Introduction to Antibacterial Therapy Clinically Relevant Microbiology and

Introduction to Antibacterial Therapy Clinically Relevant Microbiology and Antibiotic Use Edward L. Goodman, MD July 22, 2010

Outline l Basic Clinical Bacteriology l Antibiotics – Categories – Pharmacology – Mechanisms of Resistance

Goodman’s Scheme for the Major Classes of Bacterial Pathogens l Gram Positive Cocci l Gram Negative Rods l Fastidious Gram Negative Organisms l Anaerobes

Gram Positive Cocci Gram stain: clusters l Catalase pos = Staph l Coag pos = S aureus l Coag neg = variety of species l Chains and pairs l Catalase neg = streptococci l Classify by hemolysis l Type by specific CHO l

Staphylococcus aureus >95% produce penicillinase (beta lactamase) = penicillin resistant l At PHD ~60% of SA are hetero (methicillin) resistant = MRSA (less than national average) l Glycopeptide (vancomycin) intermediate (GISA) l – MIC 8 -16 – Eight nationwide l First VRSA reported July 5, 2002 MMWR – Seven isolates reported (5/7 from Michigan) – MICs 32 - >128 – No evidence of spread w/in families or hospital
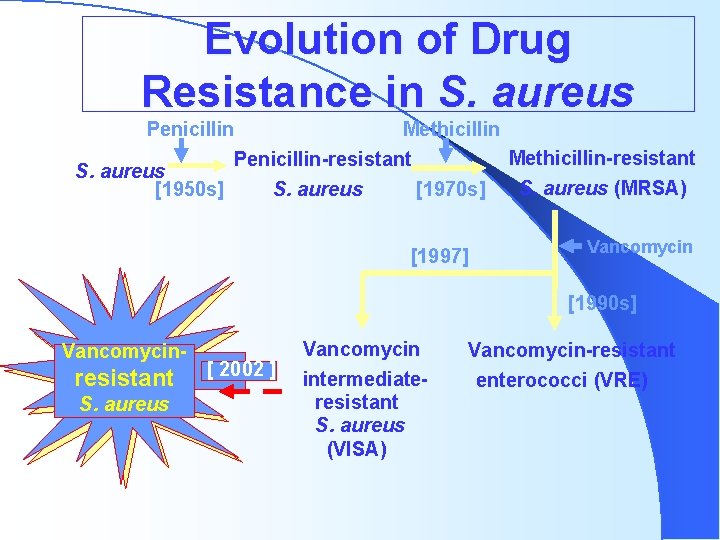
Evolution of Drug Resistance in S. aureus Penicillin Methicillin-resistant Penicillin-resistant S. aureus (MRSA) [1970 s] [1950 s] S. aureus [1997] Vancomycin [1990 s] Vancomycin- resistant S. aureus [ 2002 ] Vancomycin intermediateresistant S. aureus (VISA) Vancomycin-resistant enterococci (VRE)
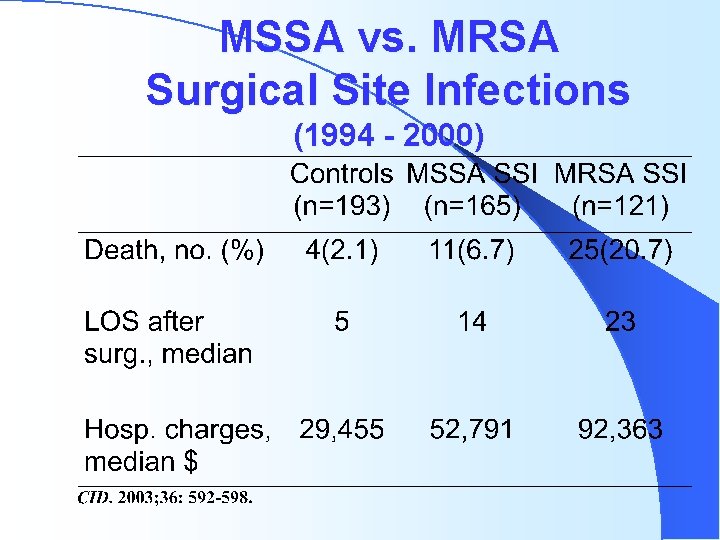
MSSA vs. MRSA Surgical Site Infections (1994 - 2000)

Coagulase Negative Staph l Many species – S. epidermidis most common l Mostly methicillin resistant (65 -85%) l Often contaminants or colonizers – use specific criteria to distinguish – Major cause of overuse of vancomycin l S. lugdunensis is rarely a contaminant – Causes destructive endocarditis
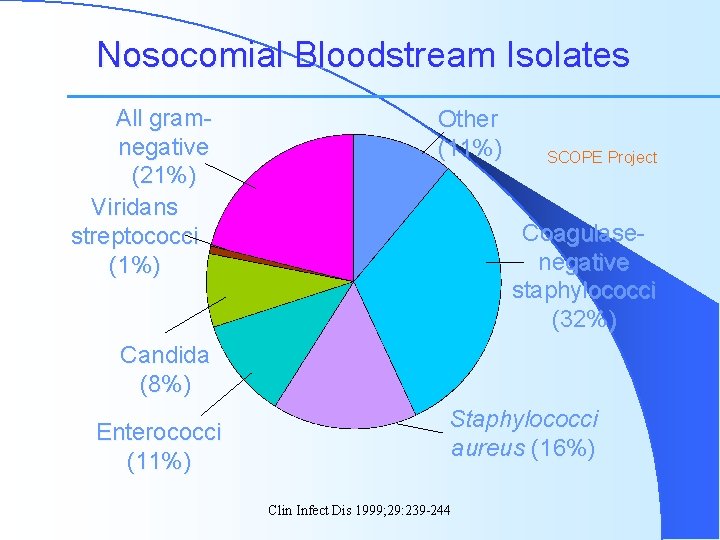
Nosocomial Bloodstream Isolates All gramnegative (21%) Viridans streptococci (1%) Other (11%) SCOPE Project Coagulasenegative staphylococci (32%) Candida (8%) Enterococci (11%) Staphylococci aureus (16%) Clin Infect Dis 1999; 29: 239 -244

Streptococci l Beta hemolysis: Group A, B, C etc. l Invasive – mimic staph in virulence l S. pyogenes (Group A) – Pharyngitis, – Soft tissue Invasive l TSS l – Non suppurative sequellae: ARF, AGN

Other Beta hemolytic l S. agalactiae (Group B) – Peripartum/Neonatal – Diabetic foot – Bacteremia/endocarditis/metastatic foci l Group C/G Streptococcus – large colony variants: similar clinical illness as GAS plus bacteremia, endocarditis, septic arthritis – Small colony variants = Strept milleri

Viridans group l Anginosus sp. l Bovis sp. : Group D l Mutans sp. l Salivarius sp. l Mitis sp.

Streptococcus anginosus Group l Formerly ‘Streptococcus milleri’ or ‘Streptococcus intermedius’. l S. intermedius; S. constellatus; S. anginosus l Oral cavity, nasopharynx, GI and genitourinary tract.

S. anginosus Group Propensity for invasive pyogenic infections ie. abscesses. l Grow well in acidic environment l polysaccharide capsule resists phagocytosis l produce hydrolytic enzymes: hyaluronidase, deoxyribonucleotidase, chondroitin sulfatase, sialidase

S. anginosus Group l Oral and maxillofacial infections l Brain, epidural and subdural abscesses l intraabdominal abscesses l empyema and lung abscesses l bacteremias usually secondary to an underlying focus of infection. l Look for the Abscess!

Enterococci l l l l Formerly considered Group D Streptococci now a separate genus Bacteremia/Endocarditis Bacteriuria Part of mixed abdominal/pelvic infections Intrinsically resistant to cephalosporins No bactericidal single agent (except ? Dapto) Role in mixed flora intra-abdominal infection trivial- therapy for 2° peritonitis need not cover

Gram Negative Rods l l l Fermentors Oxidase negative Facultative anaerobes Enteric flora Numerous genera – Escherischia – Enterobacter – Serratia, etc l UTI, IAI, LRTI, 2°B Non-fermentors l Pure aerobes l Pseudomonas (oxidase +) and Acinetobacter (oxidase -) l – Nosocomial LRTI, bacteremia, UTI – Opportunistic – Inherently resistant

Fastidious Gram Negatives Neisseria, Hemophilus, Moraxella, HACEK l Require CO 2 for growth l Culture for Neisseria must be plated at bedside l – Chocolate agar with CO 2 – Ligase chain reaction (like PCR) has reduced number of GU cultures for N. gonorrhea l l Can’t do MIC without culture Increasing resistance to FQ not detected w/o culture

Anaerobes Gram negative rods – Bacteroides (gut/gu flora) – Fusobacteria (oral and gut) – Prevotella (mostly oral) l Gram positive rods – Clostridia (gut) – Proprionobacteria (skin) l Gram positive cocci – Peptostreptococci and peptococci (oral, gut, gu) l

Anaerobic Gram Negative Rods l Fastidious l Produce beta lactamase l Endogenous flora l When to consider – Part of mixed infections – Confer foul odor – Heterogeneous morphology – Gram stain shows GNR but routine cults negative

Antibiotic Classification according to Goodman l Narrow Spectrum – Active against only one of the four classes of bacteria l Broad Spectrum – Active against more than one of the classes l Boutique – Highly specialized use – Restricted to ID physicians

Narrow Spectrum l Active mostly against only one of the classes of bacteria – gram positive: glycopeptides, linezolid, daptomycin, telavancin – aerobic gram negative: aminoglycosides, aztreonam – anaerobes: metronidazole
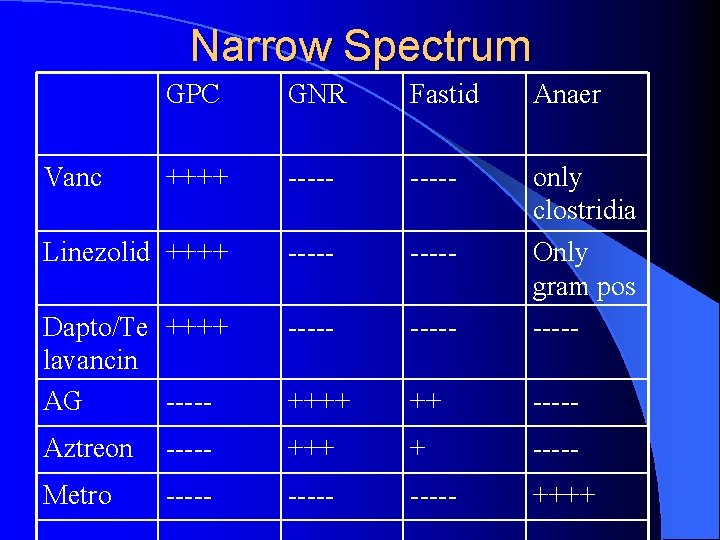
Narrow Spectrum GPC GNR Fastid Anaer ++++ ----- Linezolid ++++ ----- Dapto/Te ++++ lavancin AG ----- only clostridia Only gram pos ----- ++++ ++ ----- Aztreon ----- +++ + ----- Metro ----- ++++ Vanc

Broad Spectrum Active against more than one class GPC (incl many MRSA) and anaerobes: clindamycin l GPC (not MRSA*) and GNR: cephalosporins, penicillins, sulfonamides, TMP/Sulfa (*include MRSA), FQ l GPC (not MRSA*), GNR and anaerobes: ureidopenicillins + BLI, carbapenems, tigecycline (*MRSA), tetracyclines (*MRSA), moxiflox l GPC and fastidious: macrolides l l
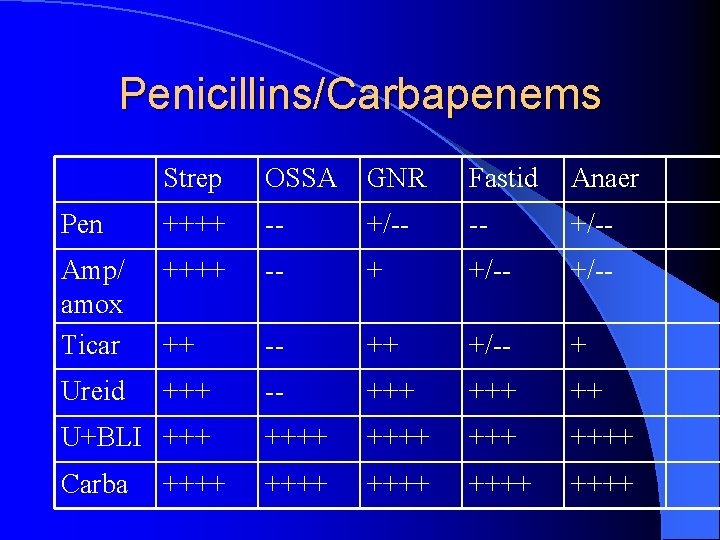
Penicillins/Carbapenems Strep OSSA GNR Fastid Anaer Pen ++++ -- +/-- Amp/ amox Ticar ++++ -- + +/-- ++ +/-- + Ureid +++ -- +++ ++ U+BLI ++++ +++ ++++ Carba ++++ ++++
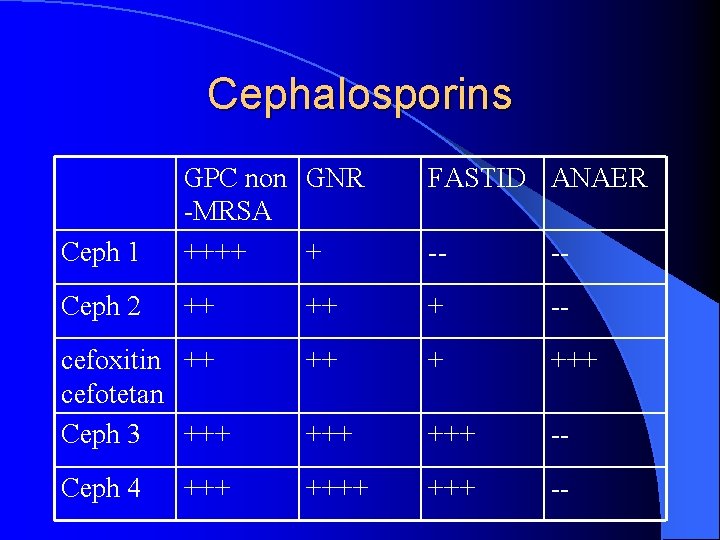
Cephalosporins FASTID ANAER Ceph 1 GPC non GNR -MRSA ++++ + -- -- Ceph 2 ++ ++ + -- cefoxitin ++ cefotetan Ceph 3 +++ +++ +++ -- Ceph 4 ++++ -- +++

Pharmacodynamics l MIC=lowest concentration to inhibit growth l MBC=the lowest concentration to kill l Peak=highest serum level after a dose l AUC=area under the concentration time curve l PAE=persistent suppression of growth following exposure to antimicrobial
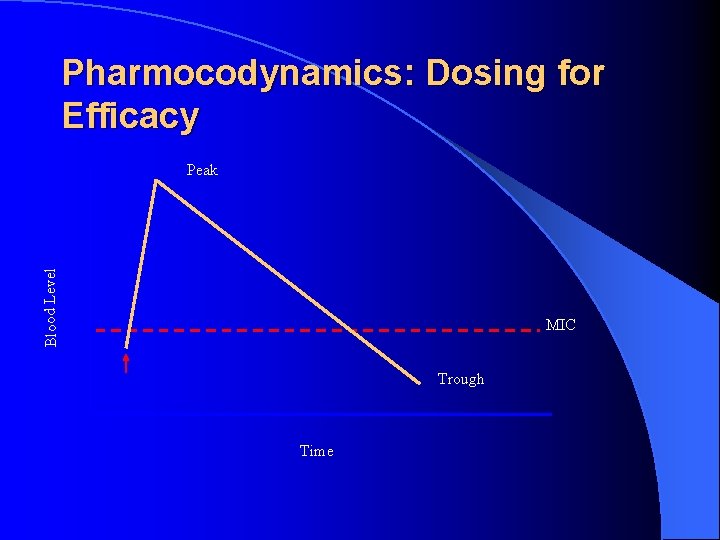
Pharmocodynamics: Dosing for Efficacy Blood Level Peak MIC Trough Time

Parameters of antibacterial efficacy Time above MIC (non concentration killing) - beta lactams, macrolides, clindamycin, glycopeptides l 24 hour AUC/MIC - aminoglycosides, fluoroquinolones, azalides, tetracyclines, glycopeptides, quinupristin/dalfopristin l Peak/MIC (concentration dependent killing) aminoglycosides, fluoroquinolones, daptomycin l

Time over MIC For beta lactams, should exceed MIC for at least 50% of dose interval l Higher doses may allow adequate time over MIC l For most beta lactams, optimal time over MIC can be achieved by continuous infusion (except unstable drugs such as imipenem, ampicillin) l For Vancomycin, evolving consensus that troughs should be >15 for most serious MRSA infections, especially pneumonia and bacteremia l – If MRSA MIC is 1. 5 - 2, should avoid vancomycin in favor of daptomycin, linezolid or tigecycline

Higher Serum/tissue levels are associated with faster killing l Aminoglycosides – Peak/MIC ratio of >10 -12 optimal – Achieved by “Once Daily Dosing” – PAE helps l Fluoroquinolones – 10 -12 ratio achieved for enteric GNR l PAE helps – not achieved for Pseudomonas – Not always achieved for Streptococcus pneumoniae l Daptomycin – Dose on actual body weight

AUC/MIC = AUIC l For Streptococcus pneumoniae, FQ should have AUIC >= 30 l For gram negative rods where Peak/MIC ratio of 10 -12 not possible, then AUIC should >= 125.
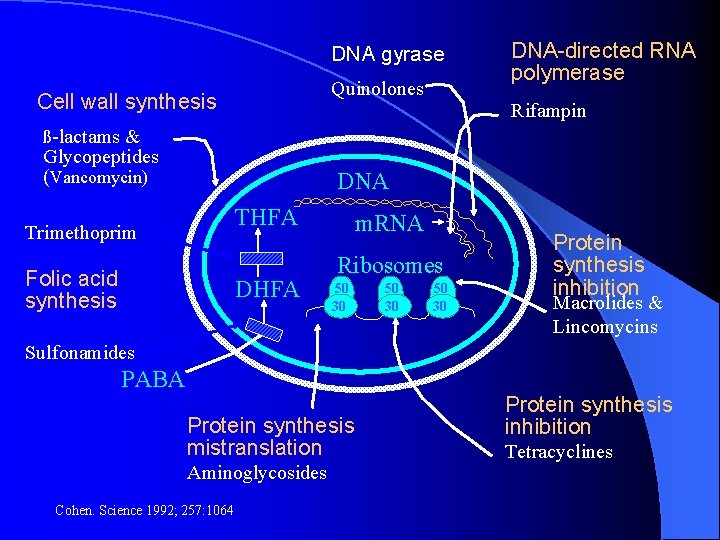
DNA gyrase Quinolones Cell wall synthesis DNA-directed RNA polymerase Rifampin ß-lactams & Glycopeptides (Vancomycin) DNA THFA Trimethoprim Folic acid synthesis DHFA m. RNA Ribosomes 50 30 Protein synthesis inhibition Macrolides & Lincomycins Sulfonamides PABA Protein synthesis mistranslation Aminoglycosides Cohen. Science 1992; 257: 1064 Protein synthesis inhibition Tetracyclines

Pathways of Common Resistance Mechanisms l Impede access of drug to target – – – l Beta lactamases: multiple classes Aminoglycoside altering enzymes Chloramphenicol altering enzymes Altered porin channels – carbapenems Efflux pumps - macrolides Alterations in target – – Altered binding proteins: MRSA, DRSP Methylation of ribosomes: macrolides Bypass metabolic pathways: TMP/Sulfa Alteration in gyrases

Some Background on Enterobacteriaceae β-lactam antibiotics (derivatives of penicillin) have long been the mainstay of treating infections caused by Enterobacteriaceae. l However, resistance to β-lactams emerged several years ago and has continued to rise. l – Extended spectrum β-lactamase producing Enterobacteriaceae (ESBLs) – Plasmid-mediated Amp. C-type enzymes

Extended Spectrum Beta Lactamases (ESBL) l Hyper production derived from TEM beta lactamases l Predominantly in Klebsiella and E coli l Confer resistance to penicillins, cephalosporins, monobactams – Plasmids also confers R to FQ/AG – Indications for carbapenems*

Amp C Beta Lactamases l Chromosomal cephalosporinases active against – 1 st - 3 rd generation cephalosporins, penicillins even with BLI l l Constituent or Inducible Reside in periplasmic space – Not easily detected when in low numbers l SPICE organisms possess Amp C – – – l Serratia Pseudomonas Indole + Proteii Citrobacter Enterobacter Indication for carbapenems* (imipenem, meropenem, ertapenem, doripenem)

The Last Line of Defense l Fortunately, our most potent β-lactam class, carbapenems, remained effective against almost all Enterobacteriaceae. Doripenem, Ertapenem, Imipenem, Meropenem l Unfortunately, “Antimicrobial resistance follows antimicrobial use as surely as night follows day”

Klebsiella Pneumoniae Carbapenemase l KPC is a class A b-lactamase – Confers resistance to all b-lactams including extended- spectrum cephalosporins and carbapenems l Occurs in Enterobacteriaceae – Most commonly in Klebsiella pneumoniae – Also reported in: K. oxytoca, Citrobacter freundii, Enterobacter spp. , Escherichia coli, Salmonella spp. , Serratia spp. , l Also reported in Pseudomonas aeruginosa (South America)

Susceptibility Profile of KPC-Producing K. pneumoniae Antimicrobial Interpretation Amikacin I Chloramphenicol R Amox/clav R Ciprofloxacin R Ampicillin R Ertapenem R Aztreonam R Gentamicin R Cefazolin R Imipenem R Cefpodoxime R Meropenem R Cefotaxime R Pipercillin/Tazo R Cetotetan R Tobramycin R Cefoxitin R Trimeth/Sulfa R Ceftazidime R Polymyxin B MIC >4μg/ml Ceftriaxone R Colistin MIC >4μg/ml Cefepime R Tigecycline S
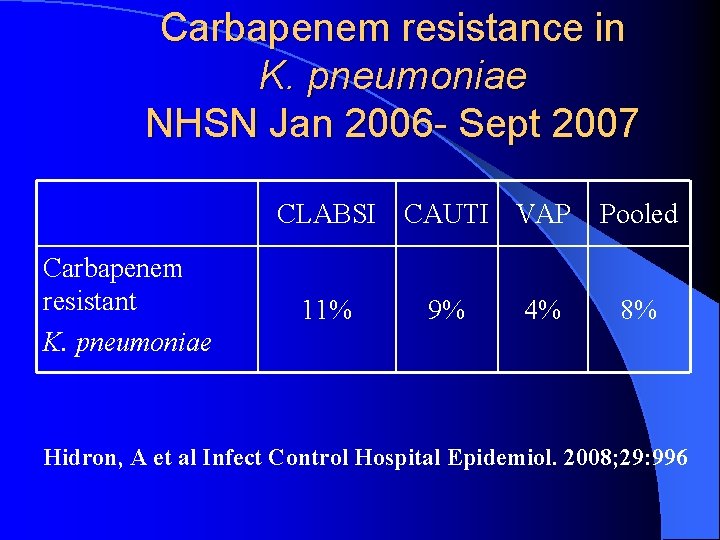
Carbapenem resistance in K. pneumoniae NHSN Jan 2006 - Sept 2007 CLABSI CAUTI VAP Carbapenem resistant K. pneumoniae 11% 9% 4% Pooled 8% Hidron, A et al Infect Control Hospital Epidemiol. 2008; 29: 996
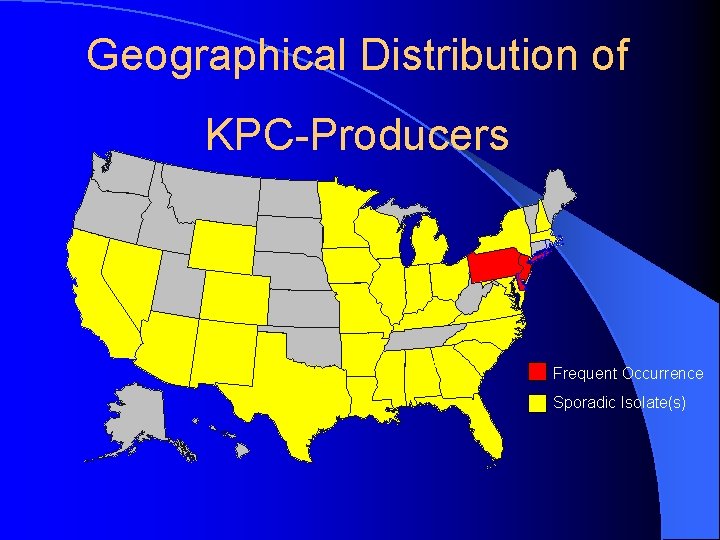
Geographical Distribution of KPC-Producers Frequent Occurrence Sporadic Isolate(s)

Antibiotic Use and Resistance l Strong epidemiological evidence that antibiotic use in humans and animals associated with increasing resistance l Subtherapeutic dosing encourages resistant mutants to emerge; conversely, rapid bactericidal activity discourages l Hospital antibiotic control programs have been demonstrated to reduce resistance

Historic overview on treatment of infections l 2000 BC: Eat this root l 1000 AD: Say this prayer l 1800’s: Take this potion l 1940’s: Take penicillin, it is a miracle drug l 1980’s – 2000’s: Take this new antibiotic, it is a bigger miracle! l ? 2011: Eat this root!

Antibiotic Armageddon “There is only a thin red line of ID practitioners who have dedicated themselves to rational therapy and control of hospital infections” Kunin CID 1997; 25: 240

Thanks to l Shahbaz Hasan, MD for allowing me to use slides from his 6/6/07 Clinical Grand Rounds on Streptococci l Eliane S Haron, MD for allowing me to use the “Eat this root” slide l Jean B. Patel, Ph. D and CDR Arjun Srinivasan, MD, Division of Healthcare Quality Promotion at CDC for Kpc slides
- Slides: 46