INTRODUCTION TO ACID BASE BALANCE 1 Dr Sadia

INTRODUCTION TO ACID BASE BALANCE 1 Dr. Sadia Haroon Lecture # 2

OBJECTIVES Explain how the p. H of the blood is stabilized by bicarb buffer and define the terms acidosis and alkalosis. Explain how the acid-base balance of the blood is affected by C 02 and HC 03 -, and describe the roles of the lungs and kidneys in maintaining acid-base balance. Explain how C 02 affects blood p. H, and hypoventilation and hyperventilation affect acidbase balance. Explain how the interaction between plasma K+ and H+ concentrations affects the tubular secretion of these.

ACID BASE HOMEOSTASIS Acid-Base homeostasis involves chemical and physiologic processes responsible for the maintenance of the acidity of body fluids at levels that allow optimal function of the whole individual 3

ACID BASE HOMEOSTASIS The chemical processes represent the first line of defense to an acid or base load and include the extracellular and intracellular buffers The physiologic processes modulate acid-base composition by changes in cellular metabolism and by adaptive responses in the excretion of volatile acids by the lungs and fixed acids by the kidneys 4
ACIDS Acids can be defined as a proton (H+) donor Hydrogen containing substances which dissociate in solution to release H+ Click Here 5
ACIDS Physiologically important acids include: �Carbonic acid (H 2 CO 3) �Phosphoric acid (H 3 PO 4) �Pyruvic acid (C 3 H 4 O 3) �Lactic acid (C 3 H 6 O 3) These acids are dissolved in body fluids Lactic acid Phosphoric acid Pyruvic acid 6

TYPES OF ACIDS IN THE BODY Volatile acids: � Can leave solution and enter the atmosphere. � H 2 C 03 (carbonic acid). � Pco 2 is most important factor in p. H of body tissues.

TYPES OF ACIDS IN THE BODY Fixed Acids: � Acids that do not leave solution. � Sulfuric and phosphoric acid. � Catabolism of amino acids, nucleic acids, and phospholipids.

TYPES OF ACIDS IN THE BODY Organic Acids: � Byproducts of aerobic metabolism, during anaerobic metabolism and during starvation, diabetes. � Lactic acid, ketones.
BASES Bases can be defined as: �A proton (H+) acceptor �Molecules capable of accepting a hydrogen ion (OH-) Click Here 10
BASES Physiologically important bases include: �Bicarbonate (HCO 3 - ) �Biphosphate (HPO 4 -2 ) Biphosphate 11
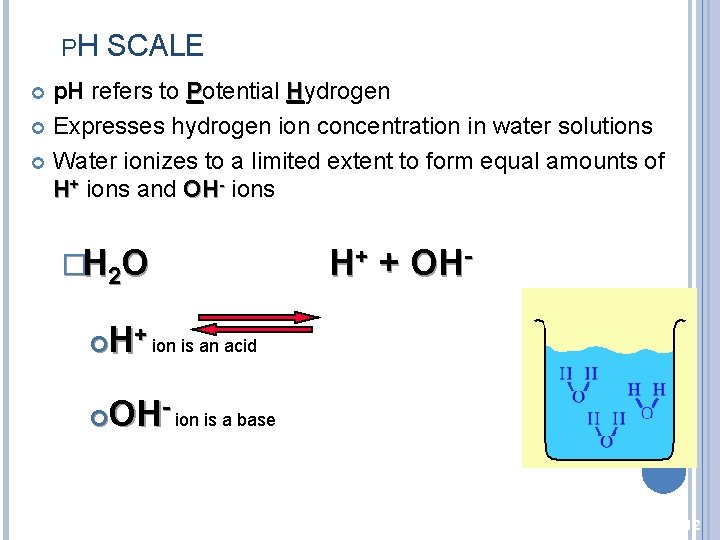
PH SCALE p. H refers to Potential Hydrogen Expresses hydrogen ion concentration in water solutions Water ionizes to a limited extent to form equal amounts of H+ ions and OH- ions �H 2 O H+ + OH- H+ ion is an acid OH- ion is a base 12
ACID-BASE BIOCHEMISTRY METHODS p. H electrode

PH SCALE p. H equals the logarithm (log) to the base 10 of the reciprocal of the hydrogen ion (H+) concentration p. H = log 1 / H+ concentration in extracellular fluid (ECF) 4 X 10 -8 (0. 00000004) 14
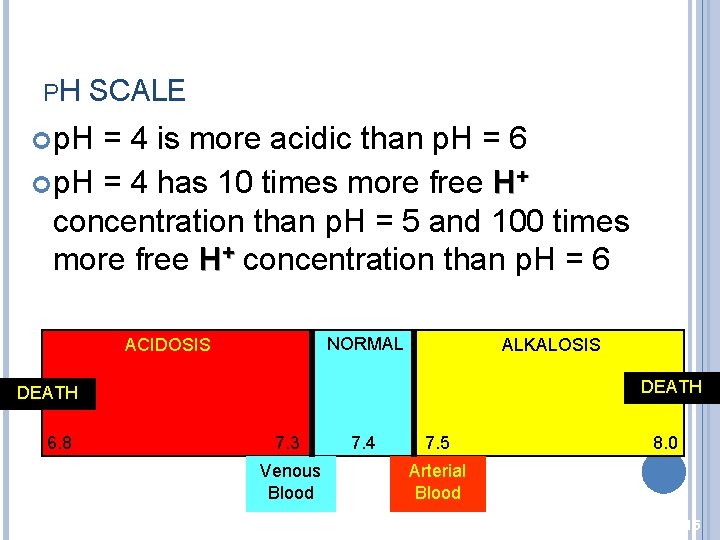
PH SCALE p. H = 4 is more acidic than p. H = 6 p. H = 4 has 10 times more free H+ concentration than p. H = 5 and 100 times more free H+ concentration than p. H = 6 NORMAL ACIDOSIS ALKALOSIS DEATH 6. 8 7. 3 Venous Blood 7. 4 7. 5 8. 0 Arterial Blood 15
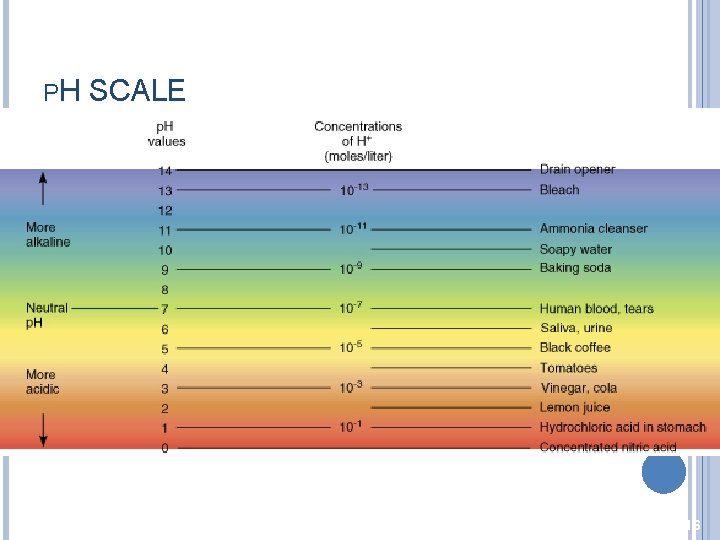
PH SCALE 16

ACIDOSIS / ALKALOSIS 17

ACIDOSIS / ALKALOSIS Acidosis �A condition in which the blood has too much acid (or too little base), frequently resulting in a decrease in blood p. H Alkalosis �A condition in which the blood has too much base (or too little acid), occasionally resulting in an increase in blood p. H 18
CHANGES IN CELL EXCITABILITY p. H decrease (more acidic) depresses the central nervous system �Can lead to loss of consciousness p. H increase (more basic) can cause over-excitability �Tingling sensations, nervousness, muscle twitches 19
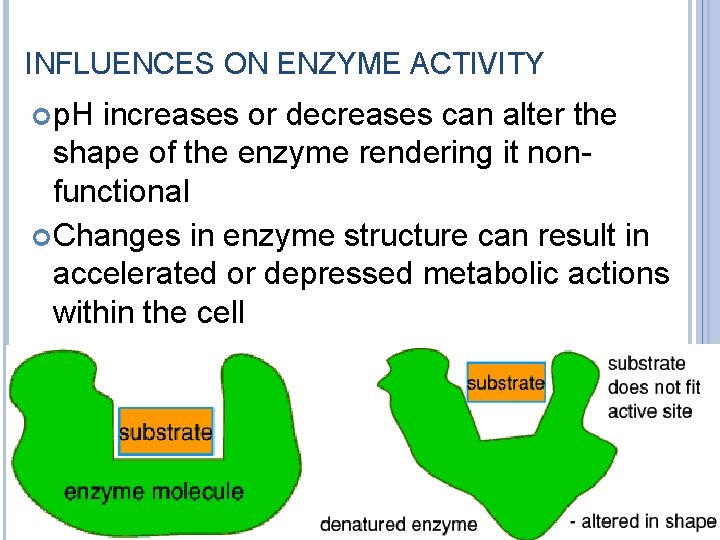
INFLUENCES ON ENZYME ACTIVITY p. H increases or decreases can alter the shape of the enzyme rendering it nonfunctional Changes in enzyme structure can result in accelerated or depressed metabolic actions within the cell 20
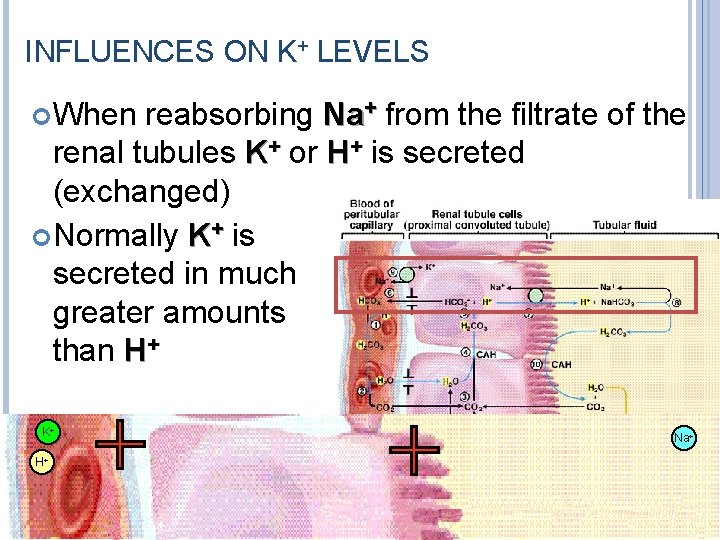
INFLUENCES ON K+ LEVELS When reabsorbing Na+ from the filtrate of the renal tubules K+ or H+ is secreted (exchanged) Normally K+ is secreted in much greater amounts than H+ K+ Na+ H+ K+ 21

ACID-BASE REGULATION 23

BUFFER SYSTEMS Provide or remove H+ and stabilize the p. H. Include weak acids that can donate H+ and weak bases that can absorb H+. Does NOT prevent a p. H change.
![Henderson-Hasselbach Equation 1) Ka = [H+][A-] [HA] HA = weak acid A- = Conjugate Henderson-Hasselbach Equation 1) Ka = [H+][A-] [HA] HA = weak acid A- = Conjugate](http://slidetodoc.com/presentation_image_h/323a10962a385c468166378b7bda0743/image-25.jpg)
Henderson-Hasselbach Equation 1) Ka = [H+][A-] [HA] HA = weak acid A- = Conjugate base 2) [H+] = Ka [HA] [A-] 3) -log[H+] = -log Ka -log [HA] [A-] 4) -log[H+] = -log Ka +log [A-] [HA] 5) p. H = p. Ka +log [A-] [HA] * H-H equation describes the relationship between p. H, p. Ka and buffer concentration
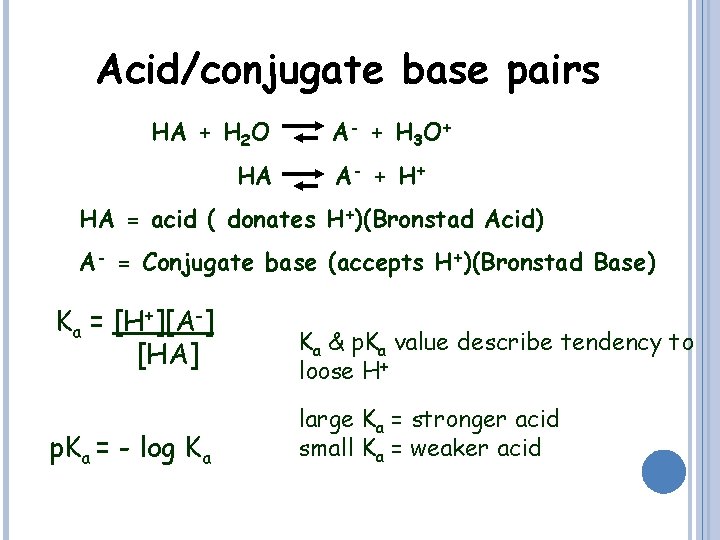
Acid/conjugate base pairs HA + H 2 O HA A - + H 3 O + A - + H+ HA = acid ( donates H+)(Bronstad Acid) A- = Conjugate base (accepts H+)(Bronstad Base) Ka = [H+][A-] [HA] p. Ka = - log Ka Ka & p. Ka value describe tendency to loose H+ large Ka = stronger acid small Ka = weaker acid

HENDERSON-HASSELBACH EQUATION Consider the dissociation of a general acid HA HA H + + A- We can define a dissociation constant (K) where Rearranging gives Taking logarithms on both sides and multiplying by -1 gives: -log[H+] = -log. K – log [HA]/[A-] or p. H = p. K + log [A-]/[HA]
![Henderson-Hasselbach Equation 1) Ka = [H+][A-] [HA] HA = weak acid A- = Conjugate Henderson-Hasselbach Equation 1) Ka = [H+][A-] [HA] HA = weak acid A- = Conjugate](http://slidetodoc.com/presentation_image_h/323a10962a385c468166378b7bda0743/image-29.jpg)
Henderson-Hasselbach Equation 1) Ka = [H+][A-] [HA] HA = weak acid A- = Conjugate base 2) [H+] = Ka [HA] [A-] 3) -log[H+] = -log Ka -log [HA] [A-] 4) -log[H+] = -log Ka +log [A-] [HA] 5) p. H = p. Ka +log [A-] [HA] * H-H equation describes the relationship between p. H, p. Ka and buffer concentration

Henderson-Hasselbalch Equation • This equation can be used to determine the p. H if the p. K and ratio of the ionised and unionised forms is known. • The p. Ka (a for acid) is the –ve log of the dissociation constant of the acid. It is the p. H at which the ratio of the ionised and unionised species is equal to 1. ie the molar concentration of the ionised and unionsed species is the same. • Similarly p. Kb is –ve log of the dissociation constant of the base

REGULATION OF H+ CONCENTRATION Concentration of hydrogen ions is regulated sequentially by: Chemical buffer systems –act within seconds The respiratory center in the brain stem –acts within 1 -3 min Renal mechanisms –require hours to days to effect p. H changes Sources of hydrogen ions anaerobic and aerobic respiration of glucose incomplete oxidation of fatty acids oxidation of sulfur-containing amino acids hydrolysis of phosphoproteins and nucleic acids

Buffers • Definition: A weak acid plus its conjugate base that cause a solution to resist changes in p. H when an acid or base are added Effectiveness of a buffer is determined by: 1) the p. H of the solution, buffers work best within 1 p. H unit of their p. Ka 2) the concentration of the buffer; the more present, the greater the buffering capacity
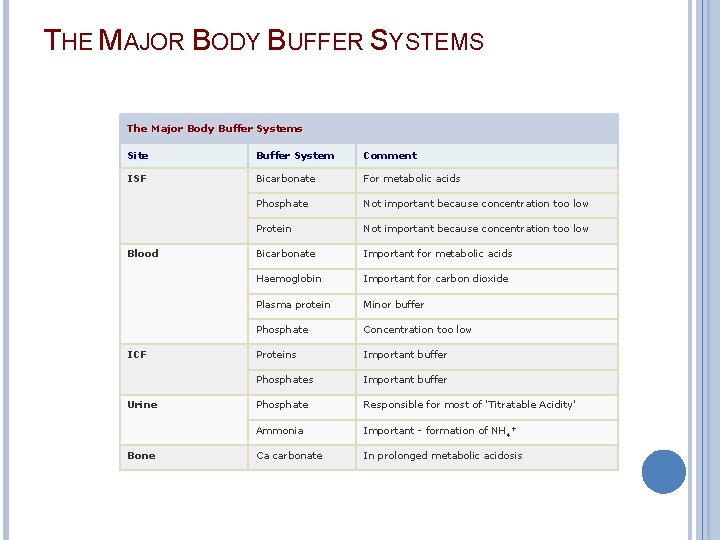
THE MAJOR BODY BUFFER SYSTEMS The Major Body Buffer Systems Site Buffer System Comment ISF Bicarbonate For metabolic acids Phosphate Not important because concentration too low Protein Not important because concentration too low Bicarbonate Important for metabolic acids Haemoglobin Important for carbon dioxide Plasma protein Minor buffer Phosphate Concentration too low Proteins Important buffer Phosphate Responsible for most of 'Titratable Acidity' Ammonia Important - formation of NH 4+ Ca carbonate In prolonged metabolic acidosis Blood ICF Urine Bone
34

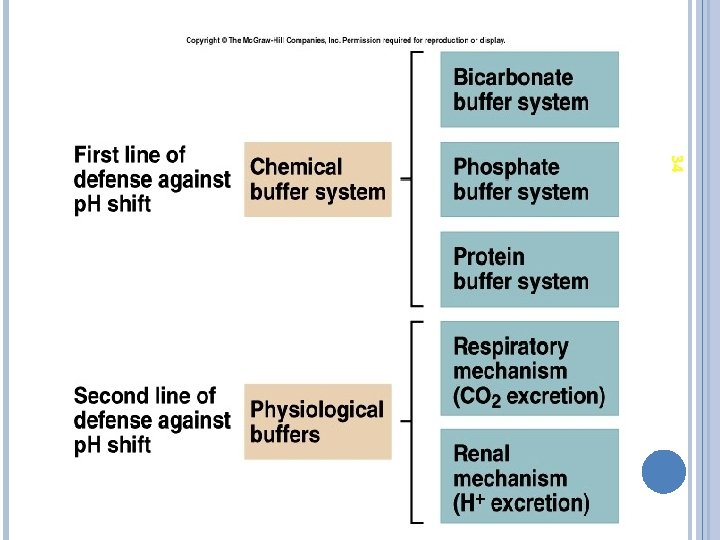
ACID-BASE REGULATION Maintenance of an acceptable p. H range in the extracellular fluids is accomplished by three mechanisms: � 1) Chemical Buffers React very rapidly (less than a second) � 2) Respiratory Regulation Reacts rapidly (seconds to minutes) � 3) Renal Regulation Reacts slowly (minutes to hours) 35
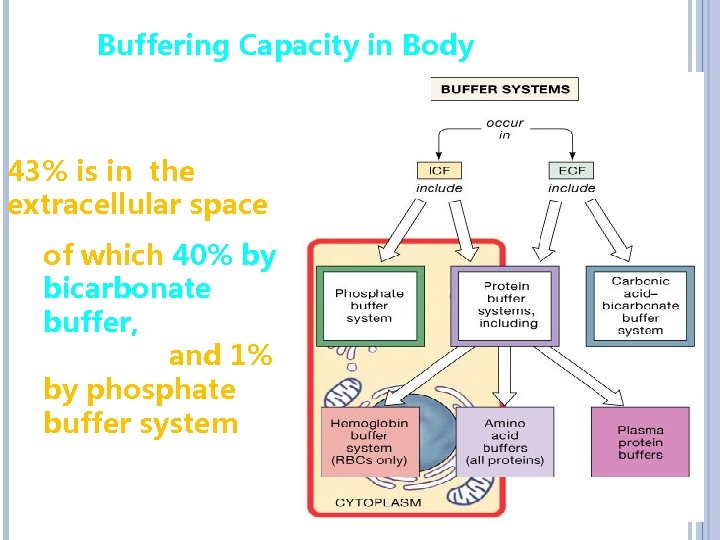
Buffering Capacity in Body 52% is in cells, 5% is in RBCs 43% is in the extracellular space of which 40% by bicarbonate buffer, 1% by proteins and 1% by phosphate buffer system 36

ORGANS INVOLVED IN THE REGULATION OF A-B-BALANCE � CO 2 production from complete oxidation of substrates � metabolism of organic acid anions � conversion of NH 4+ to urea in the liver results in an equivalent production of H+ Production of plasma proteins � such as lactate, ketones and amino acids metabolism of ammonium � 20% of the body’s daily production esp. albumin contributing to the anion gap Bone inorganic matrix consists of hydroxyapatite crystals (Ca 10(PO 4)6(OH)2] bone can take up H+ in exchange for Ca 2+, Na+ and K+ (ionic exchange) or release of HCO 3 -, CO 3 - or HPO 42 -

BUFFER CAPACITY The buffer capacity of a system is already defined as the amount of strong acid or base added to one litre (l) of the system in order to change the p. H one unit

CONTROL OF ACIDS Buffer systems Take up H+ or release H+ as conditions change Buffer pairs – weak acid and a base Exchange a strong acid or base for a weak one Results in a much smaller p. H change

CONTROL OF ACIDS Buffer systems Take up H+ or release H+ as conditions change Buffer pairs – weak acid and a base Exchange a strong acid or base for a weak one Results in a much smaller p. H change

CHEMICAL BUFFERS Act within fraction of a second. Protein. HCO 3 -. Phosphate.

ACID-BASE REGULATION Chemical Buffers � The body uses p. H buffers in the blood to guard against sudden changes in acidity � A p. H buffer works chemically to minimize changes in the p. H of a solution Buffer
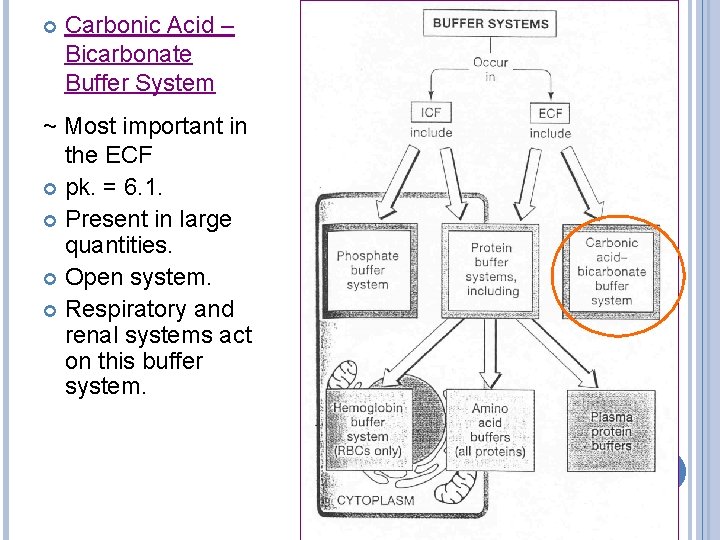
Carbonic Acid – Bicarbonate Buffer System ~ Most important in the ECF pk. = 6. 1. Present in large quantities. Open system. Respiratory and renal systems act on this buffer system.

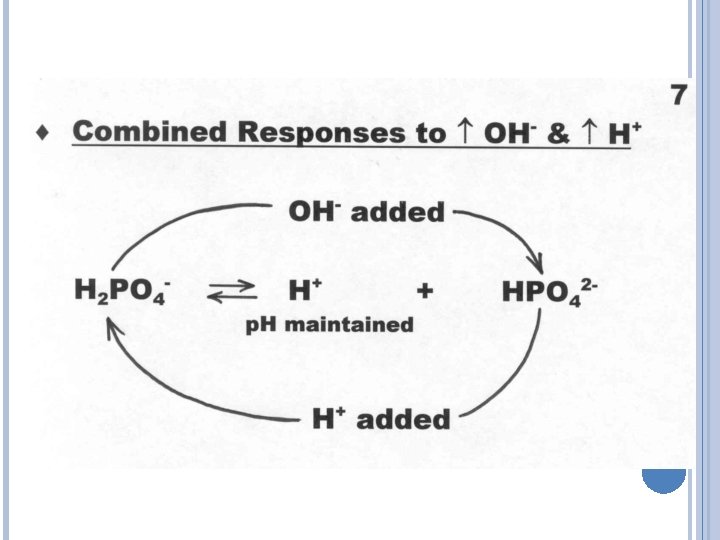
BICARBONATE BUFFER SYSTEM Present in intra-and extracellular fluid � Bicarbonate ion acts as weak base, carbonic acid acts as a weak acid � Bicarbonate ions combine with excess hydrogen ions to form carbonic acid � Carbonic acid dissociates to release bicarbonate ions and hydrogen ions H+ + HCO 3 - H 2 CO 3 H+ + HCO 3 -
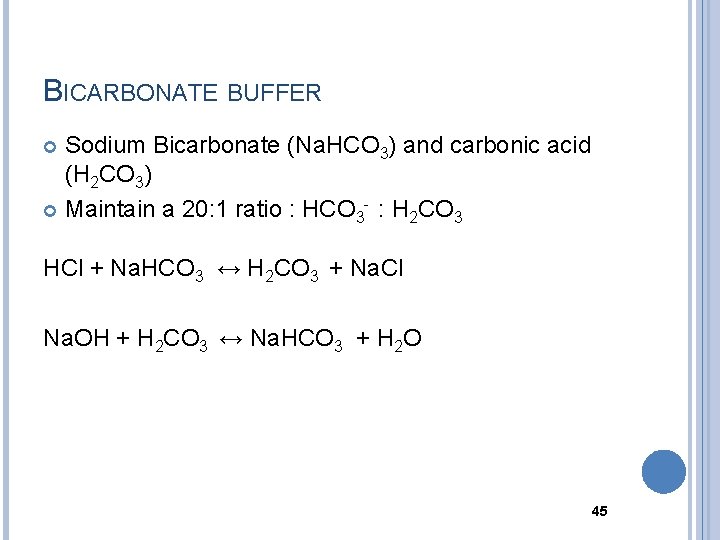
BICARBONATE BUFFER Sodium Bicarbonate (Na. HCO 3) and carbonic acid (H 2 CO 3) Maintain a 20: 1 ratio : HCO 3 - : H 2 CO 3 HCl + Na. HCO 3 ↔ H 2 CO 3 + Na. Cl Na. OH + H 2 CO 3 ↔ Na. HCO 3 + H 2 O 45

HCO 3 - LIMITATIONS Cannot protect ECF from respiratory problems. Cannot protect ECF from elevated or decreased CO 2. Limited by availability of HCO 3 -.

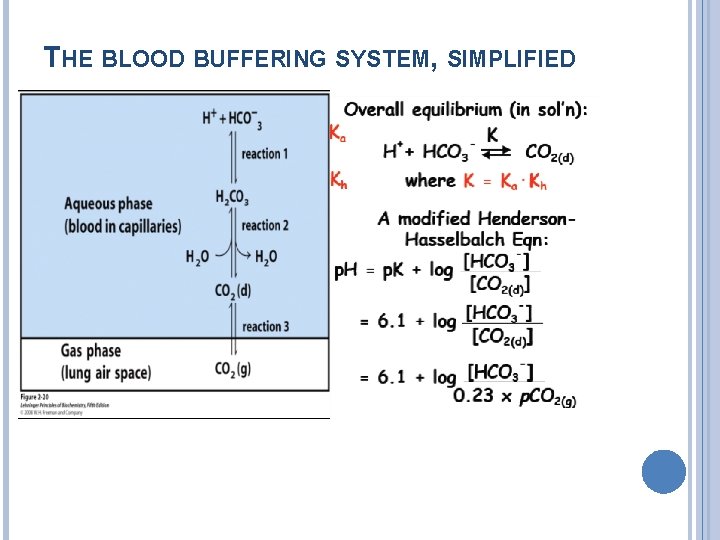
THE BLOOD BUFFERING SYSTEM, SIMPLIFIED

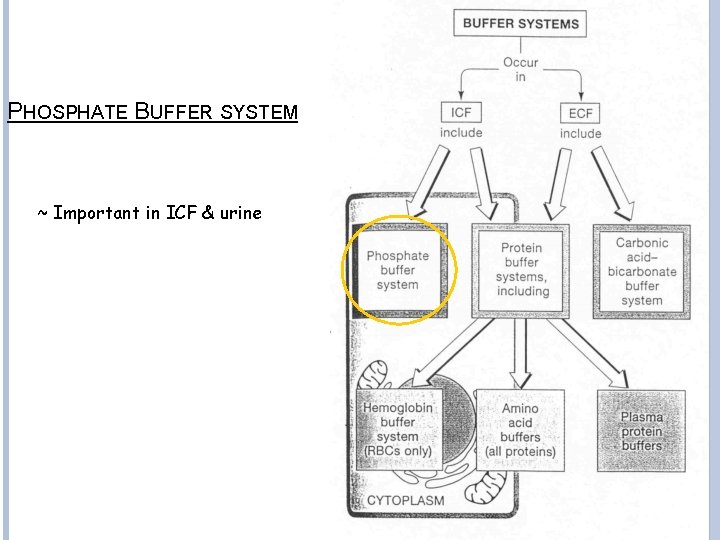
PHOSPHATE BUFFER SYSTEM ~ Important in ICF & urine

PHOSPHATE BUFFER SYSTEM Important in intracellular fluid and urine p. H regulation Consists of two phosphate ions � Monohydrogenphosphate ions act as a weak base and combine with hydrogen ions to form dihydrogenphosphate � Dihydrogenphosphate dissociates to release hydrogen ions H+ + HPO 4 -2 H 2 PO 4 - H+ + HPO 4 -2

PHOSPHATE & INTRACELLULAR BUFFERS Both Intra and Extra cellular phosphate act as a buffer. But its role is minor compared to HB or HCO 3. Intracellular buffers are needed because H doesn’t cross Plasma Membrane. Intracellular PH is more acidic. (7. 2)
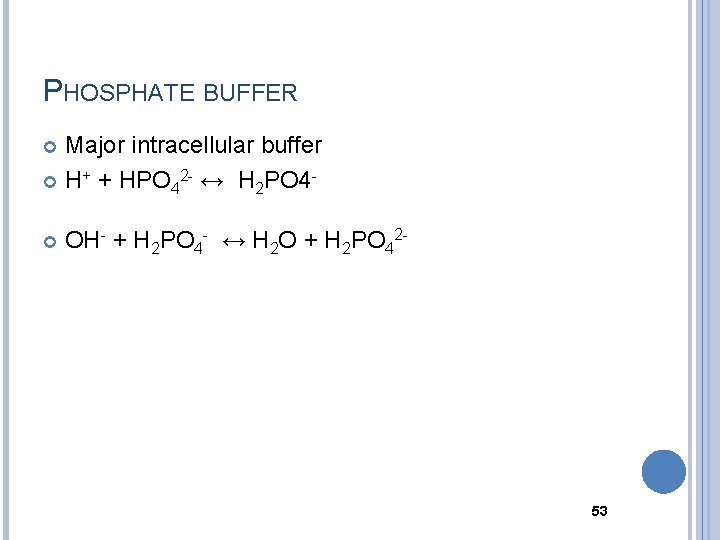
PHOSPHATE BUFFER Major intracellular buffer H+ + HPO 42 - ↔ H 2 PO 4 OH- + H 2 PO 4 - ↔ H 2 O + H 2 PO 42 - 53

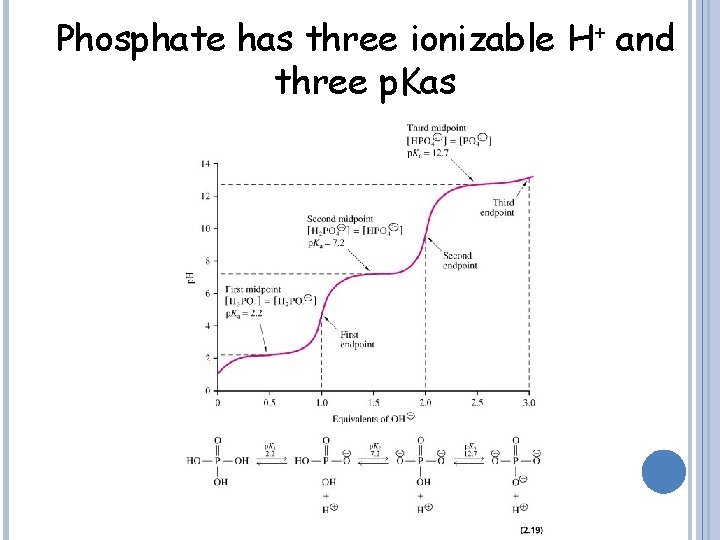
Phosphate has three ionizable H+ and three p. Kas
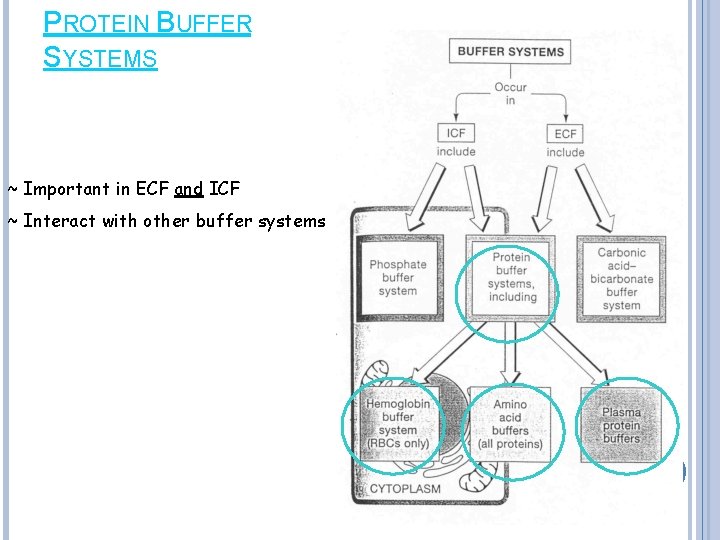
PROTEIN BUFFER SYSTEMS ~ Important in ECF and ICF ~ Interact with other buffer systems

PROTEIN BUFFER SYSTEM Consists of Plasma Proteins (albumin, hemoglobin) Remember proteins are just chains of AAThe exposed amine group of the AA (NH 2) accepts H+ ions when conditions are acidic The exposed carboxyl group of AA can release H+ ions when conditions are basic Proteins can act as Acids or Bases

PROTEIN BUFFERS Includes hemoglobin, work in blood and ISF Carboxyl group gives up H+ Amino Group accepts H+ Side chains that can buffer H+ are present on 27 amino acids. 59

HEMOGLOBIN IS AN IMPORTANT BLOOD BUFFER PARTICULARLY FOR BUFFERING CO 2 Protein buffers in blood include haemoglobin (150 g/l) and plasma proteins (70 g/l). Buffering is by the imidazole group of the histidine residues which has a p. Ka of about 6. 8. This is suitable for effective buffering at physiological p. H. Haemoglobin is quantitatively about 6 times more important then the plasma proteins as it is present in about twice the concentration and contains about three times the number of histidine residues per molecule. For example if blood p. H changed from 7. 5 to 6. 5, haemoglobin would buffer 27. 5 mmol/l of H+ and total plasma protein buffering would account for only 4. 2 mmol/l of H+.
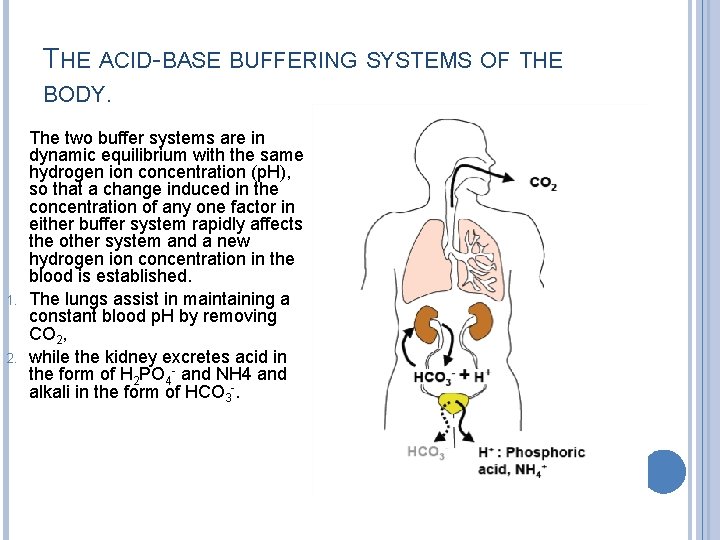
THE ACID-BASE BUFFERING SYSTEMS OF THE BODY. 1. 2. The two buffer systems are in dynamic equilibrium with the same hydrogen ion concentration (p. H), so that a change induced in the concentration of any one factor in either buffer system rapidly affects the other system and a new hydrogen ion concentration in the blood is established. The lungs assist in maintaining a constant blood p. H by removing CO 2, while the kidney excretes acid in the form of H 2 PO 4 - and NH 4 and alkali in the form of HCO 3 -.

CHEMICAL BUFFERS Act within fraction of a second. Protein. HCO 3 -. Phosphate.

ACID-BASE REGULATION Respiratory Regulation � Carbon dioxide is an important by-product of metabolism and is constantly produced by cells � The blood carries carbon dioxide to the lungs where it is exhaled CO Cell 2 CO CO 2 2 Metabolism 63

RESPIRATORY BUFFER SYSTEMS The respiratory system regulation of acidbase balance is a physiological buffering system There is a reversible equilibrium between: � Dissolved carbon dioxide and water � Carbonic acid and the hydrogen and bicarbonate ions CO 2+ H 2 O ↔H 2 CO 3↔H++ HCO 3¯
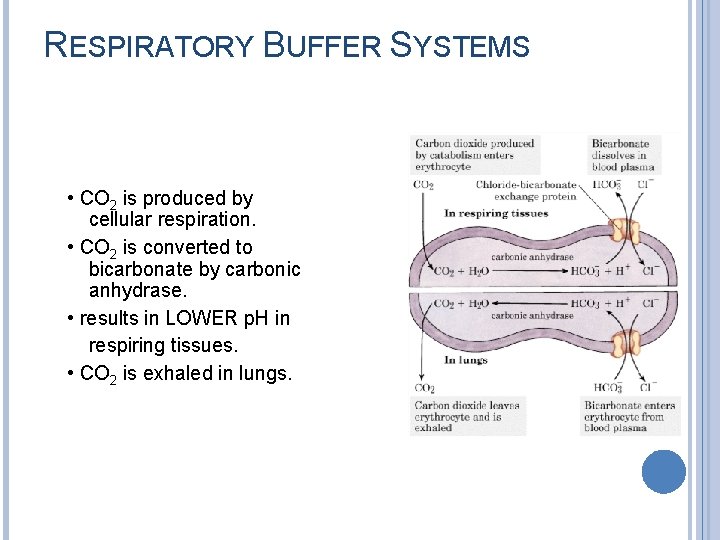
RESPIRATORY BUFFER SYSTEMS • CO 2 is produced by cellular respiration. • CO 2 is converted to bicarbonate by carbonic anhydrase. • results in LOWER p. H in respiring tissues. • CO 2 is exhaled in lungs.
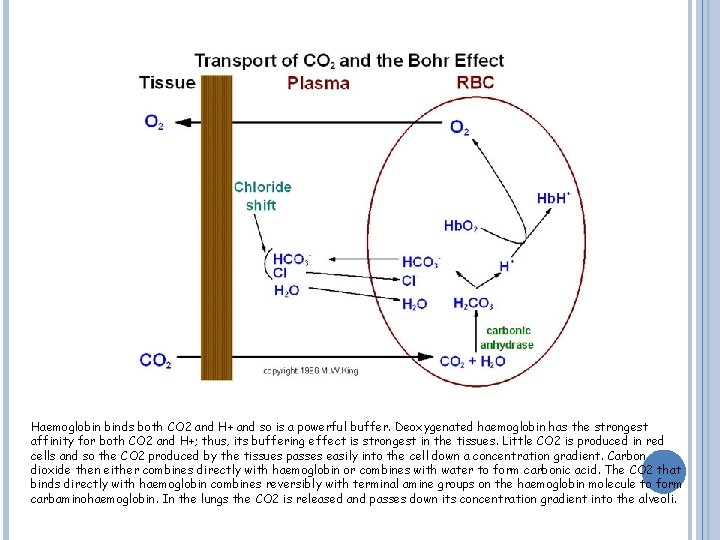
Haemoglobin binds both CO 2 and H+ and so is a powerful buffer. Deoxygenated haemoglobin has the strongest affinity for both CO 2 and H+; thus, its buffering effect is strongest in the tissues. Little CO 2 is produced in red cells and so the CO 2 produced by the tissues passes easily into the cell down a concentration gradient. Carbon dioxide then either combines directly with haemoglobin or combines with water to form carbonic acid. The CO 2 that binds directly with haemoglobin combines reversibly with terminal amine groups on the haemoglobin molecule to form carbaminohaemoglobin. In the lungs the CO 2 is released and passes down its concentration gradient into the alveoli.

RESPIRATORY SYSTEM 2 nd line of defense. Acts within min. maximal in 12 -24 hrs. H 2 CO 3 produced converted to CO 2, and excreted by the lungs. Alveolar ventilation also increases as p. H decreases (rate and depth). Coarse , CANNOT eliminate fixed acid.

ACID-BASE REGULATION Kidney Regulation � Excess acid is excreted by the kidneys, largely in the form of ammonia � The kidneys have some ability to alter the amount of acid or base that is excreted, but this generally takes several days 68
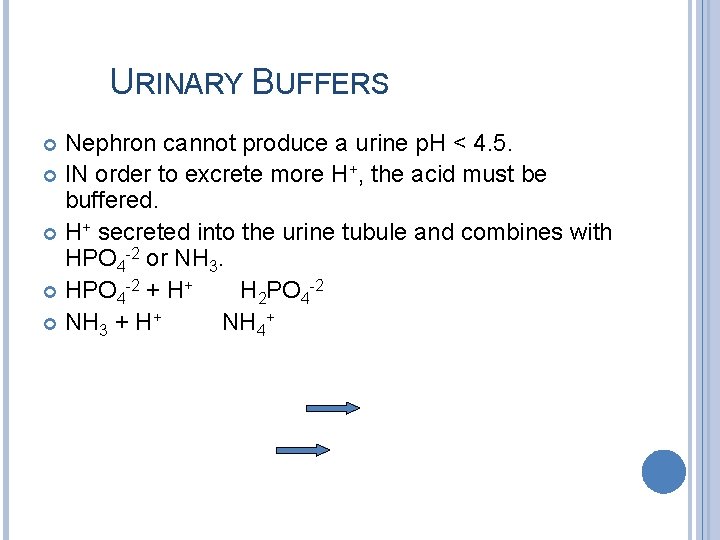
URINARY BUFFERS Nephron cannot produce a urine p. H < 4. 5. IN order to excrete more H+, the acid must be buffered. H+ secreted into the urine tubule and combines with HPO 4 -2 or NH 3. HPO 4 -2 + H+ H 2 PO 4 -2 NH 3 + H+ NH 4+
- Slides: 69