Introduction to Acids and Bases Chapter 19 Sections

Introduction to Acids and Bases Chapter 19 Sections 1, 2, plus some of 3 and 4 Honors Chemistry Dr. Knorr Adapted from http: //www. chemistryconnections. com/acids%20 bases%20 intro. ppt 1

Some Common Acids HCl hydrochloric acid HNO 3 nitric acid H 3 PO 4 phosphoric acid H 2 SO 4 sulfuric acid HC 2 H 3 O 2 acetic acid (vinegar) lemon juice 2

Acids • Electrolytes • Taste sour • Corrode metals • React with many bases to form salts and water 3

Some Common Bases Na. OH sodium hydroxide KOH potassium hydroxide Mg(OH)2 magnesium hydroxide Al(OH)3 aluminum hydroxide NH 3 ammonia 4

Bases l. Taste bitter, chalky l. Are electrolytes l. Feel soapy, slippery l. React with many acids to form salts and water 5

Arrhenius Acids and Bases l Acids produce H+ in aqueous solutions HCl water H+(aq) + Cl- (aq) l Bases produce OH- in aqueous solutions water Na. OH Na+(aq) + OH- (aq) 6
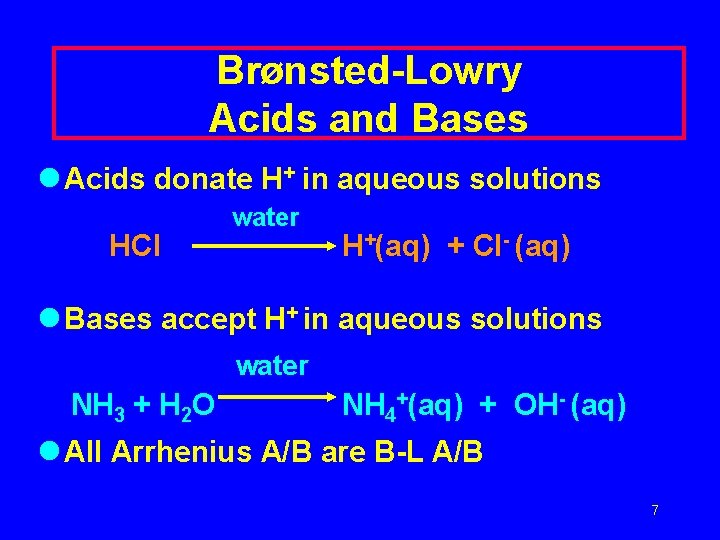
Brønsted-Lowry Acids and Bases l Acids donate H+ in aqueous solutions HCl water H+(aq) + Cl- (aq) l Bases accept H+ in aqueous solutions water NH 3 + H 2 O NH 4+(aq) + OH- (aq) l All Arrhenius A/B are B-L A/B 7

Lewis Acids and Bases l Acids accept electron pairs in aqueous solutions l Bases donate electron pairs in aqueous solutions H H N H F + B F F H H N H F B F F 8

Conjugate Acids and Bases A conjugate acid-base pair is related by loss/gain of one hydrogen ion. gain H+ NH 3 + H 2 O NH 4+(aq) + OH- (aq) Base conjugate acid conjugate base lose H+ 9
![p. H l Indicates the acidity [H+] of the solution l p. H = p. H l Indicates the acidity [H+] of the solution l p. H =](http://slidetodoc.com/presentation_image/414c16a21a36a6958ffee5a3859b5760/image-10.jpg)
p. H l Indicates the acidity [H+] of the solution l p. H = - log [H+] p. H 1 x 10 -5 M 5 1 x 10 -9 M 9 1 x 10 -11 M 11 10
![p. OH l Indicates the basicity (aka alkalinity) [OH-] of the solution l p. p. OH l Indicates the basicity (aka alkalinity) [OH-] of the solution l p.](http://slidetodoc.com/presentation_image/414c16a21a36a6958ffee5a3859b5760/image-11.jpg)
p. OH l Indicates the basicity (aka alkalinity) [OH-] of the solution l p. OH = - log [OH-] p. OH 1 x 10 -5 M 5 1 x 10 -9 M 9 1 x 10 -11 M 11 11
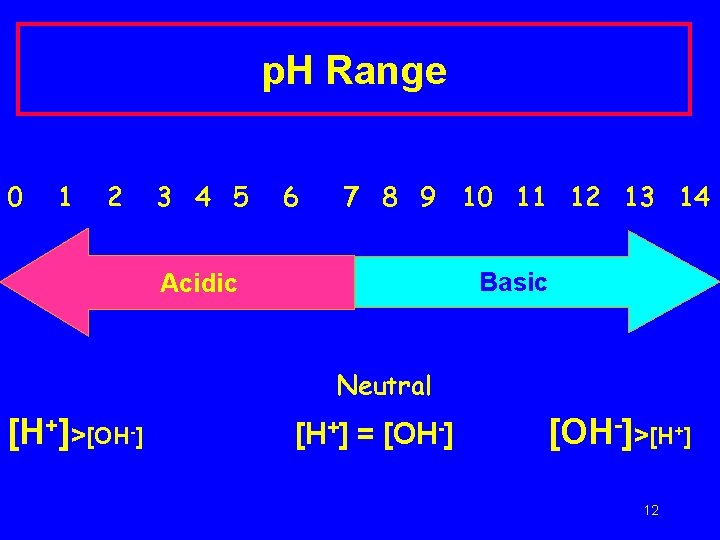
p. H Range 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Basic Acidic Neutral [H+]>[OH-] [H+] = [OH-]>[H+] 12

Why 0 14? • Autoionization of water H 2 O H+ + OH- In pure water, [H+] = [OH-] = 1 x 10 -7 M so [H+] x [OH-] = 1 x 10 -14 • Another important relationship: p. H + p. OH = 14 13
![p. H, p. OH, [H+], and [OH-] [H+] p. H p. OH [OH-] 1 p. H, p. OH, [H+], and [OH-] [H+] p. H p. OH [OH-] 1](http://slidetodoc.com/presentation_image/414c16a21a36a6958ffee5a3859b5760/image-14.jpg)
p. H, p. OH, [H+], and [OH-] [H+] p. H p. OH [OH-] 1 x 10 -5 M 5 9 1 x 10 -9 M 9 5 1 x 10 -5 M 1 x 10 -11 M 11 3 1 x 10 -3 M 3. 98 x 10 -3 M 2. 4 11. 6 2. 51 x 10 -12 M 14
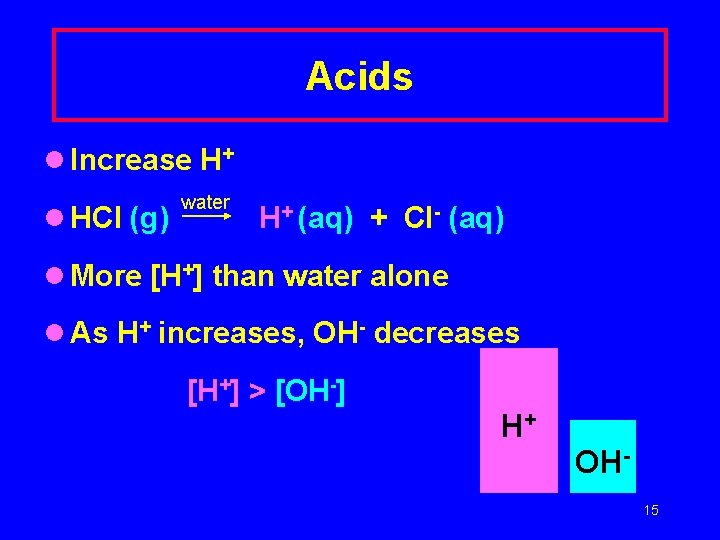
Acids l Increase H+ l HCl (g) water H+ (aq) + Cl- (aq) l More [H+] than water alone l As H+ increases, OH- decreases [H+] > [OH-] H+ OH 15
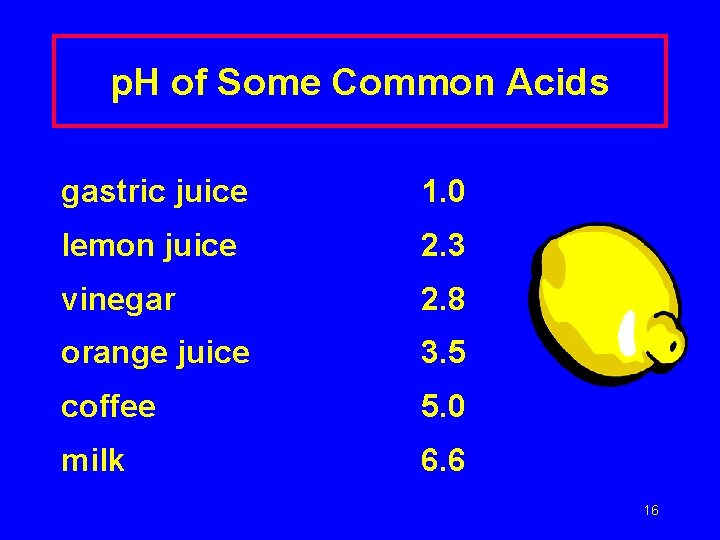
p. H of Some Common Acids gastric juice 1. 0 lemon juice 2. 3 vinegar 2. 8 orange juice 3. 5 coffee 5. 0 milk 6. 6 16

Acids Strong Acids (the big 7): HCl, HBr, HI, HNO 3, H 2 SO 4, HCl. O 3 Example: HCl(aq) H+ (aq) + Cl- (aq) **Strong acids completely ionize 17

Acids Weak acids: HC 2 H 3 O 2, HF, (and all others) Example: HF(aq) H+ (aq) + F-(aq) ** Weak acids ionize only slightly 18
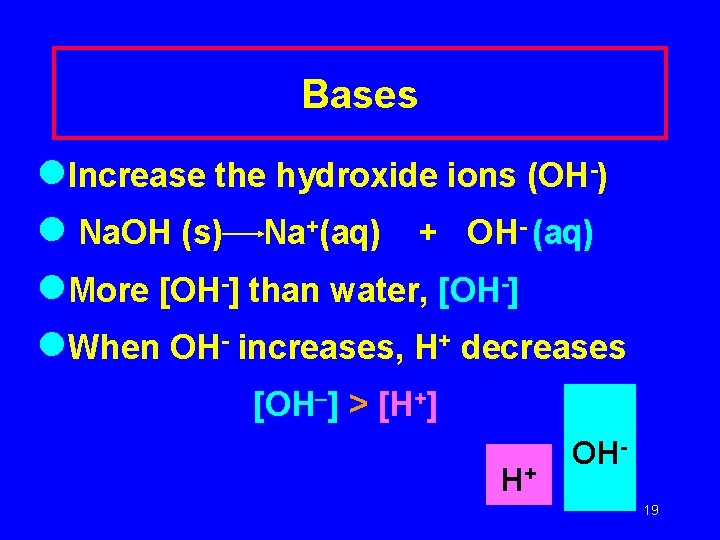
Bases l. Increase the hydroxide ions (OH-) l Na. OH (s) Na+(aq) + OH- (aq) l. More [OH-] than water, [OH-] l. When OH- increases, H+ decreases [OH ] > [H+] H+ OH 19

p. H of Some Common Bases blood 7. 4 tears 7. 4 seawater 8. 4 milk of magnesia 10. 6 household ammonia 11. 0 20

Bases • Strong Bases (the big 8) • Group I metal hydroxides • Heavier group II metal hydroxides Ca(OH)2, Ba(OH)2, Sr(OH)2 Example: Na. OH(aq) Na+ (aq) + OH- (aq) **Strong bases completely ionize 21

Bases • Weak Bases NH 3 (ammonia) Example: NH 3(aq) +H 2 O NH 4+ (aq) + OH-(aq) ** Weak bases ionize only slightly 22

Neutralization Reactions When STRONG acids and STRONG bases are mixed in the right amounts, the resulting solution is neutral. Na. OH (aq) base + HCl(aq) acid Ca(OH)2 (aq) + 2 HCl(aq) base acid Na. Cl(aq) salt + H 2 O(l) water Ca. Cl 2(aq) + 2 H 2 O(l) salt water **An acid/base reaction is a special form of a double replacement reaction 23

Neutralization The cation from the base and the anion from the acid combine to form a salt. Simultaneously, H+ and OH- combine to produce water. Na. OH(aq) H+ + HCl(aq) + from acid Na+ + from base OH- Na. Cl(aq) from base Clfrom acid + H 2 O(l) H 2 O (or HOH) water Na. Cl salt 24

Learning Check AB 1 Describe the solution in each of the following as: 1) acid 2) base or 3)neutral. A. ___soda B. ___soap C. ___coffee D. ___ wine E. ___ water F. ___ grapefruit 25

Solution AB 1 Describe each solution as: 1) acid 2) base or 3) neutral. A. _1_ soda B. _2_ soap C. _1_ coffee D. _1_ wine E. _3_ water F. _1_ grapefruit 26

Learning Check AB 2 Identify each as characteristic of an A) acid or B) base ____ 1. Sour taste ____ 2. ____ 3. ____ 4. ____ 5. Produces OH- in aqueous solutions Chalky taste Is an electrolyte Produces H+ in aqueous solutions 27

Solution AB 2 Identify each as a characteristic of an A) acid or B) base _A_ 1. Sour taste _B_ 2. Produces OH- in aqueous solutions _B_ 3. Chalky taste A, B 4. Is an electrolyte _A_ 5. Produces H+ in aqueous solutions 28

Learning Check AB 3 Acid, Base or Salt Name Ca. Cl 2 ____________ KOH ____________ Ba(OH)2 ____________ HBr ____________ H 2 SO 4 ____________ 29

Solution AB 3 Acid, Base or Salt Name Ca. Cl 2 salt calcium chloride KOH base potassiuim hydroxide Ba(OH)2 base barium hydroxide HBr acid hydrobromic acid H 2 SO 4 acid sulfuric acid 30
- Slides: 30