Introduction 2007 HIV Diagnostics Conference Bernard M Branson

Introduction: 2007 HIV Diagnostics Conference Bernard M. Branson, M. D. Associate Director for Laboratory Diagnostics Divisions of HIV/AIDS Prevention National Center for HIVAIDS, Viral Hepatitis, STD, and TB Prevention Centers for Disease Control and Prevention The findings and conclusions in this presentation are those of the author and do not necessarily represent the views of the Centers for Disease Control and Prevention
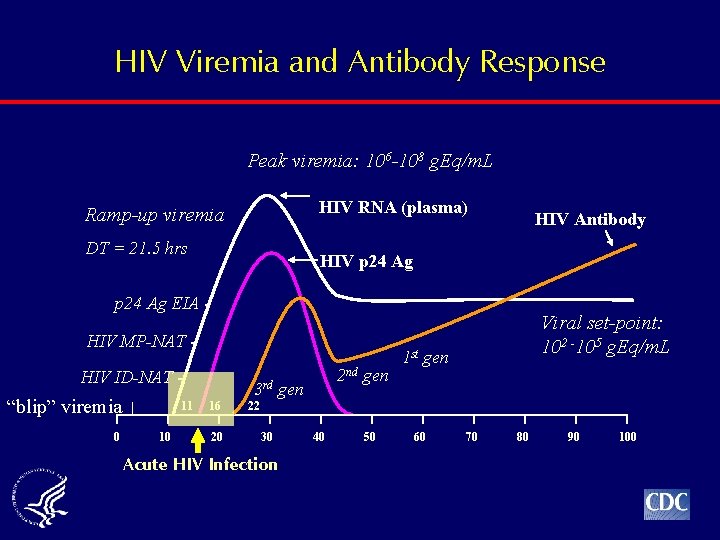
HIV Viremia and Antibody Response Peak viremia: 106 -108 g. Eq/m. L HIV RNA (plasma) Ramp-up viremia DT = 21. 5 hrs HIV Antibody HIV p 24 Ag EIA HIV MP-NAT HIV ID-NAT - “blip” viremia 0 11 10 2 nd gen 3 rd gen 16 20 Viral set-point: 102 -105 g. Eq/m. L 1 st gen 22 30 Acute HIV Infection 40 50 60 70 80 90 100

1989: State of the Art Western blot EIA


Organization Criteria APHL/CDC Any two: p 24, gp 41, gp 120/160 ARC Three or more bands, one from each gene product group: gag, pol, env Two or more bands: p 24 or p 31 and gp 41 or gp 120/160 CRSS WHO Two env bands with or without gag or pol Indeterminate: Presence of any bands

Diagnostic Algorithm: 1989 n The Public Health Service recommends that no positive test results be given to clients/patients until a screening test has been repeatedly reactive (i. e. , greater than or equal to two tests) on the same specimen and a supplemental, more specific test such as the Western blot has been used to validate those results

1989 State of the Art

WHO HIV Diagnostic Testing: Strategy III Asymptomatic; prevalence < 10% A 1+ A 1 - Negative A 2 A 1+A 2 - A 1+A 2+ Positive A 3 A 1+A 2 -A 3+ A 1+A 2 -A 3 - Positive Indeterminate
SUDS: Single Use Diagnostic System for HIV-1
Blue color with reactive HIV test


Recommendation …and a Promise n Health-care providers should provide preliminary positive test results before confirmatory results are available in situations where tested persons benefit. n When additional rapid tests become available for use in the United States, the PHS will re-evaluate algorithms using specific combinations of two or more rapid tests for screening and confirming HIV infection.
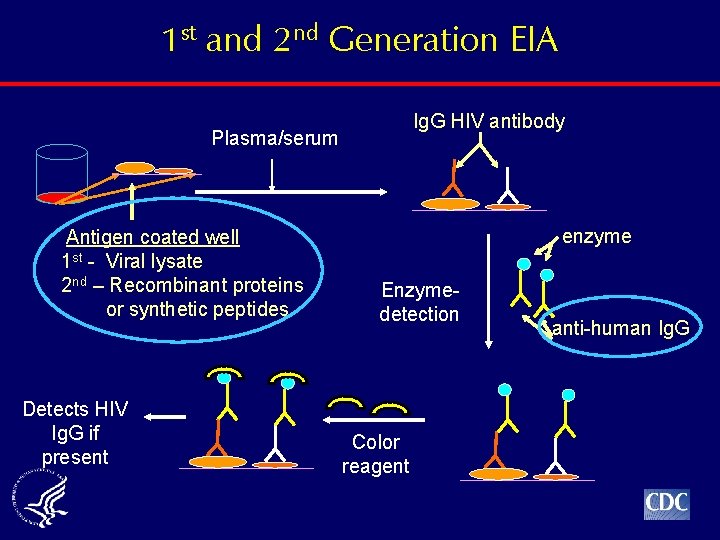
1 st and 2 nd Generation EIA Plasma/serum (1 h/37 o C) Ig. G HIV antibody Plasma/serum Antigen coated well 1 st - Viral lysate 2 nd – Recombinant proteins or synthetic peptides Detects HIV Ig. G if present enzyme Enzymedetection Color reagent anti-human Ig. G
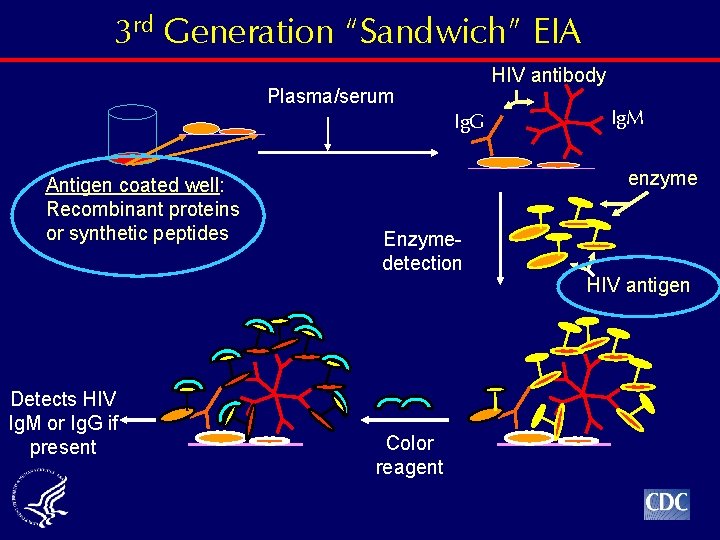
3 rd Generation “Sandwich” EIA HIV antibody Plasma/serum Ig. G Antigen coated well: Recombinant proteins or synthetic peptides Detects HIV Ig. M or Ig. G if present Ig. M enzyme Enzymedetection Color reagent HIV antigen
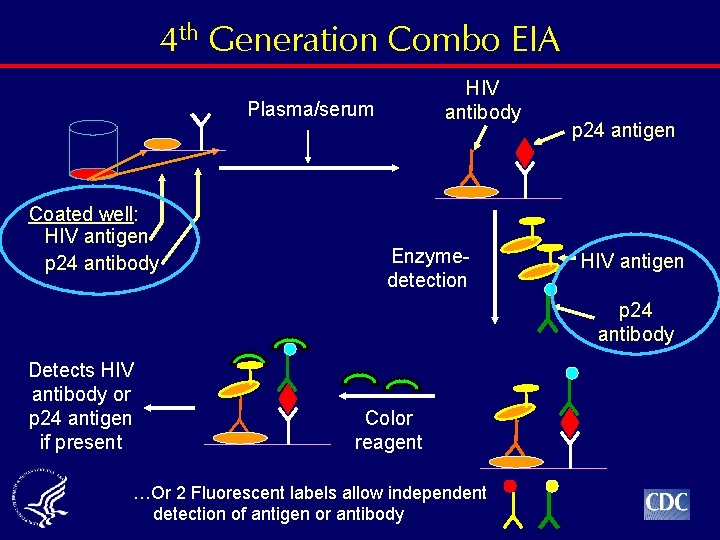
4 th Generation Combo EIA HIV antibody Plasma/serum Coated well: HIV antigen p 24 antibody Enzymedetection p 24 antigen HIV antigen p 24 antibody Detects HIV antibody or p 24 antigen if present Color reagent …Or 2 Fluorescent labels allow independent detection of antigen or antibody
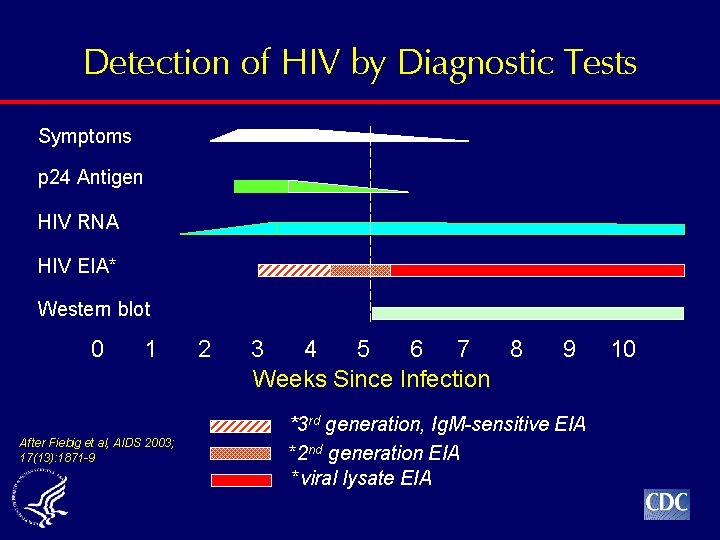
Detection of HIV by Diagnostic Tests Symptoms p 24 Antigen HIV RNA HIV EIA* Western blot 0 1 After Fiebig et al, AIDS 2003; 17(13): 1871 -9 2 3 4 5 6 7 8 Weeks Since Infection 9 *3 rd generation, Ig. M-sensitive EIA *2 nd generation EIA *viral lysate EIA 10

EIAs Used by Public Health Labs FDA approval date % used by PHL labs, 2004 Vironostika HIV-1 Microelisa 1987 63% Abbott HIVAB HIV-1/2 1992 20% Genetic Systems r. LAV 1998 20% Gen Sys HIV-1/HIV-2 2000 18% Gen Sys HIV-1/2 Plus O 2003 10% Vironostika HIV-1 Plus O 2003 2% viral lysate EIA

EIAs Used by Public Health Labs FDA approval date % used by PHL labs, 2004 Vironostika HIV-1 Microelisa 1987 63% Abbott HIVAB HIV-1/2 1992 20% Genetic Systems r. LAV 1998 20% Gen Sys HIV-1/HIV-2 2000 18% Gen Sys HIV-1/2 Plus O 2003 10% Siemens 1/O/2 e. HIV 2006 viral lysate EIA 2 nd generation EIA 3 rd generation, Ig. M-sensitive EIA

Changes in Availability of EIAs FDA approval date % used by PHL labs, 2004 Vironostika HIV-1 Microelisa 1987 63% Abbott HIVAB HIV-1/2 1992 20% Genetic Systems r. LAV 1998 20% Gen Sys HIV-1/HIV-2 2000 18% Gen Sys HIV-1/2 Plus O 2003 10% Bayer 1/O/2 e. HIV 2006 viral lysate EIA 2 nd generation EIA 3 rd generation, Ig. M-sensitive EIA

Changes in Availability of EIAs FDA approval date % used by PHL labs, 2004 Vironostika HIV-1 Microelisa 1987 63% Abbott HIVAB HIV-1/2 1992 20% Genetic Systems r. LAV 1998 20% Gen Sys HIV-1/HIV-2 2000 18% Gen Sys HIV-1/2 Plus O 2003 10% Siemens 1/O/2 e. HIV 2006 viral lysate EIA 2 nd generation EIA 3 rd generation, Ig. M-sensitive EIA

2007 State of the Art
Uni-Gold Recombigen Clearview Complete HIV 1/2 Multispot HIV-1/HIV-2 Reveal G 3 Clearview HIV ½ Stat Pak Ora. Quick Advance
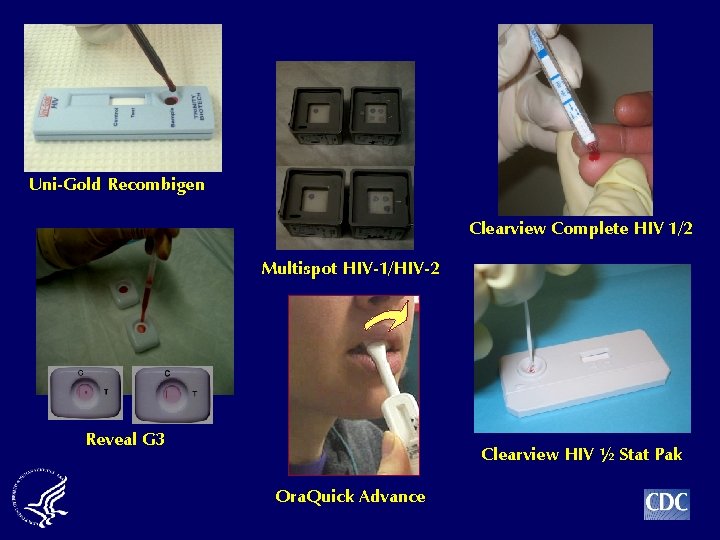
Uni-Gold Recombigen Clearview Complete HIV 1/2 Multispot HIV-1/HIV-2 Reveal G 3 Clearview HIV ½ Stat Pak Ora. Quick Advance

Reactive Control Recombinant HIV-1 Peptide HIV-2 Peptide HIV-1 Negative HIV-1 & HIV-2 Positive

The ADVIA® Centaur™ Random Access HIV 1/O/2 Enhanced (EHIV)

HIV 1/O/2 Enhanced (EHIV) n Multiple epitopes n 3 rd generation “sandwich” format n Random access EIA n FDA-approved July 2006

On-board Refrigeration of 30 Different Assays

STAT sample requests without pausing Results in ~60 minutes

APTIMA HIV-1 RNA Qualitative Assay Gen-Probe Incorporated Ø Target capture specimen processing Ø Transcription-Mediated Amplification (TMA) Ø Dual Kinetic Assay ØFDA approved September 2006

Qualitative RNA Assay: Intended Use n Aid to HIV-1 diagnosis n Diagnosis of acute HIV-1 infection in antibodynegative persons n Confirmation of HIV-1 infection in antibodypositive persons when it is reactive

Clinical Syndrome of Acute HIV n 40 -90% develop symptoms of Acute HIV n 50%-90% with symptoms seek medical care n Of those diagnosed with Acute HIV, 50% of patients seen at least 3 times before diagnosis - Kahn et al, NEJM 1998 - Weintrob et al, Arch Int Med 2003
Pooled RNA Screening for Early HIV Infection
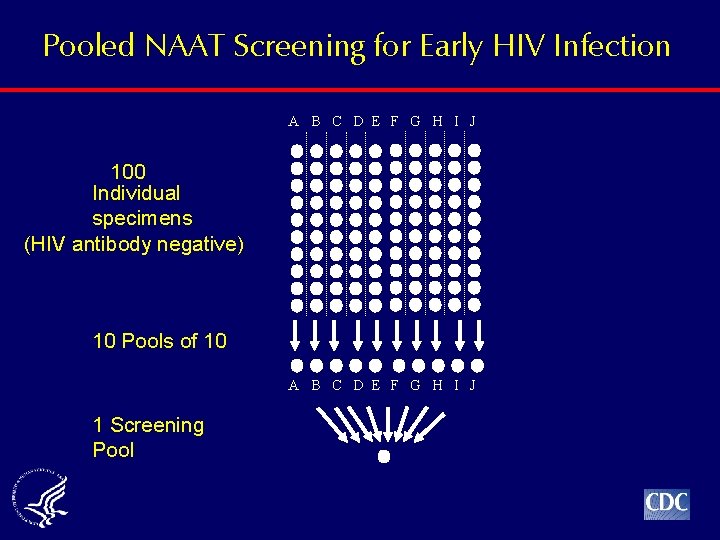
Pooled NAAT Screening for Early HIV Infection A B C D E F G H I J 100 Individual specimens (HIV antibody negative) 10 Pools of 10 A B C D E F G H I J 1 Screening Pool

Resolution Testing A Individual NAAT testing on 10 specimens 10 pools of 10 tested with NAAT Screening Pools of 100 specimens tested with NAAT
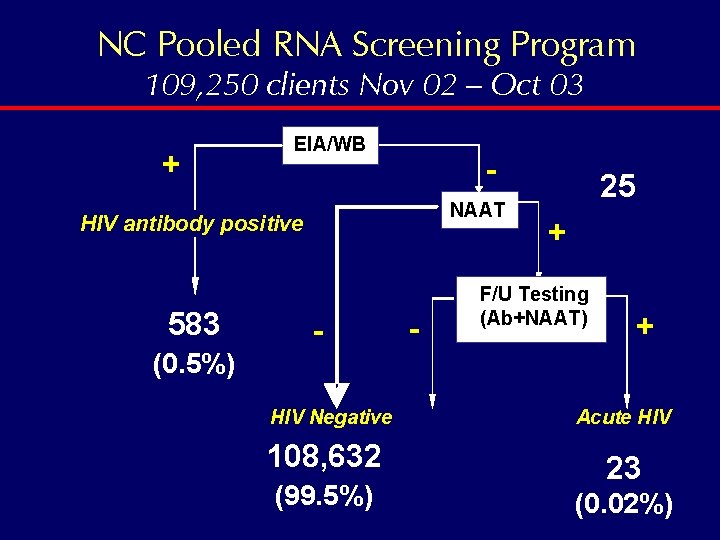
NC Pooled RNA Screening Program 109, 250 clients Nov 02 – Oct 03 + EIA/WB NAAT HIV antibody positive 583 - - 25 + F/U Testing (Ab+NAAT) + (0. 5%) HIV Negative 108, 632 (99. 5%) Acute HIV 23 (0. 02%)

3 -Test Algorithm: Antenatal Women HIV- Determine n=1179 HIV+ Confirm w/Genie II n=177 (15%) n=999 (85%) HIV- HIV+ Capillus n=8 (4. 5%) HIVHIV+ n=8 (100%) Koblavi-Deme et al, J Clin Micro 2001 n=169 (95. 5%) n=0

3 -Test Algorithm, Uganda EIA/WB Positive EIA/WB Negative Rapid Test Positive 295 166 129 Rapid Test Negative 1220 4 1218 (Determine – Unigold – Stat Pak) Sensitivity: 97. 6% Specificity: 90. 4% - Gray et al, BMJ 2007
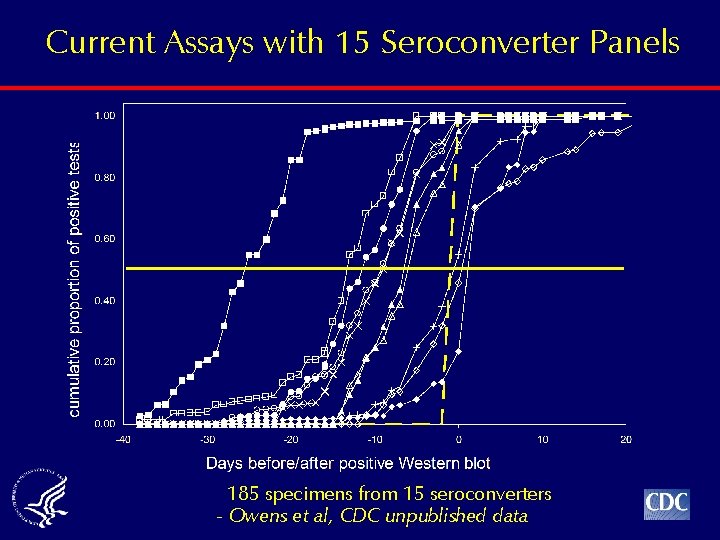
Current Assays with 15 Seroconverter Panels 185 specimens from 15 seroconverters - Owens et al, CDC unpublished data

25 20 15 10 5 Ora. Quick WB positive Uni-Gold Vironostika HIV-1 (1 st) Multi-Spot GS r. Lav (2 nd) Abbott HIVAB ½ (3 rd) Reveal G 2 WB Indeterminate Procleix RNA Bio. Rad HIV-1/2+O (3 rd) Current Assays with 15 Seroconverter Panels 0 Days before Western blot positive when 50% of Specimens Reactive 185 specimens from 15 seroconverters - Owens et al, CDC unpublished data

Dilemmas n Screening in low prevalence n False-positive Western blots? n What metric?
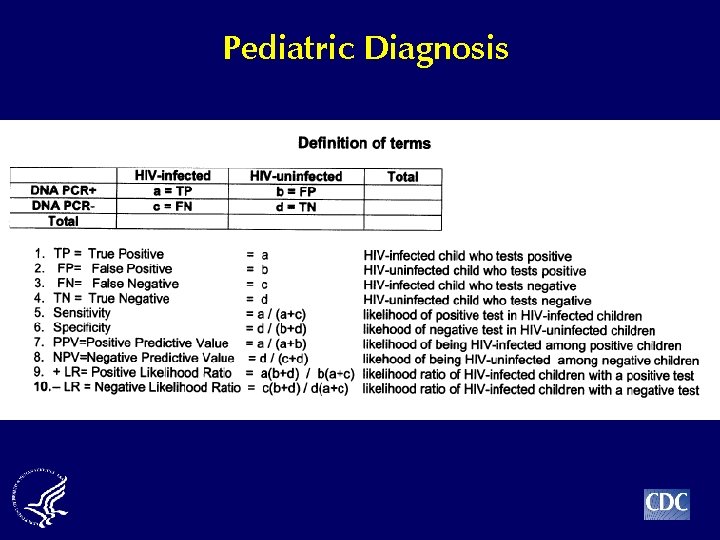
Pediatric Diagnosis
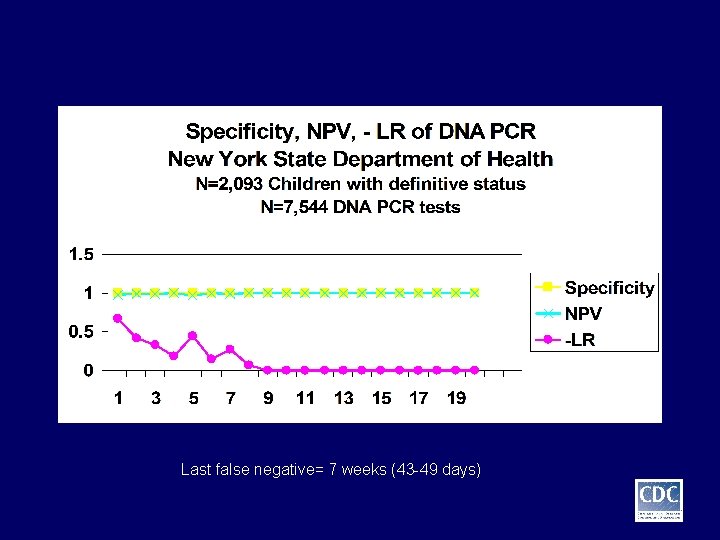
Last false negative= 7 weeks (43 -49 days)
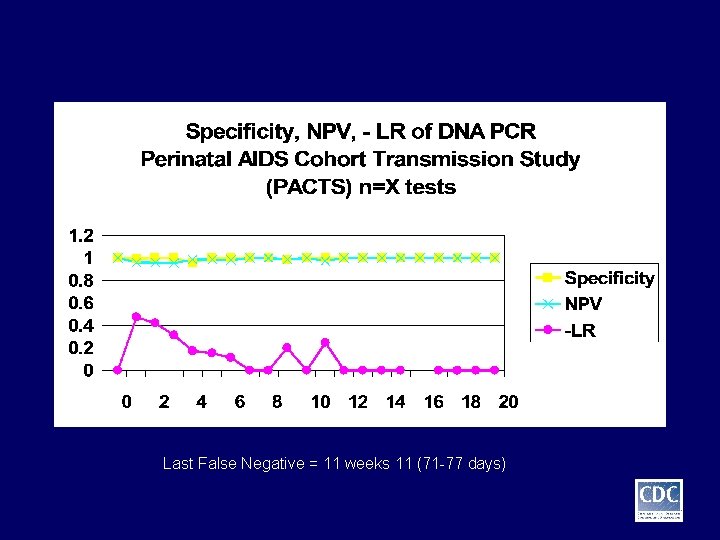
Last False Negative = 11 weeks 11 (71 -77 days)

Interpreting Test Results Negative likelihood ratio: likelihood of negative test when disease is present likelihood of negative test when disease is absent - Ratio of false negative rate specificity { true negative rate}

Interpreting Test Results Negative likelihood ratio: - Does not depend on prevalence - Ratio < 0. 1 = convincing diagnostic evidence Ratio < 0. 2 = strong diagnostic evidence - Can be used with continuous variables (viral load)
- Slides: 45