INTRODUCTION BM 5 Short history of drug regulation

INTRODUCTION BM 5 Short history of drug regulation Jan Klimas

Currently - well organized pharmaceutical industry (in western countries), systematic and compliant to international regulatory standards for manufacturing of chemical and biological drugs for human and veterinary consumption as well as medical devices, traditional herbal products and cosmetics - well-defined controlled regulatory framework - systematic manufacturing and marketing of safe, efficacious and qualitative drugs - complexity created a need for regulatory professionals

Beginning of 1900’s in US first tendencies to regulate drugs by law - first trigger: vaccines tragedies in 1901. 1) diphtheria antitoxin developed by City Health Department of St. Louis was contaminated by tetanus causing bacteria (death of 14 children) 2) smallpox vaccine contaminated (death of 9 children) Consequence - adoption of The Biologics Control Act in 1902. - mandated manufacturing and distribution licensing of biological products i. e. serum, vaccine, toxin, viruses as well as defined labelling in terms of manufacturer’s name, address, license number, identification of product and expiry date, and so started the evolution of vaccine industry.

The first general American law to regulate medicines The Federal Pure Food and Drug Act in 1906 -made mandatory labelling of ingredients and its content for drugs (i. e. alcohol, cocaine, heroin, morphine, opium). - very weak, as it was only a requirement that drugs have to be free of adulterants and there was no mention regarding drug safety or effectiveness. - the starting point for the later creation of Food and Drug Administration (FDA). Originally, The Bureau of Chemistry was used to regulate food safety, however in 1927, it was reorganized into The Bureau of Chemistry and Soils and Food, Drug and Insecticide Administration. In 1930, the current FDA came into effect after shortening of earlier organization. Since the root of FDA was born in 1906, FDA still celebrates 1906 as its establishment year.

Basis of modern regulation = drug safety (predominantly) - modern medicines regulation started after breakthrough progress in life sciences - still predominantly, as a consequence of fatal drug failures worldwide - substantial increase of legislations for drug products quality, safety and efficacy - stricter norms for Marketing Authorization (MA) and Good Manufacturing Practices (GMPs).

Fatal drug failures In 1937, over 100 people in USA died of diethylene glycol poisoning following the use of a “Sulfanilamide Elixir” The „elixir“used the chemical as a solvent without any safety testing This facilitated the introduction of The Federal Food, Drug and Cosmetic Act (in 1938) to oversee safety of medicines. With this law, pre-marketing approval of all new drugs was made mandatory and proof for scientific safety study was asked by FDA. This law also mandated the directions for safe use.

Durham-Humphrey Amendment Act of 1951 (Prescription Amendment Act) -law for categorising medicines as 1) over-the-counter (OTC) drugs and 2) prescription drugs - medicines for minor ailments (indigestion, headache) can be fall under OTC drugs and freely sold at pharmacy store without prescription - major ailments drugs defined “Prescription Only (RX)” and unsafe for self medication - mandatory to put statement a labelling as “Caution. Federal Law prohibits dispensing without a prescription”
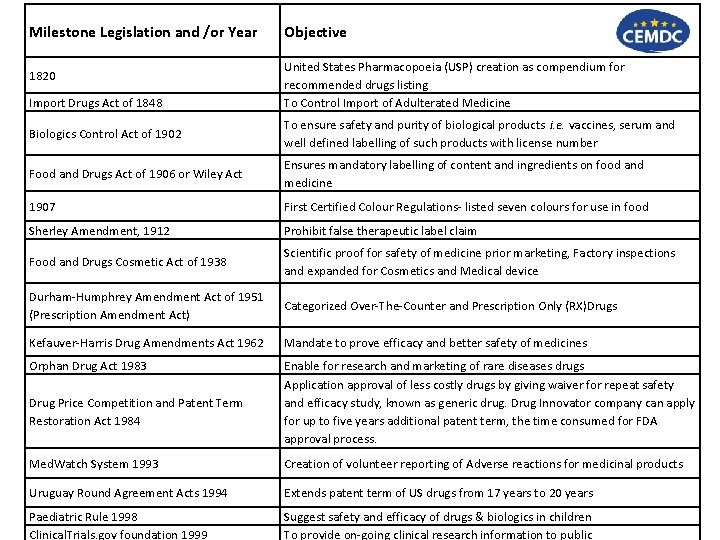
Milestone Legislation and /or Year 1820 Import Drugs Act of 1848 Objective United States Pharmacopoeia (USP) creation as compendium for recommended drugs listing To Control Import of Adulterated Medicine Biologics Control Act of 1902 To ensure safety and purity of biological products i. e. vaccines, serum and well defined labelling of such products with license number Food and Drugs Act of 1906 or Wiley Act Ensures mandatory labelling of content and ingredients on food and medicine 1907 First Certified Colour Regulations- listed seven colours for use in food Sherley Amendment, 1912 Prohibit false therapeutic label claim Food and Drugs Cosmetic Act of 1938 Scientific proof for safety of medicine prior marketing, Factory inspections and expanded for Cosmetics and Medical device Durham-Humphrey Amendment Act of 1951 (Prescription Amendment Act) Categorized Over-The-Counter and Prescription Only (RX)Drugs Kefauver-Harris Drug Amendments Act 1962 Mandate to prove efficacy and better safety of medicines Orphan Drug Act 1983 Enable for research and marketing of rare diseases drugs Application approval of less costly drugs by giving waiver for repeat safety and efficacy study, known as generic drug. Drug Innovator company can apply for up to five years additional patent term, the time consumed for FDA approval process. Drug Price Competition and Patent Term Restoration Act 1984 Med. Watch System 1993 Creation of volunteer reporting of Adverse reactions for medicinal products Uruguay Round Agreement Acts 1994 Extends patent term of US drugs from 17 years to 20 years Paediatric Rule 1998 Clinical. Trials. gov foundation 1999 Suggest safety and efficacy of drugs & biologics in children To provide on-going clinical research information to public

Milestone – the Thalidomide Disaster - The darkest episodes in pharmaceutical research history influenced the development of medicines regulation far more than any event in history worldwide - Predominantly, West Germany and Western Europe were affected - Thalidomide was synthesised (1954) by the West German Chemie Grünenthal Gmb. H, to expand the company’s product range beyond antibiotics, as an anticonvulsive drug - But, instead it, made users sleepy and relaxed. It seemed a perfect example of newly fashionable tranquilisers - During patenting and testing, scientists realized it was practically impossible to achieve an LD 50 level, or deadly overdose, of the drug (PERFECT SAFETY!) - But, animal tests did not include tests looking at the effects during pregnancy

Milestone – the Thalidomide Disaster (cont. ) - On sale already in 1956 as prescription-free OTC drug for treating influenza, later as a sedative and hypnotic, was seen to be vastly safer than barbiturates - Following anecdotal reports of benefits in the treatment of nausea and vomiting in early pregnancy, it was heavily promoted for the relief of morning sickness in the first few months of pregnancy - In 1959 reports started of babies being born with phocomelia and other internal malformations - In 1961, a possible link between these serious birth defects and thalidomide was highlighted and the drug was withdrawn - Ironically, thalidomide was never approved by FDA due in part to concerns raised about potentially irreversible neuritis and the drug’s safety

Result of thalidomide disaster - the whole regulatory system was reshaped, - in the United States, The Drug Amendments Act of 1962 was passed by Congress requiring FDA to approve all new drug applications - for the first time, demanded that a new drug should be proven to be effective and safe - the FDA was also given the authority to require compliance with current Good Manufacturing Practices (GMP), to officially register drug establishments and implement other requirements

European medicines regulation history

The United Kingdom - Committee on the Safety of Drugs (CSD) started in 1963 - had no legal powers but worked effectively with the pharmaceutical industry to advise on clinical trials and the marketing of new medicines - In 1964, this committee also set up a voluntary system of adverse drug reaction reporting for drugs by means of replypaid yellow cards; this became known as the “Yellow Card Scheme”.

Other European Countries - healthcare regulations with main objective of keeping unsafe products out of marketplace - The Helsinki Declaration has been set (1964) to avoid unethical and unsafe clinical trials and have safe and proper treatment of human subjects. - Until Thalidomide tragedy, the drugs were being sold by notification to health authority and no safety, efficacy or quality data were required to be submitted prior marketing

Other European Countries (cont. ) - formation of European Economic Commission (EEC) in 1957 (currently European Union; EU) - For medicinal products, the first directive passed by EEC was "Council Directive 65/65/EEC of 26 January 1965 on the approximation of provisions laid down by law, regulation or administrative action relating to medicinal products - this mandated that no medicines can be marketed in European Communities until and unless it is not approved by at least one competent authority within Europe. - the objective was to have standard common marketing approval for medicines process within EEC

To further develop harmonization in European community. . . 1) Directive 75/318/EEC: on approximation of the laws of member states relating to analytical, pharmaco-toxicological and clinical standards and protocols in respect of the testing of proprietary medicinal products 2) Directive 75/319/EEC established also the Committee on Proprietary Medicinal Products (since 2004 Committee for Medicinal Products for Human Use (CHMP)) as an advisory committee to the European Commission and introduced the multistate procedure known today as the Mutual Recognition Procedure (MRP) 3) Directive 87/22/EEC introduced the concentration procedure which is now known as the Centralized Procedure (CP). These directives, and following council regulations, were the landmarks for starting harmonization inside the European Union with the final longstanding aim of creating a common market for medicines 4) The Council Regulation EEC/2309/93 established the European Medicines Evaluation Agency (EMEA; now EMA)

65/65/EEC The first European medicinal council directive, about detailed marketing authorisation requirements across European community and it was applicable not only to new products to be authorized but already approved marketed products as well. 75/318/EEC Specification of legal requirements relating to analytical, pharmaco-toxicological, and clinical documentation. 75/319/EEC Establishment of the Committee on Proprietary Medicinal Products (CPMP) and introduction of the Mutual Recognition Procedure (MRP). Introduction of the Centralized Procedure (CP). Definition of requirements for immunological products, i. e. vaccines, toxins, sera and allergens. 87/22/EEC 89/342/EEC 89/381/EEC 91/356/EEC 92/27/EEC 92/28/EEC 2001/18/EEC Regulation of medicinal products derived from human blood and blood plasma Definition of GMP requirements. Definition of prescription status. Definition of medicinal products labelling and package leaflet. Definition of medicinal products advertising. Regulation of use of genetically modified micro organisms and their deliberate release into environment. It was further amended by regulations (EC) 830/2003 - establishing a clear EU system for tracking and labelling genetically modified organisms. 2001/83/EEC Above all Council Directive was unified and replaced by one common directive as 2001/83/EEC. 2002/98/EC Definition of standards of quality and safety for the collection, testing, processing, storage and distribution of human blood and blood products. Definition of analytical, pharmaco-toxicological, clinical standards and protocol for medicinal products testing, increase of biological Medicinal Products safety, establishment of a new system that simplifies approval and subsequent change procedure for human plasma derived medicinal products as well as need for Vaccine Antigen Master File (VAMF). 2003/63/EC 2003/94/EC 2004/24/EC Establishment of further GMP principles and guidelines for Medicinal products and Investigational Medicinal Products (IMPs). Definition of requirements for Traditional Herbal Medicinal products, Definition of roles and responsibility of HPMC (Committee for Herbal Medicinal Product).

Regulatory bodies

Drugs are controlled by laws and regulations Laws regulate drugs in current use, poisons, unlawful drugs, and the development of new therapeutic agents Drugs must meet the requirements of purity, potency, and quality defined in established drug standards, which provide guidelines released by regulatory institutions Additionally, drugs are tested by their manufacturers and by governments inspections from regulatory institutions – in European Medicines Agency, EMA, www. ema. europa. eu; in USA, U. S. Food and Drug Administration, FDA, www. fda. gov – to ensure that they meet all the standards Regulatory bodies also carefully control the development of new agents, regulate the phases of testing and approving the release of new medicines

The United States and the European Union account for by far the most new drug introduction in the world However, drugs are regulated to one degree or another in most if not all countries • some of them accept the same application made in Europe or the United States, • some of them make their own determinations based wholly or in large part on the decisions of the US and EU • some accept these decisions outright, while others remain more independent, using the decisions as a point to consider but by no means regarding them as binding or automatically acceptable

The process of harmonization is under way to make the regulatory process more uniform, especially between the United States and Europe. Although there are differences in the details of the approval process and, indeed, sometimes in the results of the process, the requirements and expectations are much more uniform than they were in the mid to latter third of the twentieth century. While not official policy, the European and U. S. regulatory authorities in fact pay much attention to the activities and decisions of each other.

EMA • In Europe, the regulation process has changed dramatically since the inception of the EU, with new procedures replacing the previous country-by-country approval process. • Consequently, the prerequisites for approval have become uniform in the member states. • In Europe, drug approval is regulated by both centralized and decentralized processes aimed at producing more uniform practices across member states of the EU. • The European Medicines Agency (EMA) is an agency of the EU, located in London, and responsible for the scientific evaluation of medicines developed by pharmaceutical companies for use in the EU.

EMA: • provides independent, science-based recommendations on the quality, safety and efficacy of medicines, and on more general issues relevant to public and animal health that involve medicines, • applies efficient and transparent evaluation procedures to help bring new medicines to the market by means of a single, EU-wide marketing authorisation granted by the European Commission, • implements measures for continuously supervising the quality, safety and efficacy of authorised medicines to ensure that their benefits outweigh their risks,

EMA: • provides scientific advice and incentives to stimulate the development and improve the availability of innovative new medicines, • recommends safe limits for residues of veterinary medicines used in food-producing animals, for the establishment of maximum residue limits by the European Commission, • involves representatives of patients, healthcare professionals and other stakeholders in its work, to facilitate dialogue on issues of common interest,

EMA: • publishes impartial and comprehensible information about medicines and their use, • develops best practice for medicines evaluation and supervision in Europe, and contributes alongside the Member States and the European Commission to the harmonization of regulatory standards at the international level.

FDA In the United States, the U. S. Food and Drug Administration (FDA), a branch of the Department of the Health and Human Services, approves drugs. The FDA administers most of the federal laws concerned with the pharmaceutical industry and comprises several offices and centres which regulate following areas: I. Food, II. Drugs, III. Medical Devices, IV. Vaccines, Blood & Biologics, V. Animal & Veterinary, VI. Cosmetics, VII. Radiation-Emitting Products, VIII. Tobacco Products.

ŠÚKL (SIDC) In Slovakia, the State Institute for Drug Control (SIDC; Štátny ústav pre kontrolu liečiv, ŠÚKL) in Bratislava is the institution of the Ministry of Health of the Slovak Republic responsible for ensuring surveillance of the quality, efficacy and safety of medicinal products for human use and medical devices used in health care. SIDC has following responsibilities: • controls, standardizes and evaluates the process of assessing the quality, efficacy and safety of drugs, • supervises pharmacies, wholesalers, manufacturers of medicinal products and medical devices, • prepares assessment reports on medicinal products for human use,

ŠÚKL (SIDC): • issues marketing authorizations performs pharmacovigilance and vigilance in the field of medical devices, • performs withdrawal or suspension of marketing or marketing authorization of medicinal products or medical devices, • controls advertisement of medicinal products and infant milk formula, • performs inspections of Good Manufacturing Practice (GMP), Good Laboratory Practice (GLP), Good Clinical Practice (GCP), and Good Pharmacy Practice (GPP), • keeps database of registered drugs and medicinal devices, • acts in the field of training and education.

Role and core tasks of Medicine Agencies and European Medicine Agency Medicine agencies in the EU but also outside the EU have three main duties: 1) To protect public health by ensuring that only medicines with a favourable benefit to risk profile are licensed 2) To provide appropriate information on available medicines so that prescribers and patients can make informed decisions on their use 3) Not to impose unnecessary regulatory impediments that would prevent innovative medicines from being available to improve public health. The main mission of the regulation of medicines in Europe by European Medicines Agency is to foster scientific excellence in the evaluation and supervision of medicines, for the benefit of public health.

Thank you for your attention
- Slides: 30