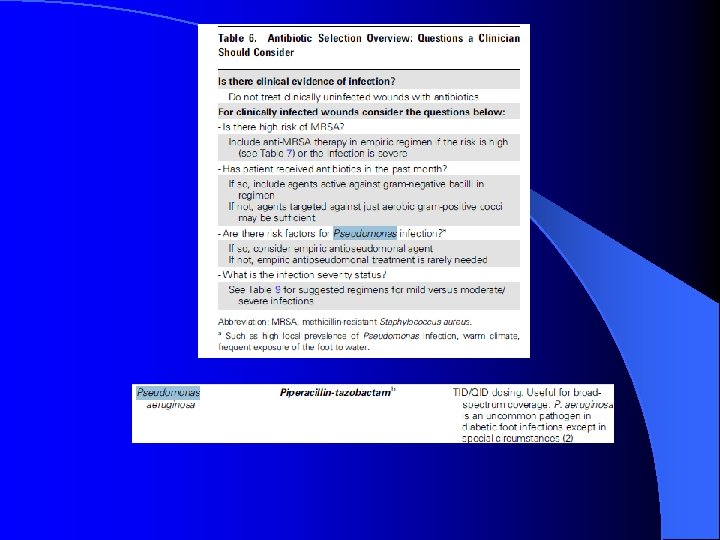
Is Pseudomonas Coverage Needed for Every Diabetic Foot Infection? CID 2012; 54 (12): 132 -173

Historic overview on treatment of infections l 2000 BC: Eat this root l 1000 AD: Say this prayer l 1800’s: Take this potion l 1940’s: Take penicillin, it is a miracle drug l 1980’s – 2000’s: Take this new antibiotic, it is a bigger miracle! l ? 2014: Eat this root!

Thanks to l Shahbaz Hasan, MD for allowing me to use slides from his 6/6/07 Clinical Grand Rounds on Streptococci l Eliane S Haron, MD for allowing me to use the “Eat this root” slide l Terri Smith, Pharm. D for collecting data from the Antibiotic Stewardship Program
- Slides: 43