INTRODUCTION REGULATION BLOOD BUFFERS RESPIRATORY RENAL ACID ABG


� INTRODUCTION � REGULATION � BLOOD BUFFERS � RESPIRATORY � RENAL � ACID � ABG OF ACID BASE BALANCE MECHANISM BASE DISORDERS ANALYSIS

� Normal blood PH : 7. 35 -7. 45 � Maintenance of blood p. H - important homeostatic mechanism of the body. � PH less than 7. 35 leads to acidosis and p. H more than 7. 45 leads to alkalosis.

� Acids are proton donors. HA ↔ H+ + AHCL ↔ H ++ CL � - Bases are proton acceptors. Acid Base NH 3+H+ ↔ NH 4+ HCO 3+H+ ↔ H 2 CO 3 • Weak and strong acids : HCL → H 2 CO 3 → H ++ CL - (COMPLETE) - Strong acid H+ + HCO 3 - ( PARTIAL) - Weak acid

� Carbonic acid - Oxidation of c-compounds � Sulphuric acid - Oxidation of sulphur containing amino acids. � Phosphoric acid-metabolism of dietary nucleoproteins, phosphatides. phosphoproteins , Organic acid- oxidation of carbohydrates , fats and proteins. e. g. pyruvic acid , lactic acid , acetoacetic acid etc. � � Iatrogenic : - certain medicine like NH 4 Cl, mandelic acid etc. NOTE: DIET RICH IN ANIMAL PROTEIN RESULTS IN MORE ACID PRODUCTION.
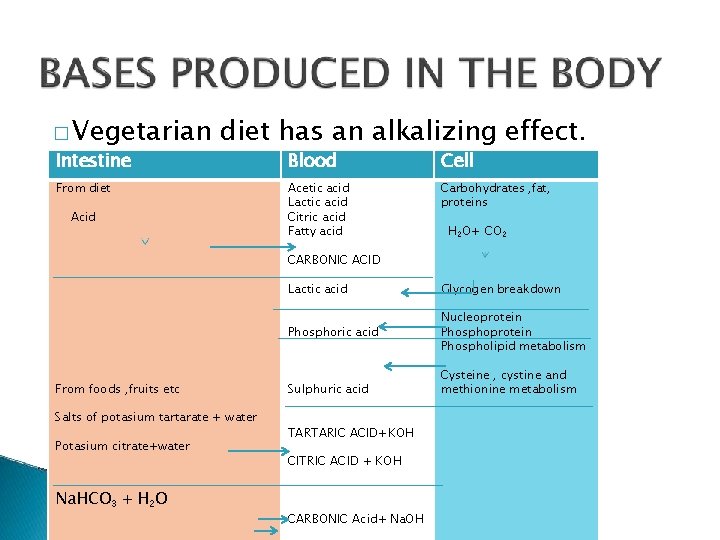
� Vegetarian Intestine diet has an alkalizing effect. From diet Acid Blood Cell Acetic acid Lactic acid Citric acid Fatty acid Carbohydrates , fat, proteins H 2 O+ CO 2 CARBONIC ACID From foods , fruits etc Salts of potasium tartarate + water Potasium citrate+water Na. HCO 3 + H 2 O Lactic acid Glycogen breakdown Phosphoric acid Nucleoprotein Phospholipid metabolism Sulphuric acid TARTARIC ACID+KOH CITRIC ACID + KOH CARBONIC Acid+ Na. OH Cysteine , cystine and methionine metabolism

� � Buffers - resist change in p. H. Two types a)Mixture of weak acids with their salt with a strong base. b)Mixture acid. of weak bases with their salt with a strong Example 1. Bicarbonate buffer (H 2 CO 3 / Na. HCO 3 ) 2. Acetate buffer (CH 3 COOH / CH 3 COONa) 3. Phosphate buffer (Na 2 HPO 4 /Na. H 2 PO 4 )

� If you go running you build up lactic acid in your muscles. � Therefore � Buffer � And your p. H will decrease. will act to increase the p. H. vice versa. So how does it work using chemistry ?
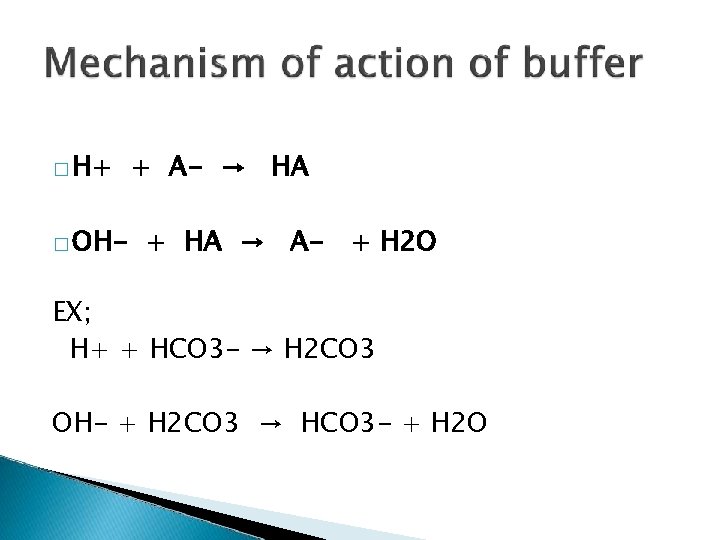
� H+ � OH- + A- → HA + HA → A- + H 2 O EX; H+ + HCO 3 - → H 2 CO 3 OH- + H 2 CO 3 → HCO 3 - + H 2 O
![� Acid add [ H+ ] therefore ↑ [ H+ ] - ↓ p. � Acid add [ H+ ] therefore ↑ [ H+ ] - ↓ p.](http://slidetodoc.com/presentation_image_h/7cb13ff064d6daff3bc261ee2d573dea/image-10.jpg)
� Acid add [ H+ ] therefore ↑ [ H+ ] - ↓ p. H � Base bind [ H+ ] therefore ↓ [ H+ ] - ↑ p. H. � Phosphate buffer H 2 PO 4 ↔ HPO 42 - + H+

� Protein � If p. H ↓ buffer Amino acid In acidic medium amino acid (NH 2) act as a base and absorbs H+. � If p. H↑ In alkaline medium amino acid (COOH) act as a acid and release H+.

� Acetate buffer �When HCl is added to the acetate buffer, the salts react with the acid forming the weak acid , acetic acid and its salts. CH 3 COONa + Hcl CH 3 COOH + Nacl When Na. OH is added, the acid reacts with its forming salt and water. CH 3 COOH + Nao. H CH 3 COONa +Nacl Thus changes in p. H is minimised.
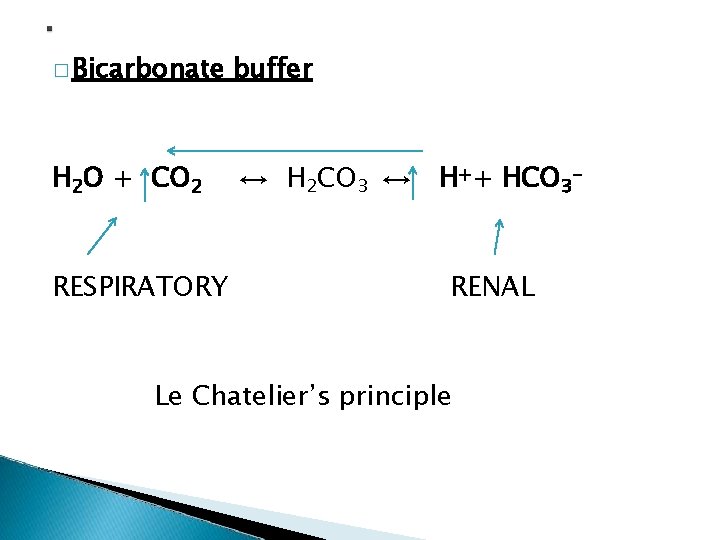
� Bicarbonate buffer H 2 O + CO 2 ↔ H 2 CO 3 ↔ H++ HCO 3 - RESPIRATORY RENAL Le Chatelier’s principle

1. Standard buffer solution are used indicator for determination of p. H. with 2. Buffer are used to check the performance of electrode used for determination of p. H. 3. Used for many chemical reactions including those catalysed by enzymes. 4. Used in the pathological laboratory to control p. H of culture media for bacteria tissues. 5. Very important in regulating the p. H of body fluids e. g. blood, interstitial fluid , lymph.

� Sodium monohydrogen phosphate (Na 2 HPO 4 ) � Sodium dihydrogen phosphate ( Na. H 2 PO 4 ) � Sodium bicarbonate ( Na. HCO 3 ) � Hydrogen � Sodium carbonate (H 2 CO 3 ) proteinate � Hydrogen proteinate

erythrocytes • • • Potassium bicarbonate Carbonic acid K 2 HPO 4 k. H 2 PO 4 Potasium proteinate Hydrogen proteinate • • • . ’ . . • Tissue cell Potassium proteinate Hydrogen proteinate Potasium bicarbonate Hydrogen bicarbonate K 2 HPO 4 k. H 2 PO 4
![� p. H = - log [H+] , dimensionless quantity. � [H+] means gm � p. H = - log [H+] , dimensionless quantity. � [H+] means gm](http://slidetodoc.com/presentation_image_h/7cb13ff064d6daff3bc261ee2d573dea/image-17.jpg)
� p. H = - log [H+] , dimensionless quantity. � [H+] means gm of hydrated H+ ion present as H 3 O+ per litre of fluid. � E. g. H 2 O contains 1/1000 0 00 gm of hydrogen ion in 1 litre , means [H+]=10 - 7. � Decrease of one p. H unit represents a ten fold increase in the H+ activity. � The p. H 7. 40 corresponds to a hydrogen ion concentration of 40 nmol/L ( European centre).

� Represents the negative logarithm of the ionization constant of a weak acid (ka). � Pk is the p. H at which an acid is half dissociated. � Acids have pk value less than 7 and bases have more than 7. �Lower pk = stronger acid �Higher pk = stronger base

1. 2. 3. 4. 5. 6. 7. 8. Enzyme activity Action potential of myelinated nerve Membrane permeability Control of respiration Heart activity Plain muscle activity Oxygen Hb dissociation curve Nerve excitability

� 3 mechanism 1. Blood buffers : first line of defence 2. Respiratory regulation : second line of defence 3. Renal regulation : third line of defence

� Can not remove H+ ions from the body. � Temporarily acts as a shock absorbant to reduce the free H+ ion. � 3 buffer system : 1. Bicarbonate buffer 2. Phosphate buffer 3. Protein buffer

� Extracellular buffer system of the body � Na. HCO 3/H 2 CO 3= � NORMAL [ SALT ] / [ACID] RATIO= 20 : 1 � Base constituent (HCO 3 ) - regulated by the kidney (Metabolic component) Acid (H 2 CO 3) respiratory regulation ( Respiratory component ).

�Neutralization of strong acid and non- volatile acid entering the ECF is achieved by bicarbonate buffer , such acid e. g. HCl , H 2 SO 4, lactic acid etc. � Strong acid react with Na. HCO 3 component. thus lactic acid is buffered as follows: � Na. HCO 3 H+ L- Na LHCO 3 H+ H 2 CO 3 (weak acid) + Na Lactate (salt) lungs(H 2 co 3)

� H 2 CO 3 � By � � H+ + HCO 3 - the law of mass action, at equilibrium Ka = [H+] [HCO 3] [H 2 CO 3 ] [H+] =Ka [H 2 CO 3 ] HCO 3 - ----(1) Ka = dissociation constant -------(2) Ka=log 1/[H+] By taking reciprocal and logarithm � Log 1/[H+] = log 1/Ka +log [HCO 3] [ H 2 CO 3 ] ----(3)
![p. H = pka + log [ HCO 3 ] -------(4) [H 2 CO p. H = pka + log [ HCO 3 ] -------(4) [H 2 CO](http://slidetodoc.com/presentation_image_h/7cb13ff064d6daff3bc261ee2d573dea/image-25.jpg)
p. H = pka + log [ HCO 3 ] -------(4) [H 2 CO 3 ] USES; 1. It determines the p. H of blood. 2. Serve as an index to understand the disturbance in acid base balance of the body.

1. High concentration 2. Alkali reserve 3. Very good physiological buffer and act as front line of defence.
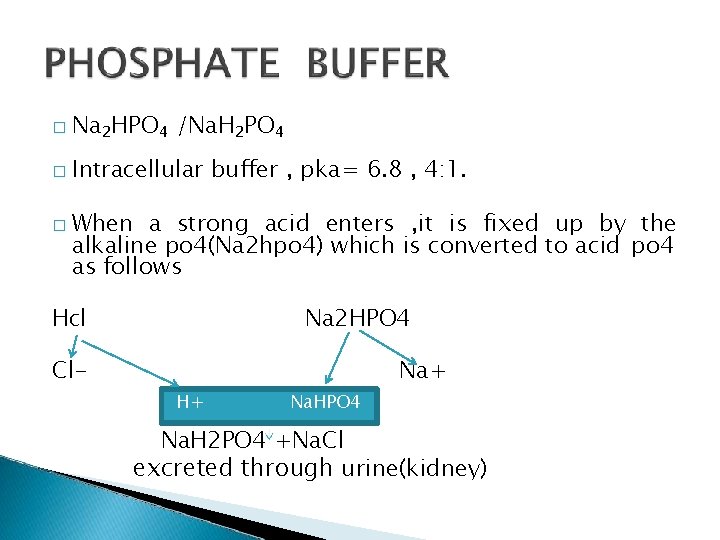
� Na 2 HPO 4 /Na. H 2 PO 4 � Intracellular � buffer , pka= 6. 8 , 4: 1. When a strong acid enters , it is fixed up by the alkaline po 4(Na 2 hpo 4) which is converted to acid po 4 as follows Na 2 HPO 4 Hcl Cl- Na+ H+ Na. HPO 4 Na. H 2 PO 4 +Na. Cl excreted through urine(kidney)
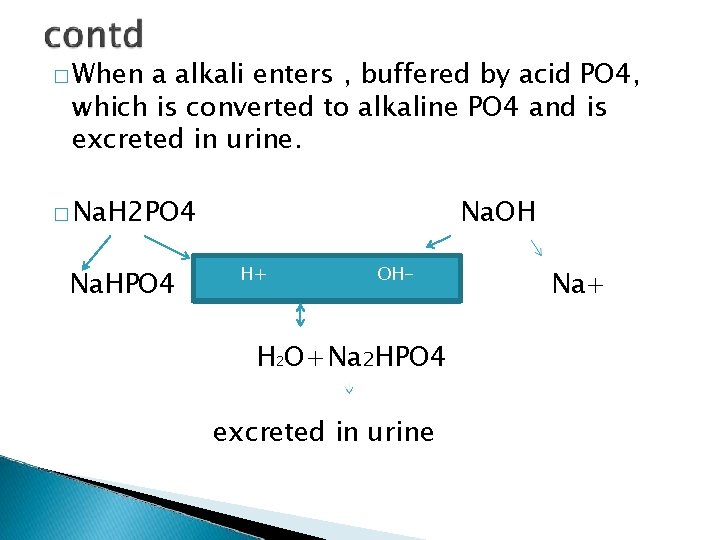
� When a alkali enters , buffered by acid PO 4, which is converted to alkaline PO 4 and is excreted in urine. � Na. H 2 PO 4 Na. HPO 4 Na. OH H+ OH- H 2 O+Na 2 HPO 4 excreted in urine Na+

� Advantage � Very effective and better, as pka approaches physiological ph.

� Plasma Protein and Hb - most important � Buffering action of protein depends on pk of ionizable group of amino acid � Effective � group- imidazole group of Histidine Pk - 6. 7

� In acidic medium, protein acts as a base, NH 2 group takes up H+ ions from the medium forming NH 3+, proteins becomes positively charged. in alkaline medium , protein act as an acid, COOH group dissociates and gives H+, forming COO-. H + combines with OH- to produce molecule of water , proteins become negatively charged

� The respiratory system helps control the acidity of the blood by regulating the elimination of CO 2 and H 2 O. � These breath. molecules are exhaled with every H 2 CO 3 H 2 O + CO 2 carbonic acid � The p. H. brain is sensitive to blood CO 2 levels and
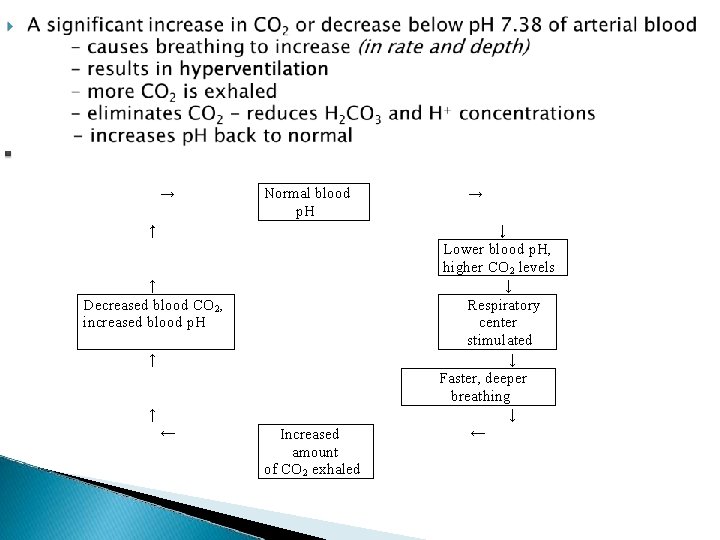
→ Normal blood p. H ↑ ↑ Decreased blood CO 2, increased blood p. H ↑ ↑ ← Increased amount of CO 2 exhaled → ↓ Lower blood p. H, higher CO 2 levels ↓ Respiratory center stimulated ↓ Faster, deeper breathing ↓ ←

A significant decrease in CO 2 or increase in p. H - causes breathing to decrease - results in hypoventilation - less CO 2 is exhaled - increases CO 2 - increases H 2 CO 3 and H+ concentrations - decreases p. H back to normal →→ Normal blood p. H ↑ ↑ Increased blood CO 2, decreased blood p. H ↑ ↑ ← Decreased amount of CO 2 exhaled →→ ↓ Higher blood p. H, lower CO 2 levels ↓ Respiratory center stimulated ↓ Slower, shallower breathing ↓ ←

• Third line of defense against change in hydrogen ion concentration • permanent solution disturbances. to the acid • Kidneys require hours to days compensate for changes in body-fluid p. H base to

1. Excretion of H+. 2. Reabsorption of bicarbonate 3. Excretion of titratable acid 4. Excretion of ammonium (NH 4+)
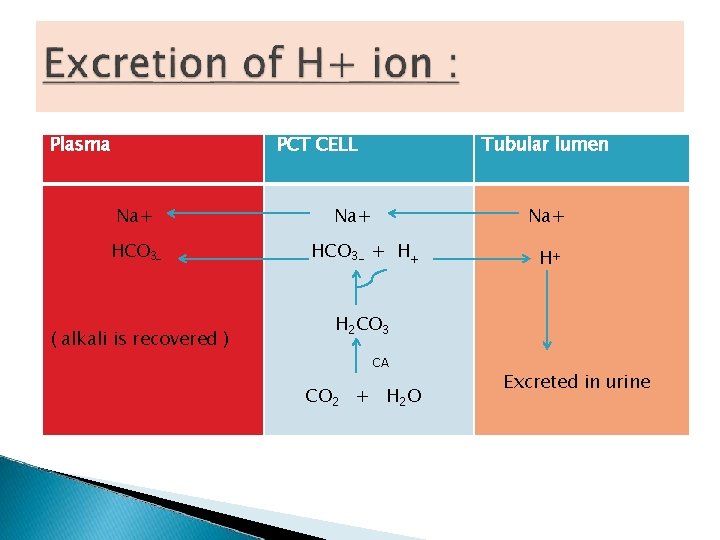
Plasma PCT CELL Na+ HCO 3 - ( alkali is recovered ) Tubular lumen Na+ HCO 3 - + H+ H+ H 2 CO 3 CA CO 2 + H 2 O Excreted in urine
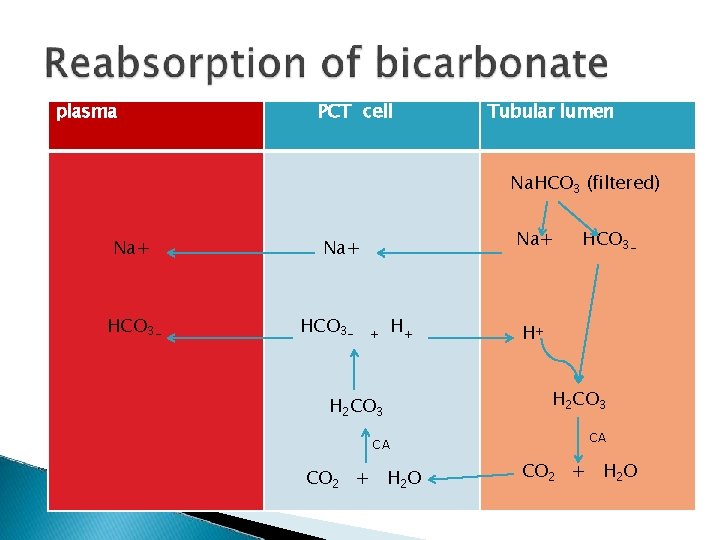
plasma PCT cell Tubular lumen Na. HCO 3 (filtered) Na+ HCO 3 - Na+ HCO 3 - + H+ H 2 CO 3 CA CO 2 + H 2 O HCO 3 - H+ H 2 CO 3 CA CO 2 + H 2 O
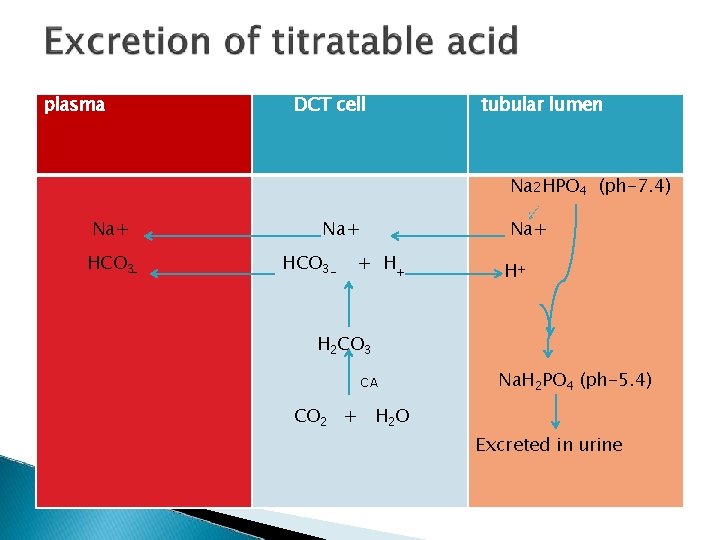
plasma DCT cell tubular lumen Na 2 HPO 4 (ph-7. 4) Na+ HCO 3 - Na+ + H+ H+ H 2 CO 3 CA Na. H 2 PO 4 (ph-5. 4) CO 2 + H 2 O Excreted in urine
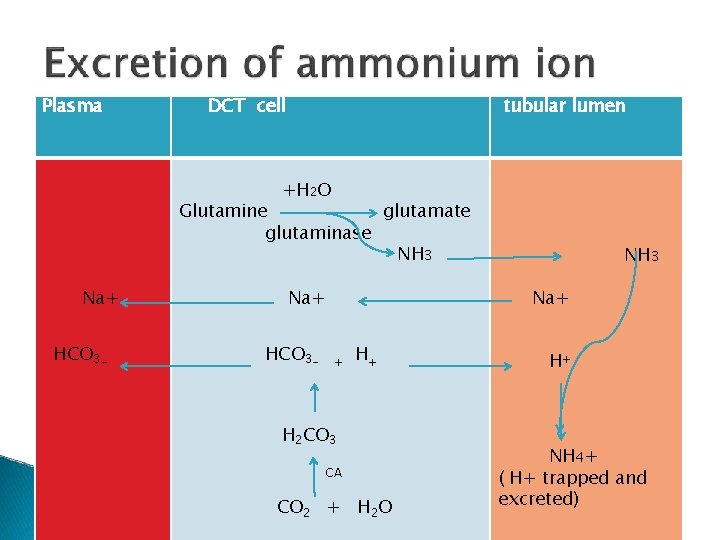
Plasma DCT cell tubular lumen +H 2 O Glutamine glutamate glutaminase NH 3 Na+ HCO 3 - Na+ HCO 3 - NH 3 Na+ + H+ H 2 CO 3 CA CO 2 + H 2 O H+ NH 4+ ( H+ trapped and excreted)

�A � � 50 year old man came to emergency department after returning from foreign travel. His symptom included persistent diarrhoea (over the past 3 days) and rapid respiration. Blood gases were drawn with following results : p. H- 7. 21 ( ) p. CO 2 - 19 mm. Hg ( ) p. O 2 - 96 mm. Hg HCO 3 - 7 mmol/l Questions: 1. What is the patient acid base status? 2. Why is the HCO 3 level is so low? 3. Why does the patient have rapid respiation?

� ACIDOSIS: PH <7. 35 a ) METABOLIC ACIDOSIS b ) RESPIRATORY ACIDOSIS ALKALOSIS : PH >7. 45 a ) METABOLIC ALKALOSIS b ) RESPIRATORY ALKALOSIS

�The sum of cations and anions in ECF is always equal , so as to maintain the electrical neutrality. �Commonly measured electrolytes in plasma are Na+, K+, Cl-, HCO 3 -. �Unmeasured anion in the plasma constitutes the anion gap.

� This is due to presence of protein anions , sulphate , phosphate and organic acids. � Anion gap = (Na + k) - ( HCO 3+ Cl- ). � Normally � Normal anion gap is about 15 m. Eq/l range = 8 -18 m. Eq/l.
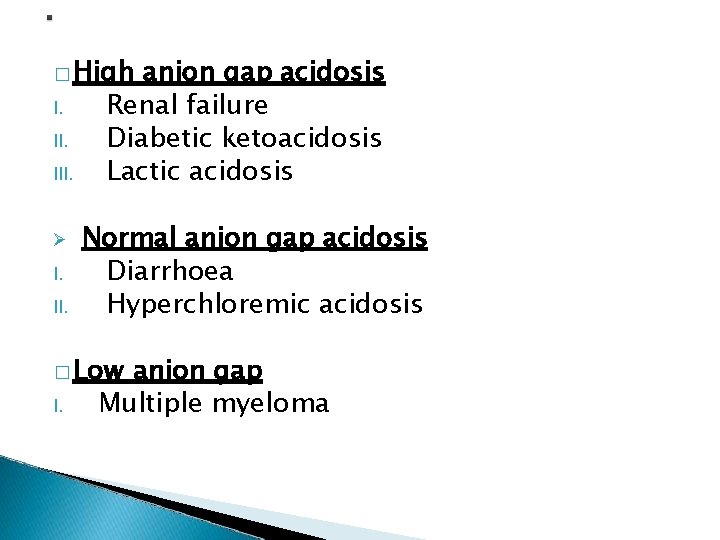
� High I. III. I. II. anion gap acidosis Renal failure Diabetic ketoacidosis Lactic acidosis Normal anion gap acidosis Diarrhoea Hyperchloremic acidosis � Low I. anion gap Multiple myeloma

� Primary deficit of bicarbonate. �Due to its utilization in buffering H+ ions, loss in urine or GIT. � Important cause-excessive production of organic acids which combine with sodium bicarbonate and deplete alkali reserve. Na. HCO 3+organic acids Na salts of o organic acids + CO 2

� Severe uncontrolled diabetes mellitus (ketoacidosis)- production of organic acids � Renal failure � Lactic acidosis � Severe � Renal diarrhoea tubular acidosis

� Increased production and accumulation of organic acid causes an elevation in anion gap. � This type is seen in ketoacidosis COMPENSATION • Hyperventilation of lungs(elimination of co 2) • Renal compensation-(3 -4 days) H+ ions excreted as NH 4+

� Primary � Causes • • • excess of carbonic acid. Severe asthma Pneumonia Cardiac arrest Depression of respiratory centre COPD
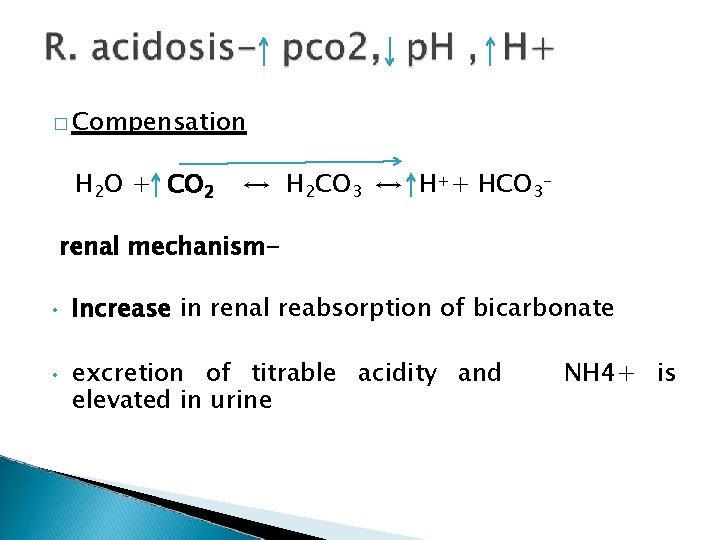
� Compensation H 2 O + CO 2 ↔ H 2 CO 3 ↔ H++ HCO 3 - renal mechanism • • Increase in renal reabsorption of bicarbonate excretion of titrable acidity and elevated in urine NH 4+ is

� Primary excess of bicarbonate Causes • Severe vomiting • Hypokalemia • Intravenous administration of bicarbonate. • Cushing syndrome

� M. alkalosis is commonly associated with hypokalemia. � In severe k+ deficiency , H+ ions are retained inside the cells to replace missing k+ ions. � In the tubular cells, H+ions are exchanged (instead of k+) with the reabsorbed Na+. � Paradoxically , the patient excretes acid urine despite alkalosis.

� COMPENSATION H 2 O + CO 2 ↔ H 2 CO 3 ↔ H++ HCO 3 - HYPOVENTILATION- to retain co 2 Renal mechanismexcretes more bicarbonate and retains H+

� Primary deficit of carbonic acid causes • Hyperventilation • High altitude • Salicylate poisoning

� Compensation H 2 O + CO 2 ↔ H 2 CO 3 ↔ H++ HCO 3 - Renal mechanism – � by increasing excretion of bicarbonate by decreasing reabsorption secretion of H+ decreases

� Potassium- affects contractility of heart � Hypokalaemia � Insulin - life threatening – increases K+ uptake by cells � Measurement of plasma k+ concentration assumes significance in acid-base disorders.

� Patients with severe uncontrolled DM acidosis) is usually with hypokalemia. (M. � When such a patient is given insulin , it stimulates k+ entry into cells. � The result is that plasma k+ level is further depleted. � Hypokalemia affects the heart functioning and is life threatening.

� Therefore in the treatment of diabetic ketoacidosis, k+ has to be given. K+ and alkalosis Hypokalemia leads to increased excretion of hydrogen ions, and thus may cause M. alkalosis.

� Assessment � Arterial of acid base status. blood Radial artery Brachial artery Femoral artery – in the Wrist - in the arm - in the groin

![parameter value [H+] 35 -43 mmol/l p. H 7. 35 -7. 45 pco 2 parameter value [H+] 35 -43 mmol/l p. H 7. 35 -7. 45 pco 2](http://slidetodoc.com/presentation_image_h/7cb13ff064d6daff3bc261ee2d573dea/image-62.jpg)
parameter value [H+] 35 -43 mmol/l p. H 7. 35 -7. 45 pco 2 4. 5 -6. 0 34 -45 1. 02 -1. 35 kpa mm. Hg mmol/l po 2 10. 5 -13. 5 kpa Bicarbonate 24 -30 mmol/l
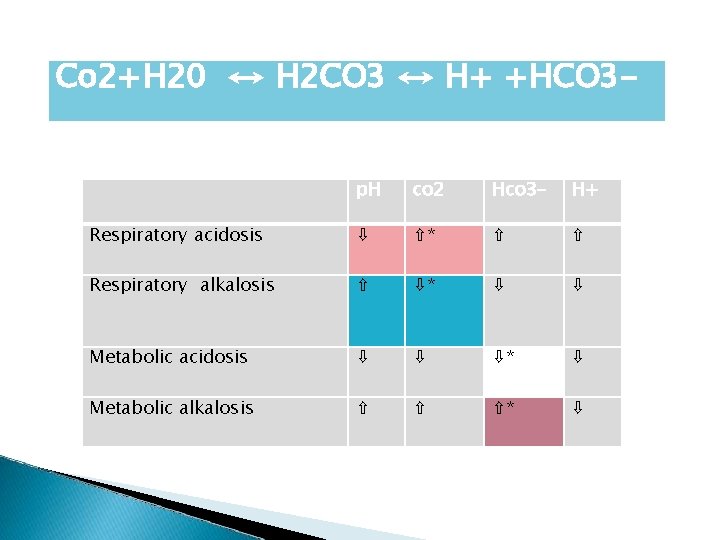
Co 2+H 20 ↔ H 2 CO 3 ↔ H+ +HCO 3 p. H co 2 Hco 3 - H+ Respiratory acidosis ⇩ ⇧* ⇧ ⇧ Respiratory alkalosis ⇧ ⇩* ⇩ ⇩ Metabolic acidosis ⇩ ⇩ ⇩* ⇩ Metabolic alkalosis ⇧ ⇧ ⇧* ⇩

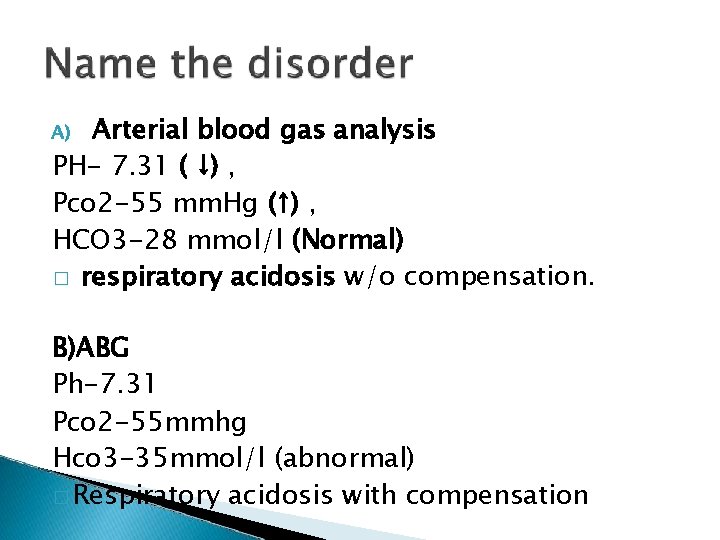
Arterial blood gas analysis PH- 7. 31 ( ↓) , Pco 2 -55 mm. Hg (↑) , HCO 3 -28 mmol/l (Normal) � respiratory acidosis w/o compensation. A) B)ABG Ph-7. 31 Pco 2 -55 mmhg Hco 3 -35 mmol/l (abnormal) � Respiratory acidosis with compensation

� TIETZ TEXT BOOK OF CLINICAL CHEMISTRY � VARLEY’S CLINICAL CHEMISTRY � MICHAEL L. BISHOP, CLINICAL CHEMISRY , SEVENTH EDITION. � DM VASUDEVAN TEXTBOOK OF BIOCHEMISTRY � U. SATYANARAYANA TEXTBOOK OF BIOCHEMISTRY � LAWERENCE A. KAPLAN A TEXT BOOK OF CLINICAL CHEMISTRY � DR. R. N. ROY A TEXT BOOK OF BIOPHYSICS

- Slides: 67