Introduction History background Basic Ethical Principles Ethical issue


Ä Introduction Ä History background Ä Basic Ethical Principles Ä Ethical issue in Human Research Ä Ethical Issue in Research to Use Data Sets Ä Ethical issue in Animal Research Ä Professional ethical misconducts Ä Guidelines of Ethical Consideration in Research Ä Informed Consent Ä IRB Ä Case Discuss

Introduction Ä Moral refers to the individual values and self-constraints Ä Ethic refers to the mechanism provided by either a committee or similar organization whose main role is to conduct ethical assessment institutional policy refers to the rules set up by the institutions themselves Ä Law refers to the government's imposition of constraints regulations、acts、decrees、orders、codes、 administrative rules or guidelines on organizations and individuals

Introduction Ä What is the research ethics? It is a criterion or principle for what is “right” or“good”in the research process. One kind of strict self-request morals or the value system that the researcher should behave himself. The researchers must have higher moral requirement than the general communicants.
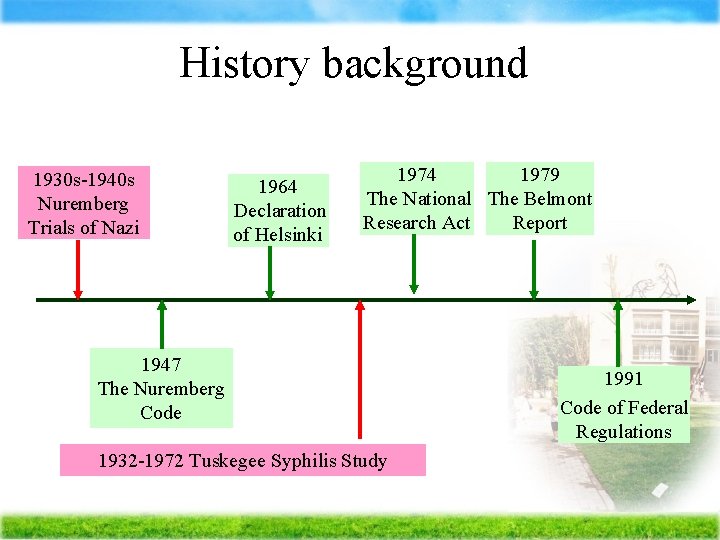
History background 1930 s-1940 s Nuremberg Trials of Nazi 1964 Declaration of Helsinki 1974 1979 The National The Belmont Research Act Report 1947 The Nuremberg Code 1932 -1972 Tuskegee Syphilis Study 1991 Code of Federal Regulations

The Nuremberg Code Ä The Nuremberg Code was adopted by the United Nations General Assembly in 1948 Ä the first major international document to provide guidelines on research ethics Ä provide a basis for modern standards on human research

The Nuremberg Code (con’t) Ä Ä Ä Ä participants are able to consent free from coercion (i. e. , outside pressure) they comprehend the risks and benefits involved states that researchers should minimize risk and harm make sure that risks do not significantly outweigh potential benefits appropriate study designs guarantee participants' freedom to withdraw at any time

Declaration of Helsinki Ä At the 18 th World Medical Assembly in Helsinki, Finland, the World Medical Association adopted 12 principles to guide physicians on ethical considerations related to biomedical research. Ä It emphasizes the distinction between medical care that directly benefits the patient and research that may or may not provide direct benefit. Ä These guidelines were revised at subsequent meetings 最新版為 2000年版

The National Research Act Ä The U. S. Congress signed this act into law, creating the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research. Ä The Commission was charged with: identifying the basic ethical principles that should govern medical research involving people, and then recommending steps to improve the Regulations for the Protection of Human Subjects

The Belmont Report Ä the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research issued Ä Ethical Principles and Guidelines for the Protection of Human Subjects of Research. Ä The report sets forth three principles underlying the ethical conduct of research: respect for persons beneficence justice

Code of Federal Regulations Ä This policy was adopted to ensure a uniform system of protections in all federal agencies and departments that conduct research Ä Policy for the protection of human subjects Ä is the main legal guideline for research requirement

Basic Ethical Principles Ä Beneficence Ä Respect Ä Justice for Persons

Beneficence Ä do no harm (passive) Physical —death, disability, infection Psychological —depression and anxiety Social —discrimination Economic —job loss Ä Nonexploitation putting the needs of researcher ahead of participants fail to correct a participants’ misconceptions about the role of researcher or the goal of study taking personal advantage of trust relationship between researcher and participant for personal gain

Beneficence (con’t) Ä promote good (active) tangible benefits as a result of participant or broader society reimbursement-mileage, stipend, payment incentives-monetary payments, special treatment more information about individual health problem obtains more attention satisfy individual curiosity Ä Risk-beneficence ratio minimal risk- equivalent to or less than the risk that are encountered in everyday life or while performing include many routine daily activities (by Federal Regulations)

Beneficence (con’t) Ä Risk-beneficence ratio (con’t) minimize risk, increase benefits stop or redesign research when the risk overweigh the benefits Ä Resolve: design the meaningful research subject IRB highly alert to the potential risk in the entire research process

Respect for Persons Ä autonomy right to self-determination to decision- making about participation free from coercion right to clarifying the research aim and process right to discontinuing research at any time right to refusing answer research question Ä full disclosure right to full information to decision-making about participation oral and written explanation special issue about respect for person

Respect for Persons(con’t) special issue about respect for person (con’t) specific level of disclosure vulnerable population ‡women of childbearing age ‡pregnant women ‡minors ‡students ‡low-income persons ‡institutionalized persons ‡incompetent persons or those with diminished capacity to give consent Ä Resolve: informed consent covert data collection deception

Justice Ä right to privacy confidential data collection anonymous data collection Ä right to fair and equitable treatment equal access to the benefits equal exposure to the risk people who decline to participate, do not participate fully, withdraw from study are treat as other participants treat with respect and civility at all times access to research staff at any time as needed

Justice (con’t) Ä Resolve: provide assurance privacy safeguards— only for necessary for the purpose of research only by participant identification numbers destroy identifying information whenever feasible and as soon as possible

Ethical Issue in Human Research
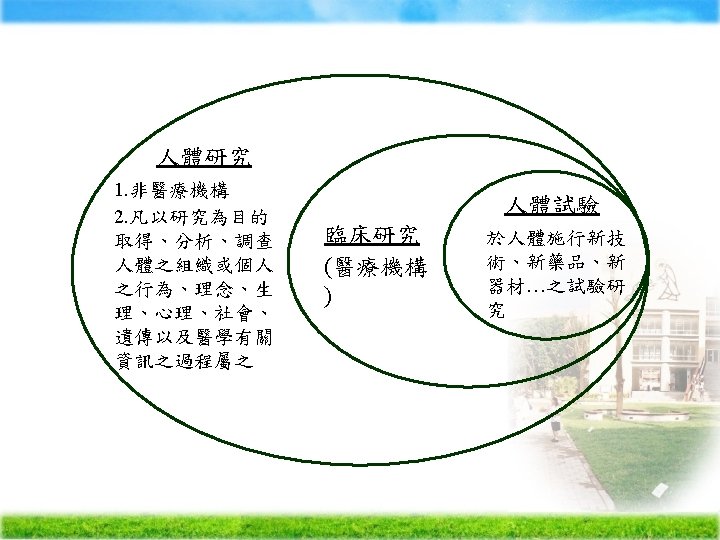



Ethical Issue in Research to Use Data Sets

Ethical Issue in Research to Use Data Sets Ä issue- privacy—personal health information lack of informed consent Ä discussion as a type of “covert “ research, but should never be used as substitute of the informed consent stripped of individual identifiers IRB’s request— Remove all of the potential identifiers Propose plan to safeguard the transfer and storage of data set exempt from full board review


Ethical Issue in Animal Research

Ethical Issue in Animal Ä Science ‘‘must’’ move forward. Ä Patients ‘‘must’’ have better medicines and treatments. Ä Laboratory animals ‘‘must’’ be treated in a responsible way. Adopt from: Different views on ethics: how animal ethics is situated in a committee culture

Different views on ethics in the name of animals Ä animal welfare Ä It is to make sure the laboratory animals have a fair existence Ä animals’ well-being, such as end points housing handling Ä social environment for the animals Ä animals have rights-one of these rights is to not suffer Adopt from: Different views on ethics: how animal ethics is situated in a committee culture

Different views on ethics in the name of science Ä defined ethics as obstacles, their aim being to stop scientific progress and destroy the hopes and expectations of many severely ill people who are dependent on animal experiments. Adopt from: Different views on ethics: how animal ethics is situated in a committee culture

Different views on ethics in the name of patients ÄI don’t care about the animals at all, I just care for humans Adopt from: Different views on ethics: how animal ethics is situated in a committee culture

Consensus Ä No research purpose is bad enough to be rejected. Ä if the research purpose is not good enough to justify animal suffering, the cost–benefit evaluation becomes complex Ä It is not unethical to use animals in research. It is unethical not to do so. Ä how to minimize the suffering of animals Ä agree on a humane end point , when the animal will be put to death Adopt from: Different views on ethics: how animal ethics is situated in a committee culture

Declaration of Helsinki Ä 動物保護法(民國97年 1月16日修正) Ä

Professional ethical misconducts

Professional ethical misconducts Ä Falsification or fabrication of data Ä Plagiarism Ä Publication of same data more than once Ä Lack of publication of research findings Ä Inadequate supervision of juniors or students

Professional ethical misconducts (con’t) Ä Authorship blunders Omission of deserving people Inclusion of undeserving people: ‘Guest authors’ ‘Ghost authors’ Ä Skewed partnerships in collaborative research Ä Conflict of interest

Professional ethical misconducts (con’t) Ä Conducting ‘morally wrong’ and unethical research Ä Misappropriation of research funds or financial Mismanagement Ä Salary discrepancies in same research project Ä Payment per participant recruited or per sample collected Ä Whistle blowing Ä Brain drain

What Makes Clinical Research Ethical?

Guidelines of Ethical Consideration in Research Ä Worthiness of research Ä competence boundaries Ä informed consent Ä Favorable Risk-Benefit Ratio Ä harm and risk Ä honesty and trust Ä Privacy, anonymity and security Ä intervention and advocacy Ä integrity and quality of research Ä ownerships of research material and Ä use and misuse of research results result

Guidelines of Ethical Consideration in Research Ä Worthiness of research Was there really a need for the research? Does the contribution of research conform to expectation of the publication and expenditure of the study? Ä Competence boundaries Is my professional accomplishment sufficient to cover the research quality? Am I ready to accept the surveillance or retraining and to seek other experts’ opinions, if my professional accomplishment is not good enough?

Guidelines of Ethical Consideration in Research Ä informed consent Does the participants fully understand information of the research? Does the participants take part in the research under free will? Ä Favorable Risk-Benefit Ratio Will the participants obtain what kind of advantage? Does participants obtain a balance between payout and reward? Ä harm and risk Will participants be harmed because enrolling in the research?

Guidelines of Ethical Consideration in Research Ä honesty and trust Do I and my object have a good interactive relationship? Do I write the findings honestly? Can we trust mutually? Ä Privacy, anonymity and security Do I invade the objects’ privacy boundary which they expect? How does my research information be kept safely? Whether my object of study in the paper be recognized easily?

Guidelines of Ethical Consideration in Research Ä intervention and advocacy When my object of study has illegal, a vulnerability or the incorrect behavior, how do I manage? When the accident occurring, should I need to defend the rights of any object of the study? If so, defend for what? Ä integrity and quality of research Does my research complete by careful and correct procedure? Does my research quality conform to the standard requirement of academic society?

Guidelines of Ethical Consideration in Research Ä ownerships of research material and result Who is the owner of my research material and result? Who has the right to own or publish my data? Whether my literature quotation infringe others’ copyright? Ä use and misuse of research results Do I have the duty to let the research results use or quote correctly? ? How will I do when my research results are used or quote wrongly?

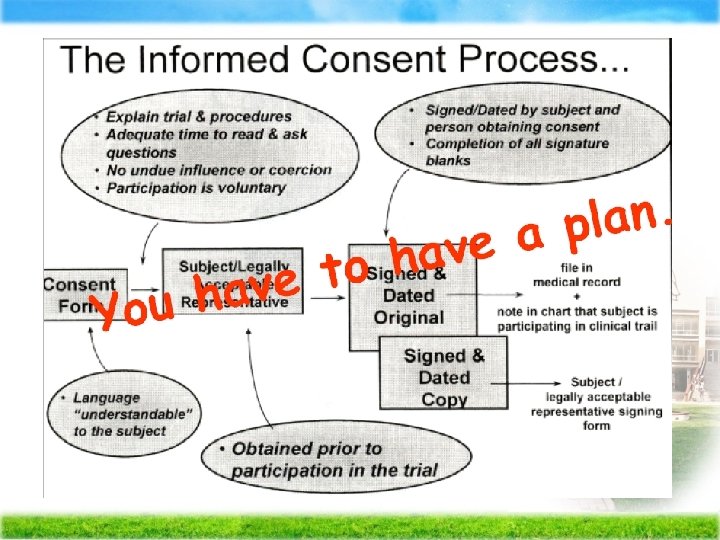
Importance of Informed Consent p. To assure that participation is voluntary and that the rights, welfare and safety of subjects are protected Tuskeegee Trial

Meaning of Informed Consent p Participants have adequate information regarding the research � are capable of comprehending the information � and have the power of free choice � enabling them to consent to or decline participation voluntarily.

Process Consent p Researcher continually renegotiates the consent� allowing participants to play a collaborative role in the decision-making process regarding on going participation
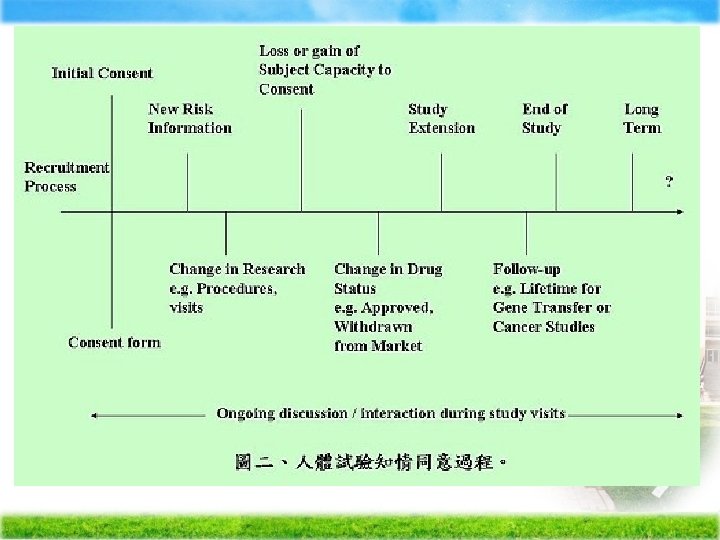
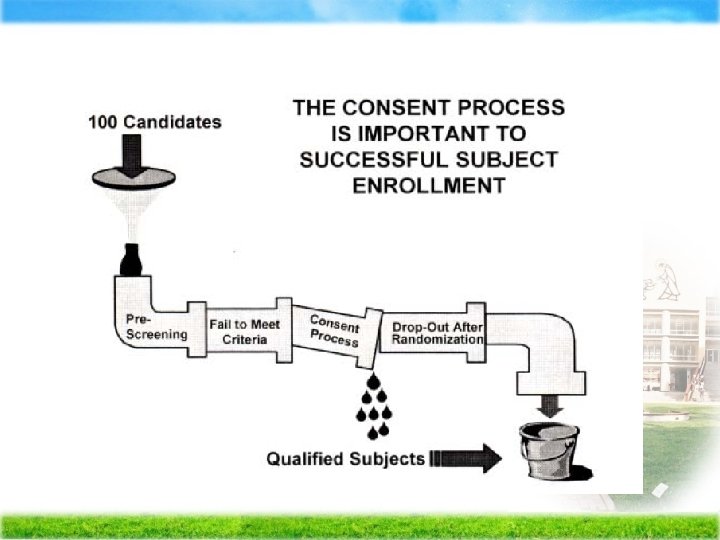

Question: Must we need the consent form?

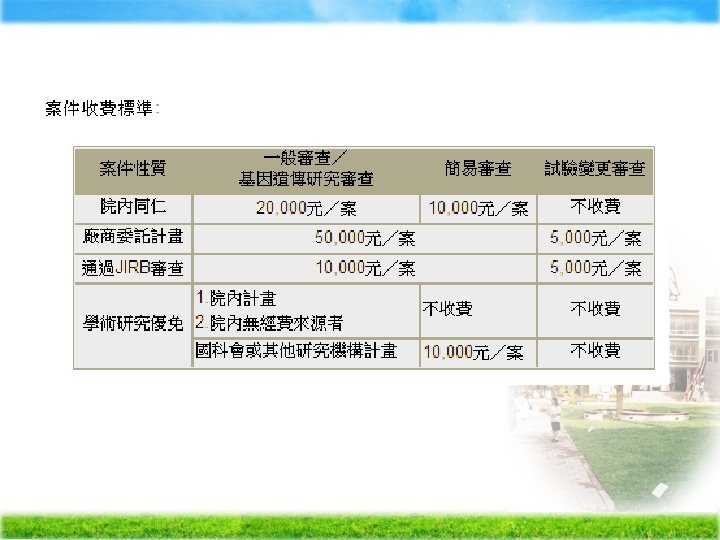
Waive to obtain informed consent Ä Ä Ä Ä Ä (1) The research involves no more than minimal risk to the subjects (2) The waiver or alteration will not adversely affect the rights and welfare of the subjects (3) The research could not practicably be carried out without the waiver or alteration (4) Whenever appropriate, the subjects will be provided with additional pertinent information after participation. 進行人的研究,即使符合免除受試者同意書要件, 還是需要預先進行IRB審查

The content of the informed consent is defined by ICH-GCP required elements 2. Country specific legal/regulatory requirements 3. Sponsor specific requirements 4. Study specific requirements 1.

The Elements of Consent Form 1. Title of research 2. Research purposes 3. How to recruit subjects 4. Explain research procedures 5. Describe risk/discomfort 6. Describe benefit/reward 7. State the possible benefits for alternative treatments or procedures

The Elements of Consent Form (con’t) 8. Describe anonymity and confidentiality 9. Sign the consent form if he/she agrees to participate in this study 10. Provide chances for asking questions 11. State the right for withdrawing at anytime of study





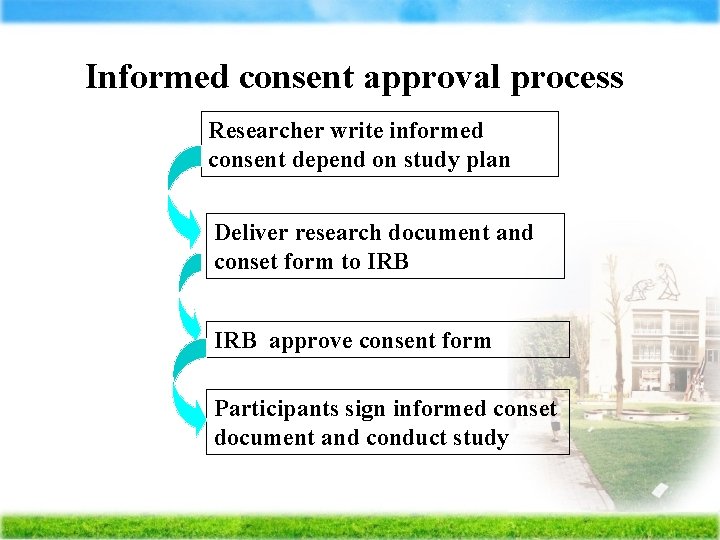
Informed consent approval process Researcher write informed consent depend on study plan Deliver research document and conset form to IRB approve consent form Participants sign informed conset document and conduct study

Consent Process Plan Ø Identify words subject may understand in consent form Ø Compile list of question the subjects may ask about the study- Informed Consent Evaluation Feedback Tool (ICE FT)

Consent Process Plan Decide who will conduct consent discussion Ø Principal Investigator Ø Investigator may delegate to a knowledgeable person

Consent Process Plan Ø Provide adequate time to explain the study and study procedures to the subject Ø Provide adequate time for subject to read and consider Ø Provide time for question to be answered

When may the consent document be revised Ø Protocol amendment Ø New relevant safety information Ø New information becomes available that might influence the subject`s decision to participate / continue in the clinical study

Comprehension of Inform Consent Oral Explanations not only Communicate but Understand Ø Simple Language Ø Avoid Jargon and Technical Ø Whitten statements for participants` reading levels educational attainment Ø Ø

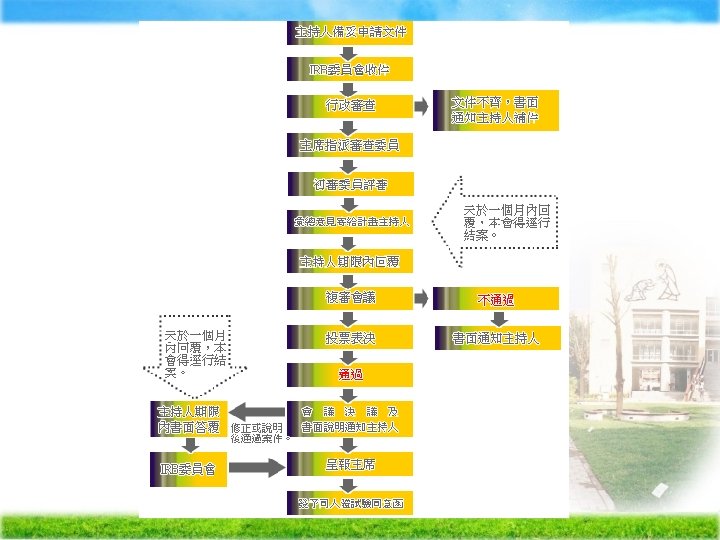
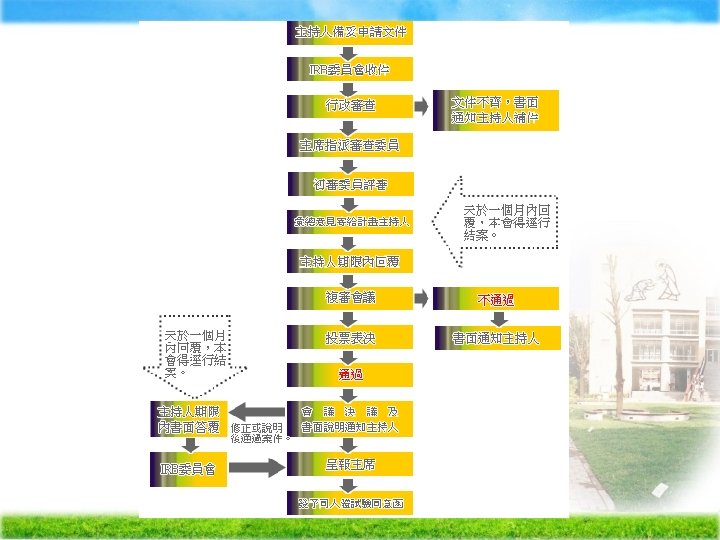
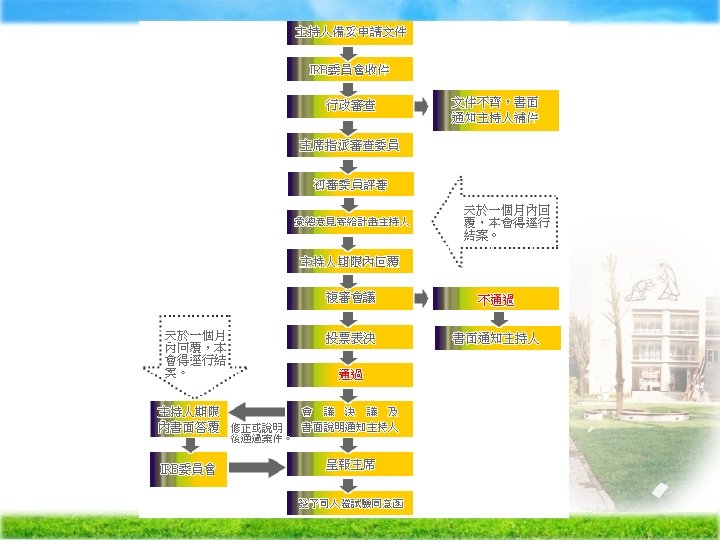
Institutional Review Broad (IRB) Ä The purpose of IRB review -assure, both in advance and by periodic review, that appropriate steps are taken to protect the rights and welfare of humans participating as subjects in the research.

Ä IRBs use a group process to review research protocols and related materials (e. g. , informed consent documents and investigator brochures) to ensure protection of the rights and welfare of human subjects of research.

IRB membership (45 CFR 46. 107) at least five members no entirely of men or women and at least one Profession Ø at least one scientific member Ø at least one member who is not otherwise affiliated with the Institution Ø Remove conflicting interest , except to provide information Ø invite individuals with competence to assist in the review of issues , but may not vote with the IRB Ø Ø


Criteria for IRB approval of research Ø Risks to subjects are minimized to subjects are reasonable in relation to anticipated benefits Ø Selection of subjects is equitable Ø Informed consent will be sought from each prospective subject Ø the subject's legally authorized representative

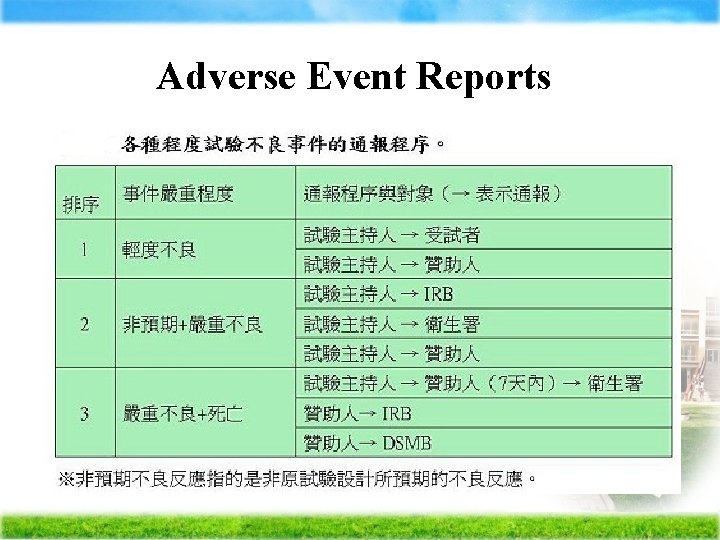
Adverse Event Reports

IRB records Ø all research proposals(consent documents, progress reports, reports of injuries to subjects) Ø minutes of IRB meetings Ø continuing review activities Ø all correspondence between the IRB and the investigators Ø a list of IRB members

Vulnerable research subject p Children p Pregnant women p Prisoners Ø Ø Mentally or emotionally disable people Severely ill or physically disabled people The terminally ill Institutionalized people









Vulnerable subjects Children If the children is developmentally mature (eg. 13 year-old) to understand the consent, it is advisable to obtain written consent from children. Ø Assent: children’s affirmative agreement to participate. Ø The child shall have the right to freedom of expression; this right shall include the freedom to seek, receive and important information and ideas of all kind, … (UNCRC, article 13(1)) Ø Ä

Vulnerable subjects-child ÄQualitative research involving children and elderly people (Jokinen, Lappalainen, Merilainen, & Pelkonen, 2002) Ø Informant-researcher relationship Home environment Age Ø Risk and benefits

Vulnerable subjects Mentally or intellectual disabled people informed consent may also obtained from someone who is interested in subject’s welfare supported decision-making benefit to participants (Iacono, 2006)

Vulnerable subjects Severely ill It might be necessary to assess their ability to make reasoned decisions about study participation. eg. mechanically ventilated patient Alternative procedure for informed consent should be used if the participants have physical impairment prevent them from reading or writing.


Vulnerable subjects The terminal ill The risk/benefit ratio needs to be carefully assessed. The health care and comfort of the participants are not compromised.

Vulnerable subjects ÄPregnant ÄA women pregnant woman cannot be involved in a study unless the purpose of the research is to meet her health needs and the risk of her and the fetus are minimized.

Review of ethical principals Ä Beneficence Ä Ä Respect for persons Ä Ä Freedom from harm Freedom from exploitation The risk/benefit ratio Autonomy/Self-determination Full disclosure Informed consent Justice Ä Ä The right to privacy The right to fair treatment

Use the example of

Beneficence 1. What is the potential benefit in this study? 2. Was any type of coercion to recruit participants? 3. What is the potential harm of participation in this study?


Respect for persons 1. Did the participants fully understand the purpose of the study? 2. Were the informed consent process appropriate used? 3. Did the participants have the right to decide whether to participate the study?

Process of informed consent

Justice 1. Were adequate steps taken to safeguard the privacy of participants? 2. Were special precautions instituted because of their vulnerable status?

- Slides: 104