INTRODUCTION Glycobiology is the study of the roles

INTRODUCTION Glycobiology is the study of the roles of sugar in health and disease Glycome is the entire complement of sugar, whether free or present in more complex molecules of an organism Glyconomics is the comprehensive study of glycome, including, genetic, physiologic, pathologic and other aspects Glycoproteins are proteins that contain oligosaccharide chains(glycans) covalently attached to their polypeptide backbone Glycosylation is the enzymatic attachment of sugar to protein Glycation is the non enzymatic attachment of sugar to protein The carbohydrate content of glycoprotein ranges from 1% to 85% by weight

NOMENCLATURE Glycoprotein, Proteoglycan, Glycosaminoglycan A glycoprotein is a compound containing carbohydrate (or glycan) covalently linked to protein. The carbohydrate may be in the form of a monosaccharide, disaccharide(s). oligosaccharide(s), or their derivatives (e. g. sulfo- or phospho-substituted). One, a few, or many carbohydrate units may be present. Proteoglycans are a subclass of glycoproteins in which the carbohydrate units are polysaccharides that contain amino sugars. Such polysaccharides are also known as glycosaminoglycans.

DIFFERENCES BETWEEN GLYCOPROTEIN AND GAG’S GLYCOPROTEINS Length of carbohydrate chain is short(2 -10 sugar units) No diglucosyl repeated units Chains may or may not be negatively charged Carbohydrate contain is variable(4 -85%) Storage diseases are called oligosaccharidosis GLYCOSAMINOGLYCANS Carbohydrate chain is long Diglucosyl repeated units are present Chains are negatively charged Carbohydrate contain is more than 95% Storage diseases are called mucopolysaccharidosis

Glycopeptides, glyco-amino-acids and glycosylamino-acids A glycopeptide is a compound consisting of carbohydrate linked to an oligopeptide composed of L- and/or D-amino acids. A glyco-amino-acid is a saccharide attached to a single amino acid by any kind of covalent bond. A glycosyl-amino- acid is a compound consisting of saccharide linked through a glycosyl linkage (O-, Nor S-) to an amino acid. (The hyphens are needed to avoid implying that the carbohydrate is necessarily linked to the amino group. )

Peptidoglycans. A peptidoglycan consists of a glycosaminoglycan formed by alternating residues of D-glucosamine and muramic acid The carboxyl group of the muramic acid is commonly substituted by a peptide containing residues of both L- and D-amino acids, whereas that of L-talosaminuronic acid is substituted by a peptide consisting of L-amino acids only.
• Almost all the plasma proteins of humanswith the notable exception of albumin-are glycoproteins. 6

• Glycosylation (enzymic attachment of sugars) is the most frequent post-translational modification of proteins. • Nonenzymic attachment of sugars to proteins can also occur, and is referred to as glycation. 7
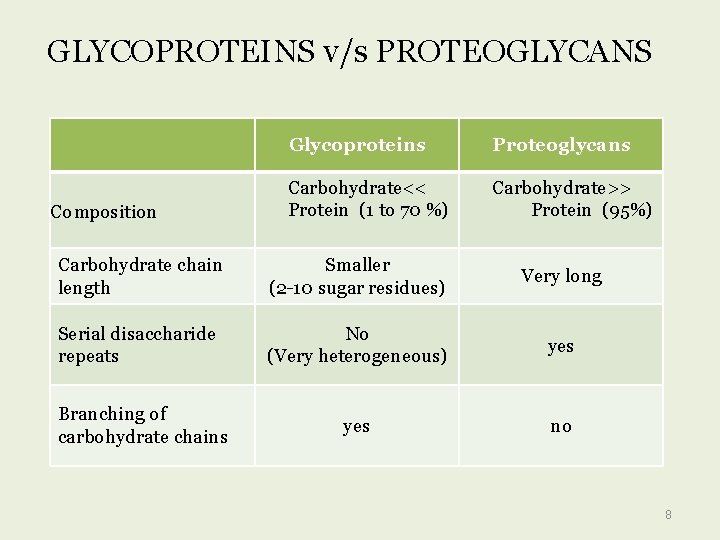
GLYCOPROTEINS v/s PROTEOGLYCANS Composition Glycoproteins Proteoglycans Carbohydrate<< Protein (1 to 70 %) Carbohydrate>> Protein (95%) Carbohydrate chain length Smaller (2 -10 sugar residues) Very long Serial disaccharide repeats No (Very heterogeneous) yes no Branching of carbohydrate chains 8
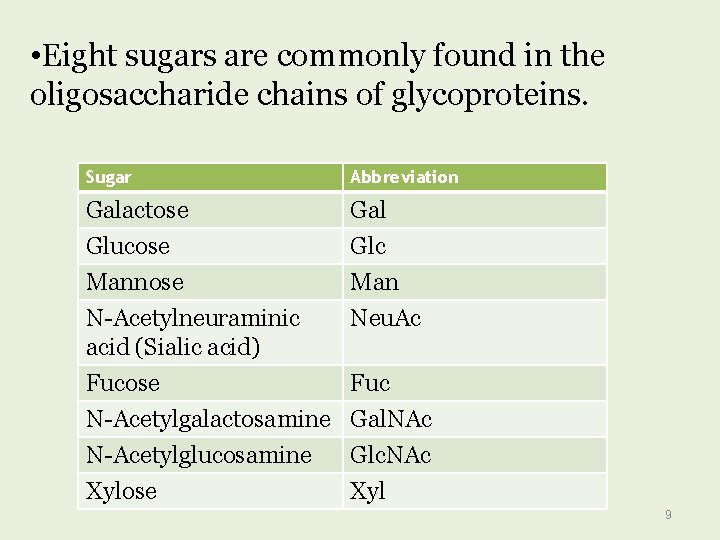
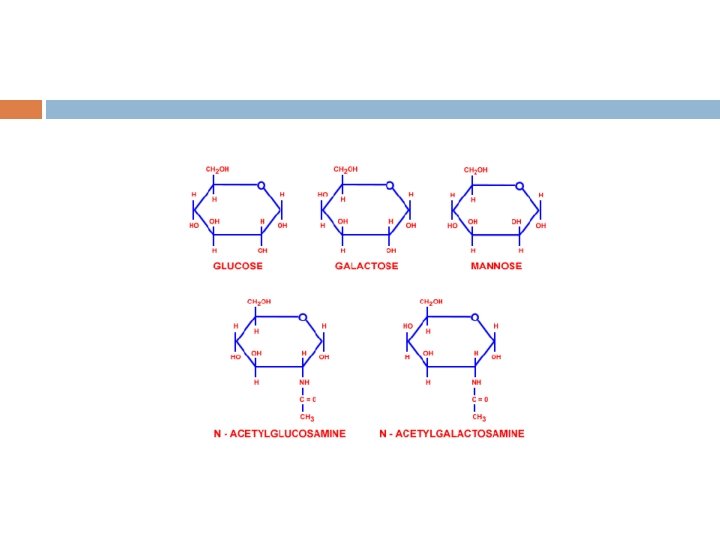
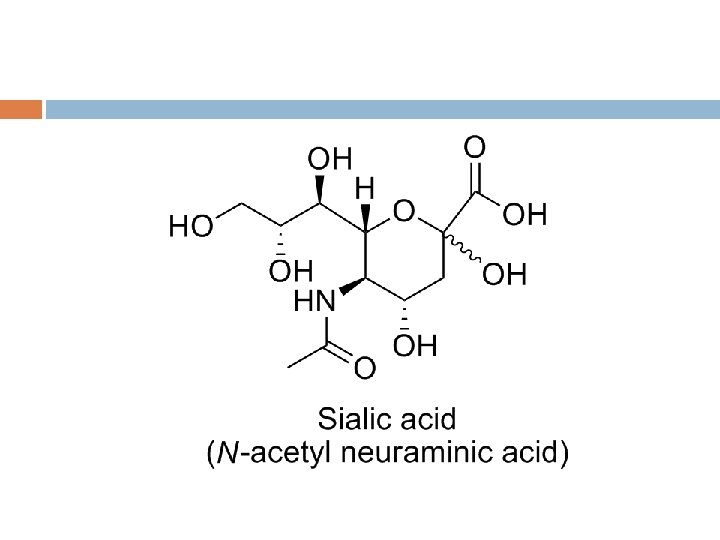
• Eight sugars are commonly found in the oligosaccharide chains of glycoproteins. Sugar Abbreviation Galactose Gal Glucose Mannose N-Acetylneuraminic acid (Sialic acid) Fucose N-Acetylgalactosamine Glc Man Neu. Ac N-Acetylglucosamine Xylose Glc. NAc Xyl Fuc Gal. NAc 9

The percentage of carbohydrate in glycoproteins is highly variable. • Some glycoproteins such as Ig. G contain low amounts (4%) of carbohydrate by weight, while glycophorin, the human red cell membrane glycoprotein, contains 60% carbohydrate 10

Functions of oligosaccharide chains of Glycoproteins: • Control of protein half-life in blood • Important determinant in receptor– ligand binding • May affect sites of metastases selected by cancer cells 11

FUNCTIONS OF GLYCOPROTEINS 12
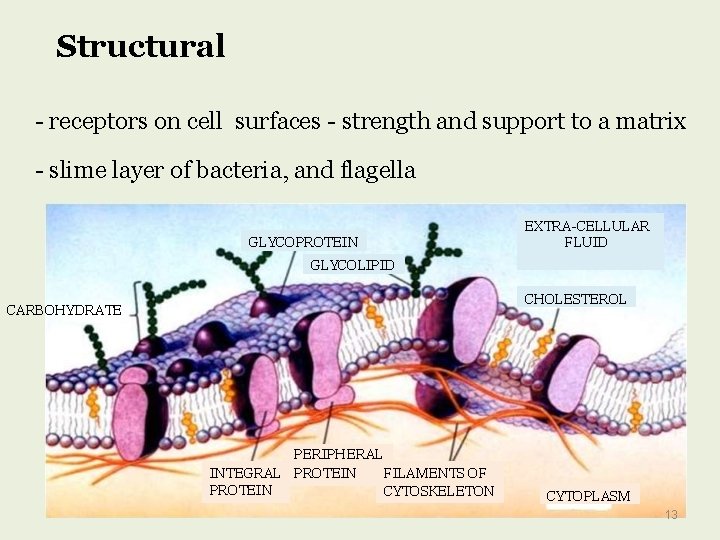
Structural - receptors on cell surfaces - strength and support to a matrix - slime layer of bacteria, and flagella GLYCOPROTEIN EXTRA-CELLULAR FLUID GLYCOLIPID CHOLESTEROL CARBOHYDRATE PERIPHERAL INTEGRAL PROTEIN FILAMENTS OF PROTEIN CYTOSKELETON CYTOPLASM 13

Protection • Mucin … form a highly viscous gel – Protect internal epithelial surfaces • Act as a lubricant – Human lacrimal glands produce a glycoprotein which protects the corneal epithelium 14
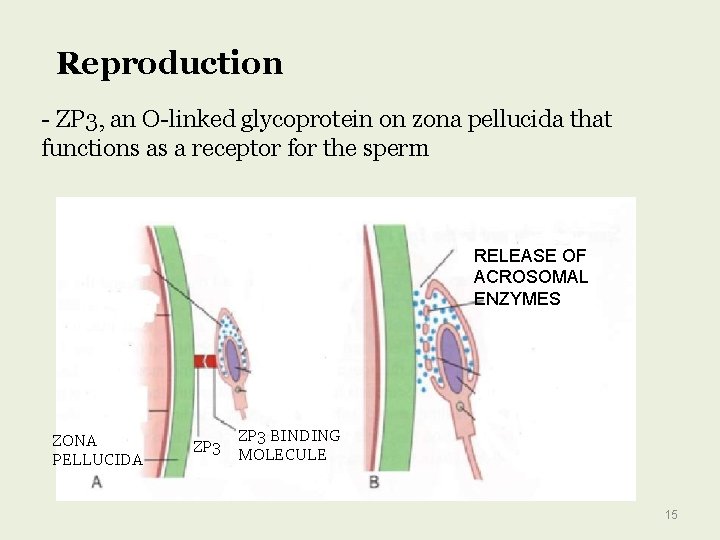
Reproduction - ZP 3, an O-linked glycoprotein on zona pellucida that functions as a receptor for the sperm RELEASE OF ACROSOMAL ENZYMES ZONA PELLUCIDA ZP 3 BINDING MOLECULE 15
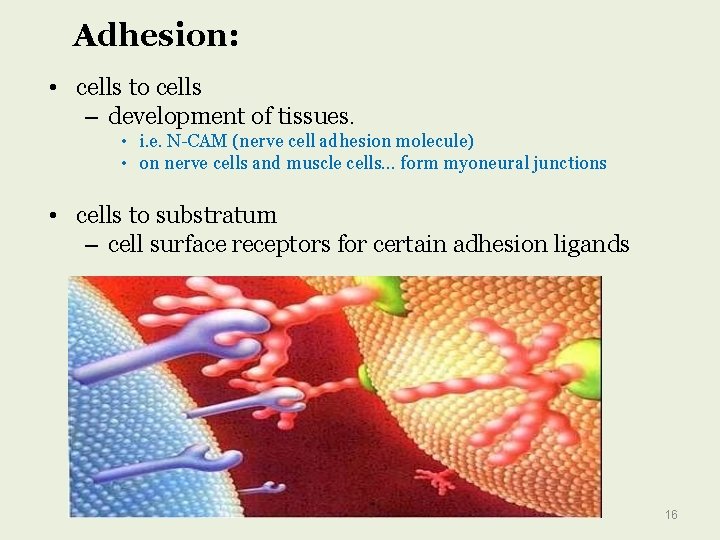
Adhesion: • cells to cells – development of tissues. • i. e. N-CAM (nerve cell adhesion molecule) • on nerve cells and muscle cells… form myoneural junctions • cells to substratum – cell surface receptors for certain adhesion ligands 16

Hormones - Chorionic gonadotropin, - Thyroid stimulating hormone (TSH) Enzymes – Oxidoreductases – Transferases – Hydrolases 17
Glycans Permeate Cellular Functions 18

Functions of glycoproteins (elaborated) http: //www. cs. stedwards. edu/chem/Chemistry/CHEM 43/Glycoproteins. HTML Carbohydrates and proteins by themselves serve in a vast number of biological functions, linking the two together results in a macromolecule with an extremely large number of functions. Structural: Glycoproteins are found throughout matrices. They act as receptors on cell surfaces that bring other cells and proteins (collagen) together giving strength and support to a matrix. In nerve tissue glycoproteins are abundant in gray matter and appear to be associated with synaptosomes, axons, and microsomes. Protection: Human lacrimal glands produce a glycoprotein which protects the corneal epithelium from desiccation and foreign particles. Human sweat glands secrete glycoproteins which protect the skin from the other excretory products that could harm the skin

Functions of glycoproteins (further elaborated-1) Prothrombin, and fibrinogen are all glycoproteins that play an intricate role in the blood clotting mechanism In certain bacteria the slime layer that surrounds the outermost components of cell walls are made up of glycoproteins of high molecular weight. In addition to forming these s-layers, glycoproteins also function as bacterial flagella. These are made up of bundles of glycoproteins protruding from the cell's surface. Their rotation provides propulsion. In plants, glycoproteins have roles in cell wall formation, tissue differentiation, & embryogenesis. Reproduction: Glycoproteins found on the surface of spermatozoa appear to increase a sperm cell's attraction for the egg by altering the electrophoretic mobility of the plasma membrane.

Functions of glycoproteins (further elaborated-2) Adhesion: Glycoproteins serve to adhere cells to cells and cells to substratum. Hormones: There are many glycoproteins that function as hormones such as human chorionic gonadotropin (HCG) which is present in human pregnancy urine. Another example is erythropoietin which regulates erythrocyte production Enzymes: Glycoprotein enzymes are of three types. These are oxidoreductases, transferases, and hydrolases.

Functions of glycoproteins (further elaborated-3) Carriers: Glycoproteins can bind to certain molecules and serve as vehicles of transport. They can bind to vitamins, hormones, cations, and other substances. Inhibitors: Many glycoproteins in blood plasma have shown antiproteolytic activity. For example, the glycoprotein a 1 -antichymotrypsin inhibits chymotrypsin. Immunological: The interaction of blood group substances with antibodies is determined by the glycoproteins on erythrocytes. Many immunoglobulins are actually glycoproteins. B and T cells contain surface glycoproteins that attract bacteria to these sites and bind them. In much the same manner, glycoproteins can direct phagocytosis. Because the HIV virus recognizes the receptor protein CD 4, it binds to helper T cells which contain it.

CELL MEMBRANE The cell membrane is a fluid mosaic of lipids, proteins, and carbohydrates. Membrane carbohydrates are usually branched oligosaccharides with fewer than 15 sugar units. Some of these oligosaccharides are covalently bonded to lipids, forming molecules called glycolipids. Most are covalently bonded to proteins, which are thereby glycoproteins. Plants produce pectin, major component of cell wall. The oligosaccharides on the external side of the plasma membrane vary from species to species The diversity of the molecules and their location on the cell's surface enable oligosaccharides to function as markers that distinguish one cell from another.

BIOMEDICAL IMPORTANCE Almost all the plasma proteins of humans—except albumin—are glycoprotein. Many proteins of cellular membranes contain substantial amounts of carbohydrate. A number of the blood group substances are glycoproteins, whereas others are glycosphingolipids. Certain hormones are glycoprotein. Hormones that are glycoprotein include: 1. Follicle-stimulating hormone 2. Luteinizing hormone 3. Thyroid-stimulating hormone 4. Human chorionic gonadotropin Alpha 5. fetoprotein Erythropoietin (EPO) 6.

Some Functions of Glycoproteins _________________________ Function Glycoprotein _________________________ 1. Structural molecule Collagens 2. Lubricant Mucins 3. Transport molecule e. g. Transferrin, Ceruloplasmin 4. Immune system Immunoglobulins, Histocompatibility antigens, Blood group determinants 5. Hormone e. g. HCG, TSH 6. Enzymes e. g. Alkaline phosphatase 7. Blood clotting e. g. Fibrinogen 8. Cell surface recognition Lectins _________________________

A major problem in cancer is metastasis, the phenomenon whereby cancer cells leave their tissue of origin (eg, the breast), migrate through the bloodstream to some distant site in the body (eg, the brain), and grow there in an unregulated manner, with catastrophic results for the affected individual. Many cancer researchers think that alterations in the structures of glycoproteins and other glyco conjugates on the surfaces of cancer cells are important in the phenomenon of metastasis
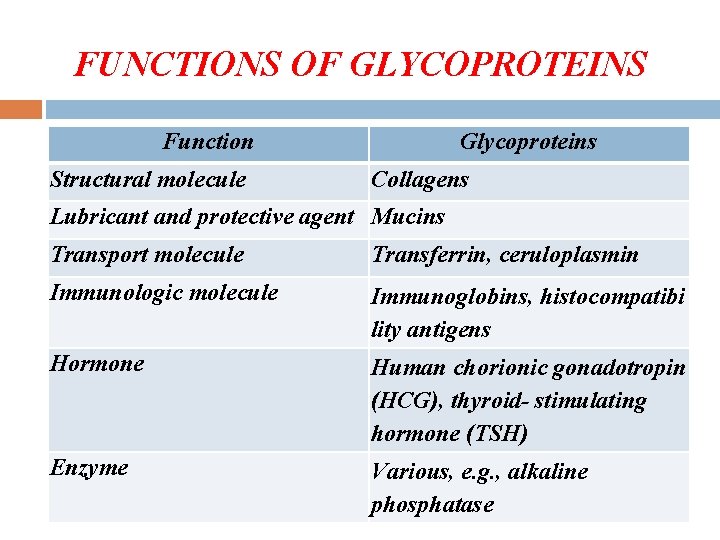
FUNCTIONS OF GLYCOPROTEINS Function Structural molecule Glycoproteins Collagens Lubricant and protective agent Mucins Transport molecule Transferrin, ceruloplasmin Immunologic molecule Immunoglobins, histocompatibi lity antigens Hormone Human chorionic gonadotropin (HCG), thyroid- stimulating hormone (TSH) Enzyme Various, e. g. , alkaline phosphatase
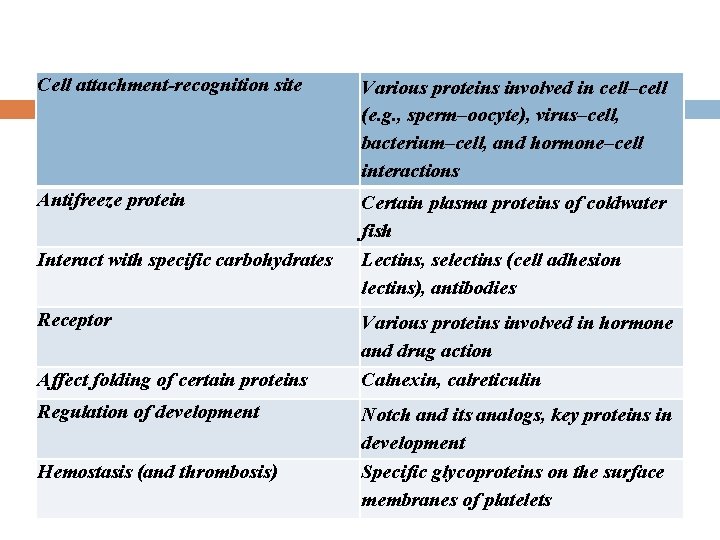
Cell attachment-recognition site Various proteins involved in cell–cell (e. g. , sperm–oocyte), virus–cell, bacterium–cell, and hormone–cell interactions Antifreeze protein Certain plasma proteins of coldwater fish Lectins, selectins (cell adhesion lectins), antibodies Interact with specific carbohydrates Receptor Affect folding of certain proteins Regulation of development Hemostasis (and thrombosis) Various proteins involved in hormone and drug action Calnexin, calreticulin Notch and its analogs, key proteins in development Specific glycoproteins on the surface membranes of platelets

SUGARS IN HUMAN GLYCOPROTEIN 200 monosaccharides are found in nature, however only 8 are commonly found in oligosaccharide chains of glycoproteins N-Acetylneuramic acid(Neu. Ac) is usually found at terminal of oligosaccharide chain, attached to subterminal galactose(Gal) or Nacetylgalactosamine(Gal-Nac) residue The other sugars are found in more internal positions Sulphate is often found in glycoproteins, usually attached to Gal, Gal. Nac, or Glc. Nac

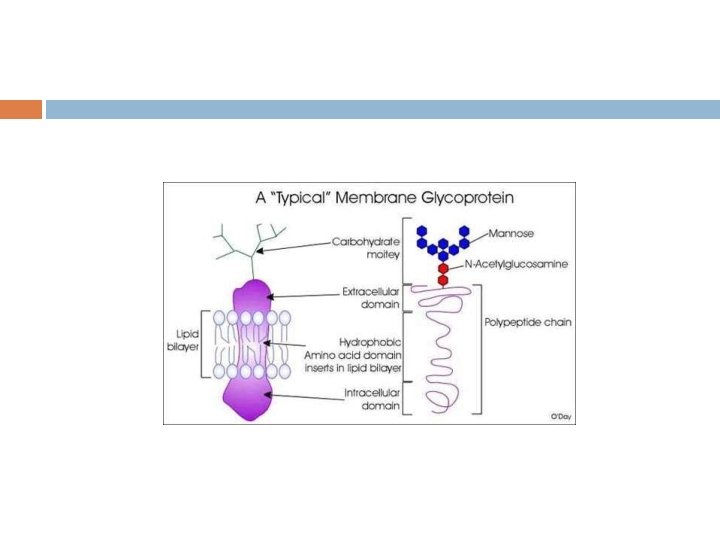
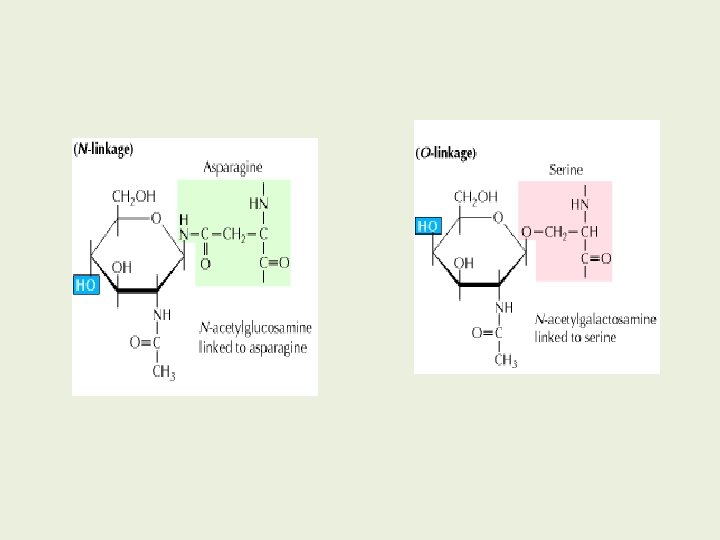
Based on the nature of the linkage between their polypeptide chains and their oligosaccharide chains, glycoproteins can be divided into three major classes : O-Linked N-Linked GPI-anchored 32
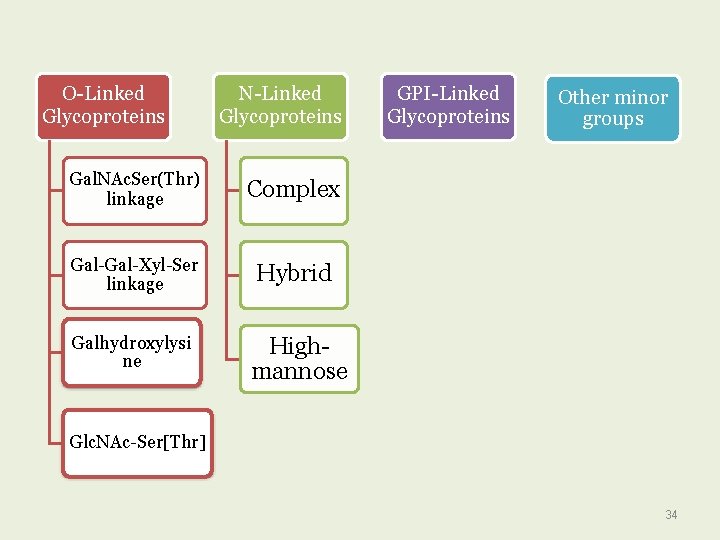
O-Linked Glycoproteins N-Linked Glycoproteins Gal. NAc. Ser(Thr) linkage Complex Gal-Xyl-Ser linkage Hybrid Galhydroxylysi ne Highmannose GPI-Linked Glycoproteins Other minor groups Glc. NAc-Ser[Thr] 34
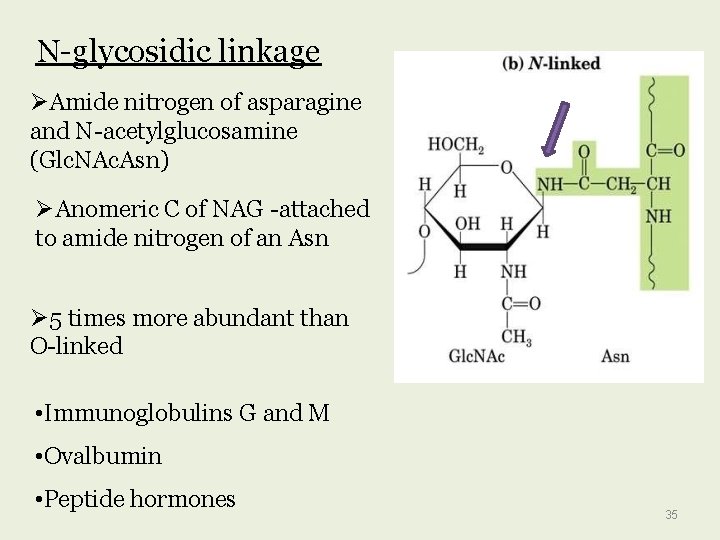
N-glycosidic linkage Amide nitrogen of asparagine and N-acetylglucosamine (Glc. NAc. Asn) Anomeric C of NAG -attached to amide nitrogen of an Asn 5 times more abundant than O-linked • Immunoglobulins G and M • Ovalbumin • Peptide hormones 35
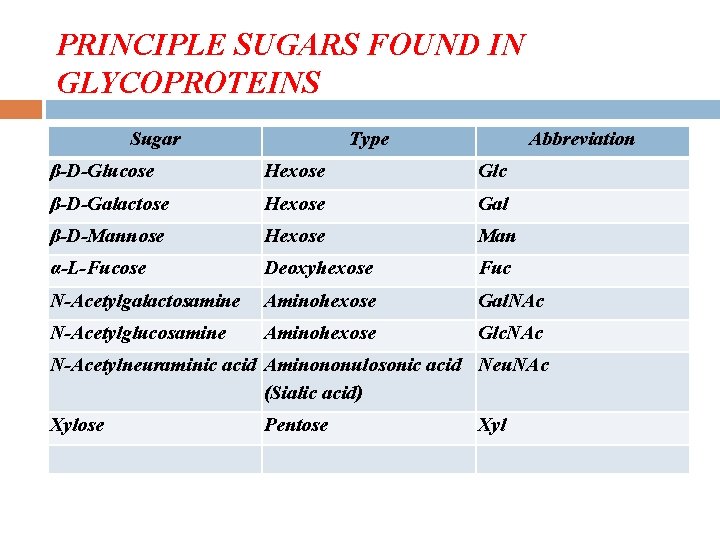
PRINCIPLE SUGARS FOUND IN GLYCOPROTEINS Sugar Type Abbreviation β-D-Glucose Hexose Glc β-D-Galactose Hexose Gal β-D-Mannose Hexose Man α-L-Fucose Deoxyhexose Fuc N-Acetylgalactosamine Aminohexose Gal. NAc N-Acetylglucosamine Aminohexose Glc. NAc N-Acetylneuraminic acid Aminononulosonic acid Neu. NAc (Sialic acid) Xylose Pentose Xyl

SOME FEATURES OF MUCIN Found in secretions of the gastrointestinal, respiratory, and reproductive tracts and also in membranes of various cells. • Exhibit high content of O-glycan chains, usually containing Neu. Ac. • Contain repeating amino acid sequences rich in serine, threonine, and proline. • Extended structure contributes to their high viscoelasticity. • Form protective physical barrier on epithelial surfaces, are involved in cell-cell interactions, and may contain or mask certain surface antigens.

LECTINS CAN BE USED TO PURIFY GLYCOPROTEINS & TO PROBE THEIR FUNCTIONS Lectins are carbohydrate-binding proteins that agglutinate cells or precipitate glycoconjugates; a number of lectins are themselves glycoproteins. Immunoglobulins that react with sugars are not considered lectins. Lectins contain at least two sugar-binding sites; proteins with a single sugar-binding site will not agglutinate cells or precipitate glycoconjugates. The specificity of a lectin is usually defined by the sugars that are best at inhibiting its ability to cause agglutination or precipitation. Enzymes, toxins, and transport proteins can be classified as lectins if they bind carbohydrate

Some functions of the oligosaccharide chains of glycoproteins Modulate physicochemical properties, eg, solubility, viscosity, charge, conformation, denaturation, and binding sites for bacteria and viruses • Protect against proteolysis, from inside and outside of cell • Affect proteolytic processing of precursor proteins to smaller products • Are involved in biologic activity, eg, of human chorionic gonadotropin (h. CG) • Affect insertion into membranes, intracellular migration, sorting and secretion • Affect embryonic development and differentiation • May affect sites of metastases selected by cancer cells

Glycoproteins Are Important in Fertilization To reach the plasma membrane of an oocyte, a sperm has to traverse the zona pellucida (ZP), a thick, transparent, noncellular envelope that surrounds the oocyte. The zona pellucida contains three glycoproteins of interest, ZP 1– 3. Of particular note is ZP 3, an O-linked glycoprotein that functions as a receptor for the sperm. A protein on the sperm surface, possibly galactosyl transferase, interacts specifically with oligosaccharide chains of ZP 3; this interaction, by transmembrane signaling, induces the acrosomal reaction, in which enzymes such as proteases and hyaluronidase and other contents of the acrosome of the sperm are released Liberation of these enzymes helps the sperm to pass through the zona pellucida and reach the plasma membrane (PM) of the oocyte

Selectins Play Key Roles in Inflammation Leukocytes play important roles in many inflammatory and immunologic phenomena. The first steps in many of these phenomena are interactions between circulating leukocytes and endothelial cells prior to passage of the former out of the circulation. Work done to identify specific molecules on the surfaces of the cells involved in such interactions has revealed that leukocytes and endothelial cells contain on their surfaces specific lectins, called selectins Selectins are single-chain Ca 2+-binding transmembrane proteins that contain a number of domains. Their amino terminal ends contain the lectin domain, which is involved in binding to specific carbohydrate ligands

Inherited Diseases I-cell disease, or leroy disease I-Cell disease is a severe and rare genetic disease, inherited in an autonomic recessive manner and caused by the lack of Golgi Glc. NAc phosphotransferase Clinical features. Typical patients present with mental retardation, coarse facial features with gingival hyperplasia, and short-trunk dwarfism; they generally die in the first decade of life
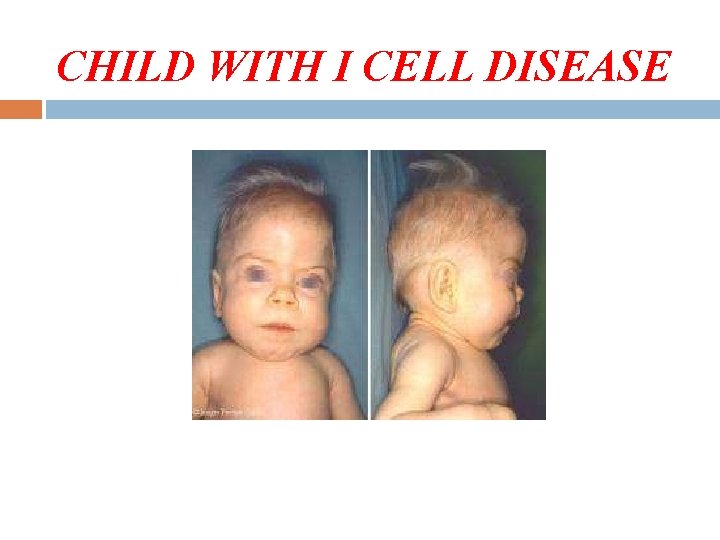
CHILD WITH I CELL DISEASE

Congenital disorders of glycosylation Another newly delineated group of carbohydrate-related genetic diseases is termed congenital disorders of glycosylation (CDG; previously known as carbohydrate-deficient glycoprotein syndrome). Two types are described, based on the enzymatic defect: type I corresponds to the CDG that involve enzymatic steps up to the assembling of the glycoprotein; type II are the CDG in which processing is involved. The most common one is CDG Ia (>300 patients worldwide), which is attributable to a deficiency in cytosolic phosphomanno mutase activity in the metabolism of mannose. Clinical features. CDG other than type Ib are multisystemic disorders involving neurological dysfunction. CDG Ia infants have a typical morphology with abnormal distribution of

Leukocyte-adhesion deficiency type ii Leukocyte-adhesion deficiency type II (LAD II) is a rare severe immunological disease. Clinical features LAD II is clinically similar to the well-described LAD I, in that is also involves severe mental retardation, short stature, and dysmorphic features. The two syndromes differ in the molecular basis of their adhesion defects. LAD I is caused by a deficiency in the CD 18 integrinadhesion molecule, whereas LAD II neutrophils are devoid of sialyl Lewis x, which is a ligand for the selectin family.

Hereditary erythroblastic multinuclearity with a positive acidified serum test (HEMPAS), also called congenital dyserythropoietic anemia type II, is a rare autosomal recessive disorder caused by membrane abnormality, with >300 known patients, and is a multifactorial disease. Clinical features. Congenital dyserythropoietic anemia type II patients suffer from a long-life anemia, hepatosplenomegaly, liver hemosiderosis, and cirrhosis.

Wiskott-aldrich syndrome Wiskott-Aldrich syndrome (WAS) is an X-linked, rare disease characterized by eczema, thrombocytopenia, and immunodeficiency. Clinical features. The clinical features begin early in life and include recurrent infections, bleeding, and eczema. The prognosis is grave. Circulating platelets are decreased in number and size. Ig. M concentrations usually are below normal, Ig. G concentrations are normal, and Ig. A and Ig. E concentrations are high. Monoclonal gammopathy is common.

Glycoproteinoses The common feature of these diseases is the genetic deficiency of any one of the lysosomal glycosidases involved in the catabolism pathway of glycoproteins (single-entity incidence between 1 in 100 000 and 1 in 250 000). Clinical features. Most of these diseases are characterized by different clinical phenotypes, depending the age of the patient. The clinical feature in the infantile form is most often dominated by neurologic symptoms. Retinal abnormalities are frequent.

PAROXSMAL NOCTURNAL HEMOGLOBINURIA(PNH) Acquired mild anaemia with presence of Hb in urine due to hemolysis of RBC, particularly in sleep Basic defect of PNH is somatic mutation of PIGA(phosphatidylinositol glycan class A) gene Product of this gene is enzyme that links PIG to GPI structure Diagnosis is by HAM’s TEST- RBC are more sensitive to hemolysis in normal serum acidified to p. H 6. 2

CONGENITAL MUSCULAR DYSTROPHY Defect in synthesis of glycan in protein alphadystroglycan(ALPHA-DG)

RHEUMATOID ARTHRITIS Associated with an alteration in glycosylation of circulating Ig. G molecules They lack galactose in their Fc regions and terminate in Glc. NAc

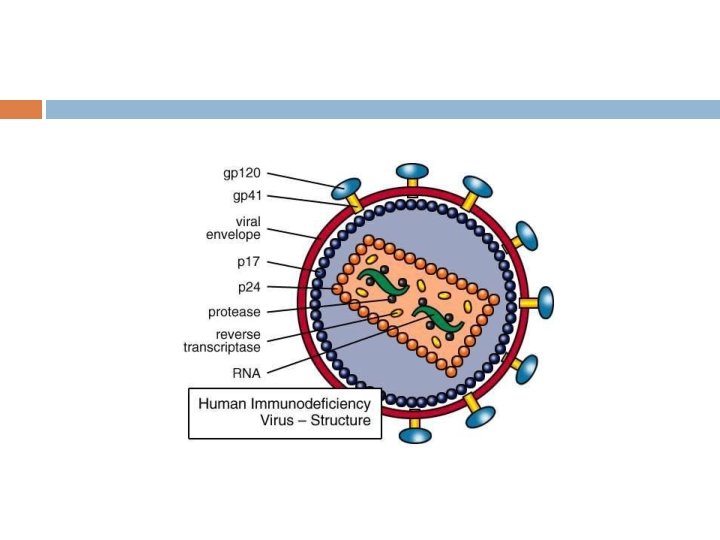
VIRAL DISEASES (1) The influenza virus possesses a neuraminidase that plays a key role in elution of newly synthesized progeny from infected cells. If this process is inhibited, spread of the virus is markedly diminished. Inhibitors of this enzyme are now available for use in treating patients with influenza. (2) HIV-1, causative agent of AIDS, attaches to cells via one of its surface glycoproteins, gp 120 and gp 41.

Advanced glycation end-product An advanced glycation end-product (AGE) is the result of a chain of chemical reactions after an initial glycation reaction. The intermediate products are known, variously, as Amadori, Schiff base and Maillard products, named after the researchers who first described them. AGEs may be less, or more, reactive than the initial sugars they were formed from. They are absorbed by the body during digestion with about 30% efficiency

Many cells in the body (for example, endothelial cells, smooth muscle, and cells of the immune system) from tissue such as lung, liver, kidney, and peripheral blood bear the Receptor for Advanced Glycation End-products (RAGE) that, when binding AGEs, contributes to age- and diabetes-related chronic inflammatory diseases such as atherosclerosis, asthma, arthritis, myocardial infarction, nephropathy, retinopathy, periodontitis a nd neuropathy They are also believed to play a causative role in the vascular complications of diabetes mellitus.

THANK YOU
- Slides: 58