Introduction basic chemical concepts Basic reading chapter 9

Introduction: basic chemical concepts Basic reading: chapter 9 of Jacob (1999) Advanced reading: chapter and 5. 3 of Brasseur and Jacob (2017)
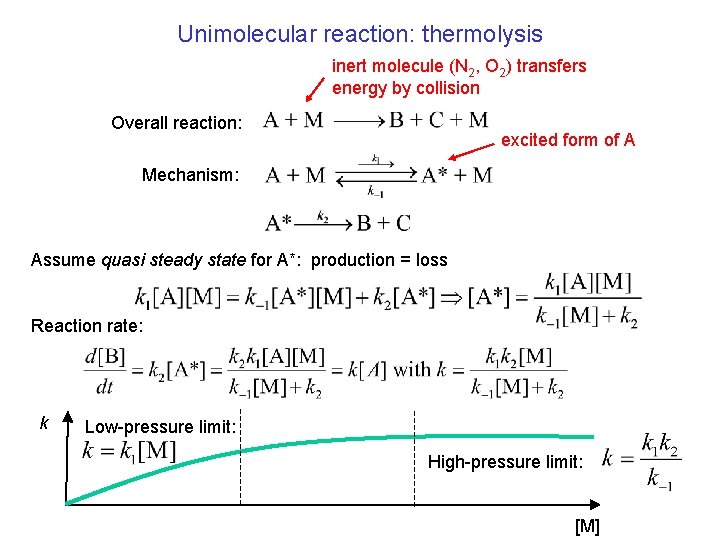
Unimolecular reaction: thermolysis inert molecule (N 2, O 2) transfers energy by collision Overall reaction: excited form of A Mechanism: Assume quasi steady state for A*: production = loss Reaction rate: k Low-pressure limit: High-pressure limit: [M]

Unimolecular reaction: photolysis k is the photolysis rate constant (also called photolysis frequency) wavelength [cm] radiation flux [photons cm-2 s-1] quantum yield: probability that photon absorption causes photolysis absorption cross-section [cm 2 molecule-1] Probability of absorption for incoming photons = σ/A photon is not absorbed Absorption cross-section s photon is absorbed Molecular cross-section A
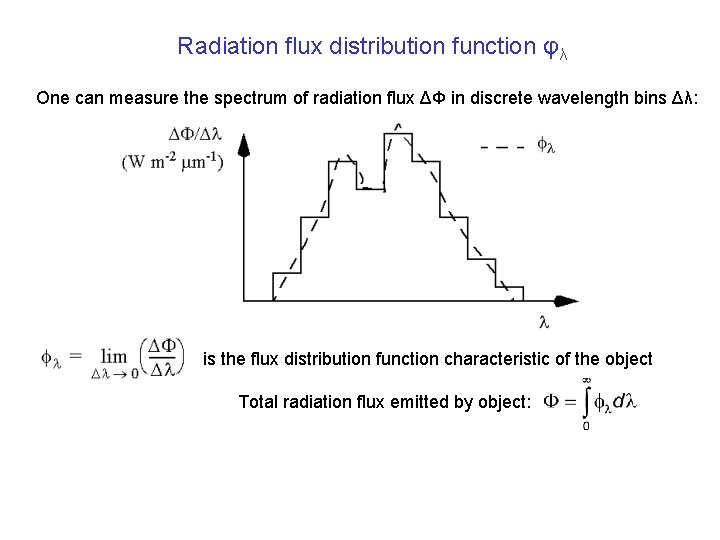
Radiation flux distribution function φλ One can measure the spectrum of radiation flux ΔΦ in discrete wavelength bins Δλ: is the flux distribution function characteristic of the object Total radiation flux emitted by object:
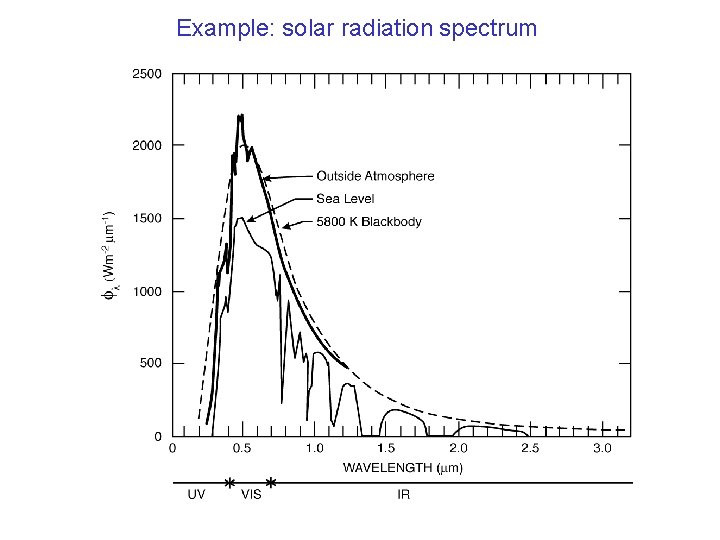
Example: solar radiation spectrum
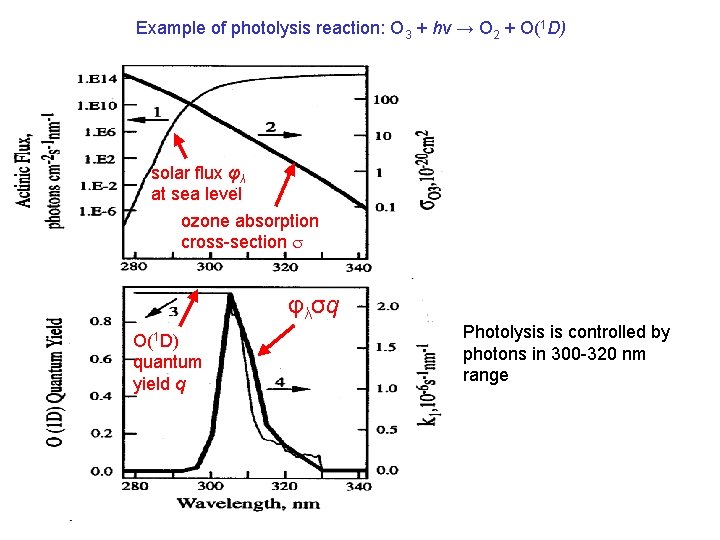
Example of photolysis reaction: O 3 + hv → O 2 + O(1 D) solar flux φλ at sea level ozone absorption cross-section s φλσq O(1 D) quantum yield q Photolysis is controlled by photons in 300 -320 nm range
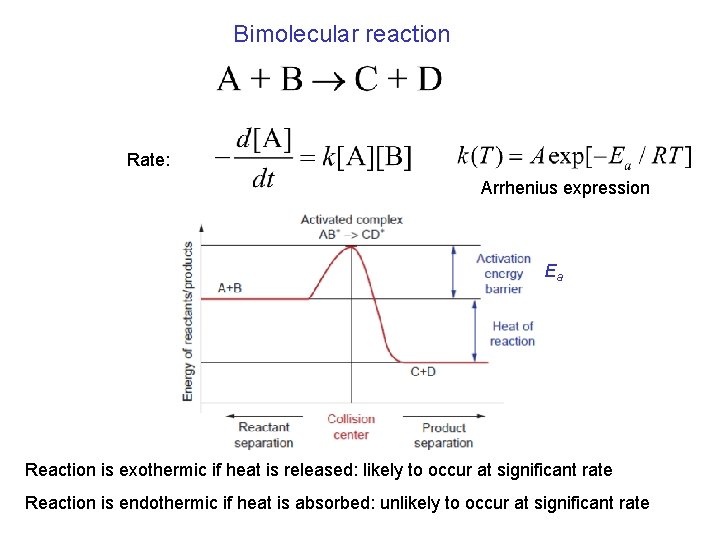
Bimolecular reaction Rate: Arrhenius expression Ea Reaction is exothermic if heat is released: likely to occur at significant rate Reaction is endothermic if heat is absorbed: unlikely to occur at significant rate

Three-body reaction Overall reaction: Mechanism: From steady state for AB*, general solution: ko Low-pressure limit: High-pressure limit: k∞ General formulation: with F is a correction factor for the transition regime
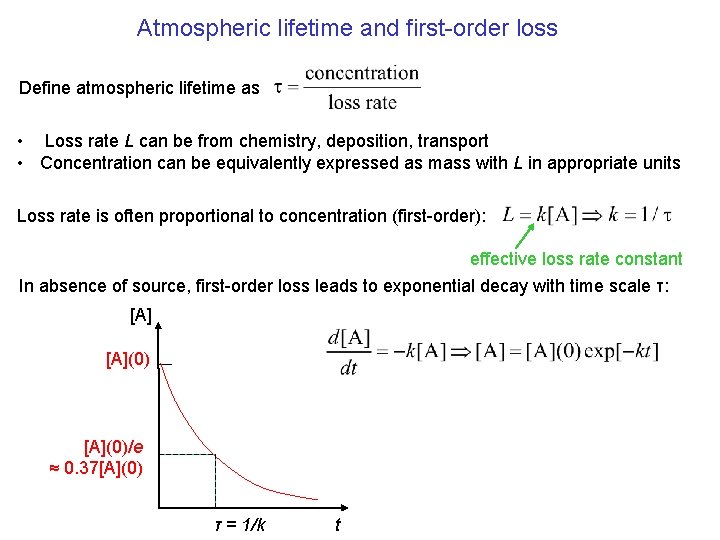
Atmospheric lifetime and first-order loss Define atmospheric lifetime as • Loss rate L can be from chemistry, deposition, transport • Concentration can be equivalently expressed as mass with L in appropriate units Loss rate is often proportional to concentration (first-order): effective loss rate constant In absence of source, first-order loss leads to exponential decay with time scale τ: [A](0)/e ≈ 0. 37[A](0) τ = 1/k t

Redox reactions oxidant I want electrons! + reductant → products I want to get rid of electrons! Let’s do it! reductant gets oxidized oxidant gets reduced An atom minimizes energy by filling lowest-energy orbitals in its outermost (valence) electron shell: this is done by acquiring or donating electrons through bonding First valence shell has 2 electrons; second has 8; third has 18 (but 8 low-energy), … In periodic table, atomic number gives number of electrons in neutral/unbound atom: this corresponds to oxidation state zero (0) for that element. Oxidation state becomes negative if atom acquires electrons, positive if it donates. Some handy rules: • A neutral molecule has total oxidation number 0 • Bound oxygen has oxidation state -2 (except -1 in peroxides) • Bound hydrogen has oxidation state +1
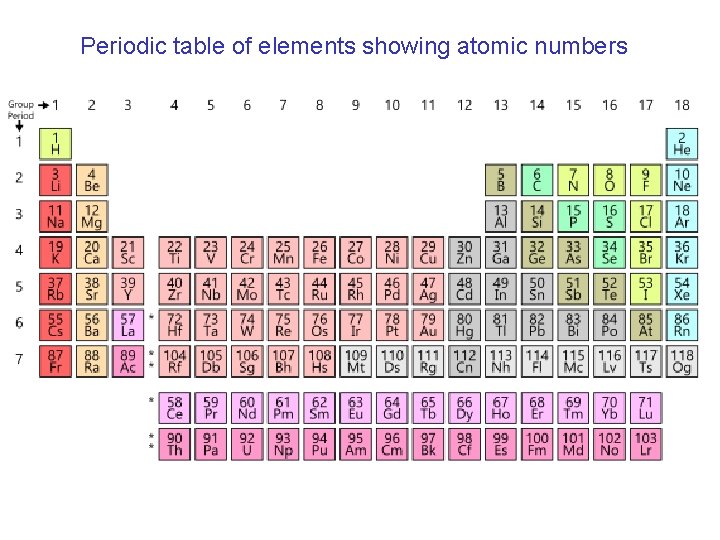
Periodic table of elements showing atomic numbers
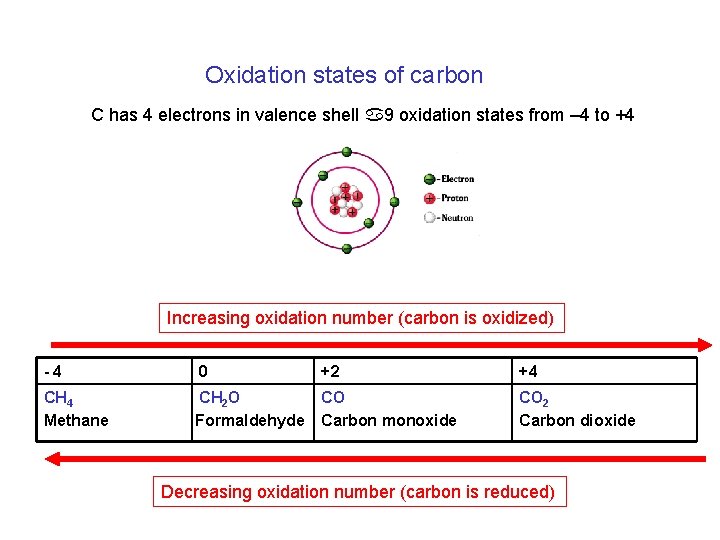
Oxidation states of carbon C has 4 electrons in valence shell a 9 oxidation states from – 4 to +4 Increasing oxidation number (carbon is oxidized) -4 0 +2 +4 CH 4 Methane CH 2 O Formaldehyde CO Carbon monoxide CO 2 Carbon dioxide Decreasing oxidation number (carbon is reduced)
- Slides: 12