INTRODUCTION TO ACIDS BASES MRS ARIEL DANTON STUDENT

INTRODUCTION TO ACIDS & BASES MRS. ARIEL D’ANTON

STUDENT OBJECTIVE At the end of this lesson, student’s will be able to: Describe acids and bases by identifying similar characteristics Derive the definition of acids and bases through examination of its properties Classify solutions as acidic or basic through use of an indicator

DO NOW: 2 MINS CATEGORIZE THE ITEMS ON THIS SLIDE AS ACIDS OR BASES IF COMPLETED BEFORE 2 MINS: BRAINSTORM OTHER HOUSEHOLD EXAMPLES OF ACIDS & BASES
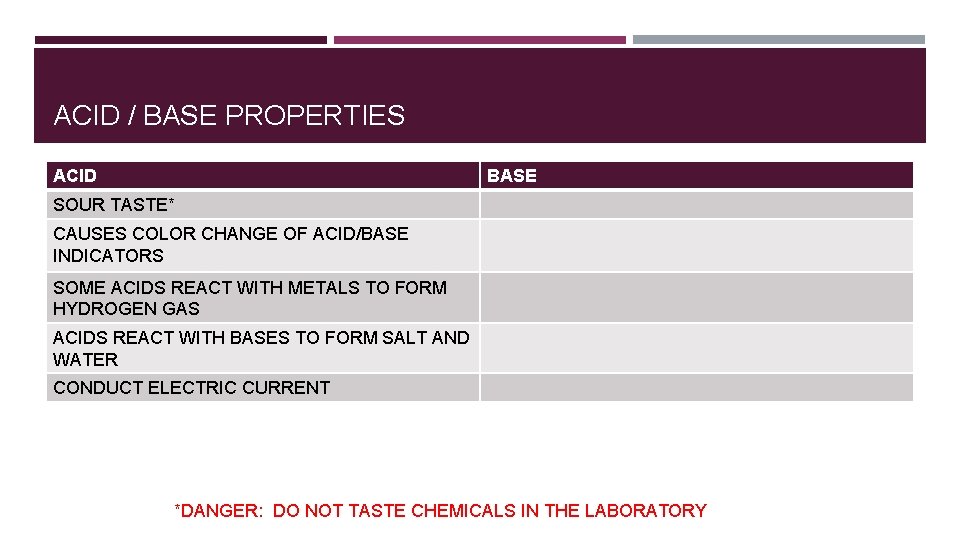
ACID / BASE PROPERTIES ACID BASE SOUR TASTE* CAUSES COLOR CHANGE OF ACID/BASE INDICATORS SOME ACIDS REACT WITH METALS TO FORM HYDROGEN GAS ACIDS REACT WITH BASES TO FORM SALT AND WATER CONDUCT ELECTRIC CURRENT *DANGER: DO NOT TASTE CHEMICALS IN THE LABORATORY

ACID / BASE PROPERTIES ACID BASE SOUR TASTE* BITTER TASTE* CAUSES COLOR CHANGE OF ACID/BASE INDICATORS SOME ACIDS REACT WITH METALS TO FORM HYDROGEN GAS DILUTE SOLUTIONS FEEL SLIPPERY ACIDS REACT WITH BASES TO FORM SALT AND BASES REACT WITH ACIDS TO FORM SALT AND WATER CONDUCT ELECTRIC CURRENT *DANGER: DO NOT TASTE CHEMICALS IN THE LABORATORY

ACID & BASE DEFINITIONS ACID: ARRHENIUS - SUBSTANCES THAT IONIZE IN WATER TO PRODUCE H+ IONS BASE: ARRHENIUS – SUBSTANCES THAT IONIZE IN WATER TO PRODUCE OH- IONS
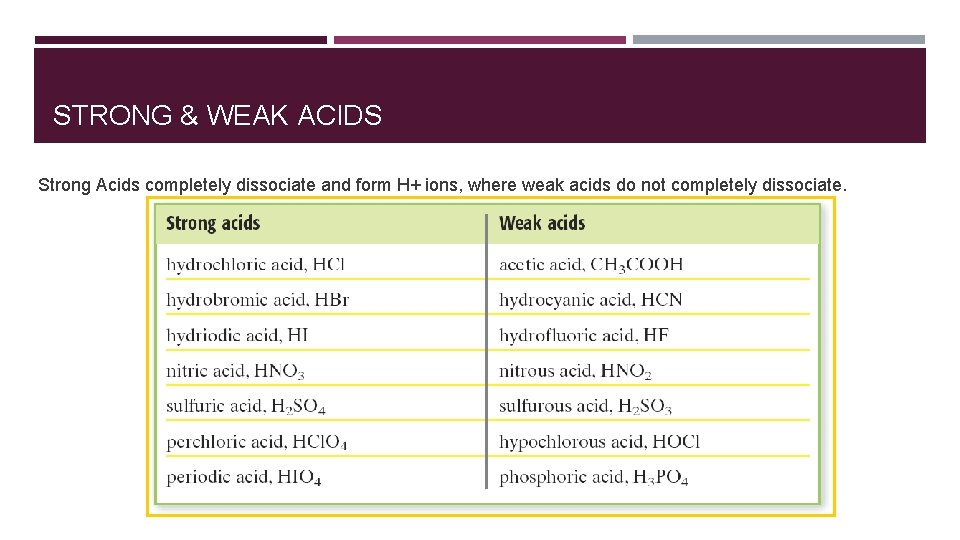
STRONG & WEAK ACIDS Strong Acids completely dissociate and form H+ ions, where weak acids do not completely dissociate.
![PH p. H: MEASUREMENT OF HYDRONIUM ION CONCENTRATION (measure of acidity) p. H=-log[H+] [H+]=10 PH p. H: MEASUREMENT OF HYDRONIUM ION CONCENTRATION (measure of acidity) p. H=-log[H+] [H+]=10](http://slidetodoc.com/presentation_image_h2/1315ad9d664dc695d9bcf68fae78d7b6/image-8.jpg)
PH p. H: MEASUREMENT OF HYDRONIUM ION CONCENTRATION (measure of acidity) p. H=-log[H+] [H+]=10 -p. H STRONG ACID 0 7 14 STRONG BASE

STRONG/WEAK BASES Strong bases completely ionize in water to form OH ions, where weak bases do not completely ionize in water.
![POH p. OH: MEASUREMENT OF HYDROXIDE CONCENTRATION (measure of alkalinity/basicity) p. OH=-log[OH-] [OH-]=10 -p. POH p. OH: MEASUREMENT OF HYDROXIDE CONCENTRATION (measure of alkalinity/basicity) p. OH=-log[OH-] [OH-]=10 -p.](http://slidetodoc.com/presentation_image_h2/1315ad9d664dc695d9bcf68fae78d7b6/image-10.jpg)
POH p. OH: MEASUREMENT OF HYDROXIDE CONCENTRATION (measure of alkalinity/basicity) p. OH=-log[OH-] [OH-]=10 -p. OH STRONG BASE 0 7 14 STRONG ACID ! ! 4 1 = H O p + H p
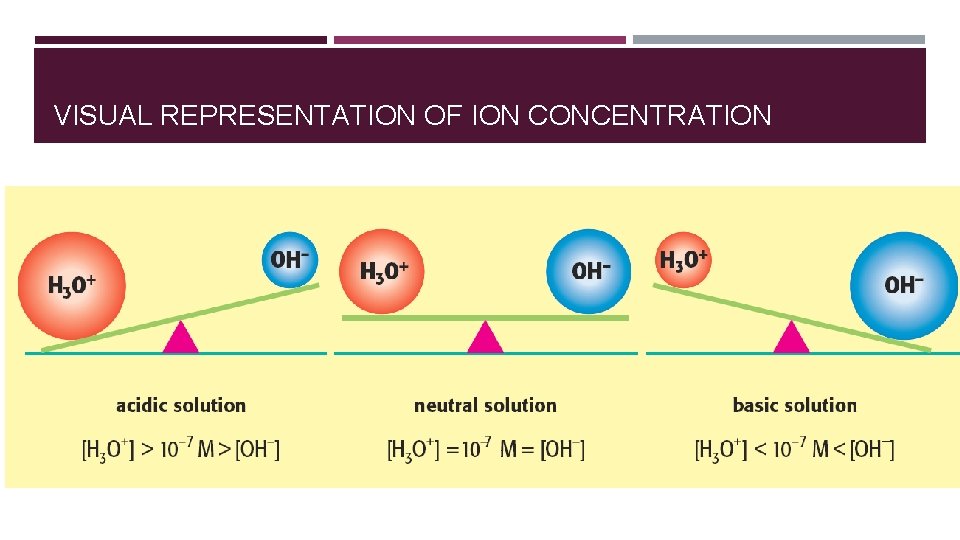
VISUAL REPRESENTATION OF ION CONCENTRATION

PH VALUES OF COMMON SUBSTANCES

INDICATORS & METERS • Certain dyes, known as indicators, turn different colors in solutions of different p. H. • • • An indicator a compound that can reversibly change color depending on the p. H of the solution or other chemical change. Thymol blue is an example of an indicator. • It is yellow in solutions whose p. H is between 3 and 8 but blue in solutions whose p. H is 10 or higher. • Universal indicator utilizes all colors of the rainbow, where red is acidic and violet basic • Litmus paper turns acidic conditions red, and basic conditions blue. • phenolphthalein is pink under basic conditions, and clear otherwise (used in titrations). p. H probes are also used to read concentration. The probe is inserted into the solution and using voltage, converts the value to a reading of p. H.

SUMMARIZING UNDERSTANDING A 4 x 6 index card has been provided to you. Please write the following: On the blank side, create a Venn Diagram listing everything you remember about acids and bases from today’s discussion (properties, definitions), & classify a new* household item that is an acid or base. On the lined side write down any clarifying question you have, or a topic you would like to know more about relating to acids & bases. Example: ACID SOUR TASTE BASE IONIZE IN WATER BITTER TASTE

LEARNER. ORG VIDEO QUESTIONS What is a hydronium ion? Is it a proton donor or proton acceptor? What is a hydroxide ion? Is it a proton donor or proton acceptor? What is dissociation? How can you make a neutral solution? What is the definition of a strong acid? What is the definition of a weak acid? Name an example of each. What is acid rain? What does the p. H scale represent? Provide some examples of the roles acids and bases play in every day life
- Slides: 15