Introducing Substantial Equivalence to the Predicate Device July

Introducing Substantial Equivalence to the Predicate Device July 16, 2019 Korea Medical Device Industry Association Legal Committee Vice Chairperson Young Soo Sul

Table of Contents 1. What is “Substantial Equivalence”? 2. Terminology and Definition 3. Regulations Related to Substantial Equivalent Devices 4. Equivalence Determination Criteria 5. Scope of Submitting Data Including Technical Document 6. Substantial Equivalence Comparison Table 7. Changes in Substantial Equivalence Review Process 8. Major Changes 9. Concerns and Anticipation of Changes 2

What is “Substantial Equivalence”? u It has the same structure and principles so that its safety and efficacy data can be substituted with the previous technology. u “Substantial equivalence review” is a pre-marketing submission to seek approval for a device with demonstrated data and safety analysis data when it has the same intended use, mechanism of action, and guaranteed safety and efficacy in a technology with sufficient safety and efficiency data. u If substantial equivalence, including structure, principles, performance, intended use, and indications for use, to the predicate device is demonstrated, submission of some approval/review data, such as clinical study data, is waived in order to alleviate industrial and administrative burdens. u Substantial equivalence is also implemented in regulatory-advanced countries, such as the USA and European Union. 3

Terminology and Definition u “Same class” refers to a device that has the same intended use, indication for use, manufacturing method, and raw materials among the devices with the same manufacturing country, manufacturer, and item name. It also refers to a class consisting of different colors or sizes, or serial models with modified/added components. u “New device” refers to a device that does not have the same intended use, mechanism of action, or raw material with the predicate device. u “Modified device” refers to a device with a change in performance, test specification, or indication for use, while its intended use, mechanism of action, and raw material (only applicable for medical products) are the same as the predicate device. In-vitro diagnostic medical device refers to a device that has the same intended use and mechanism actions as the predicate device, but with a different raw material or performance. u “Equivalent device” refers to a device with the same intended use, mechanism of action, raw material (only applicable for medical product), performance, test specification, and indications for use as the predicate device. In-vitro diagnostic medical device refers to a device with the same intended use, mechanism of action, raw material, and performance as the predicate device. Source: Article 2 of Regulations on Approval, Notification, and Review of the Medical Device 4

Regulations Related to Substantial Equivalent Devices u Article 3 (Application for Approval, Certification, and Notification of the Medical Device) - If a device is applicable to the same class, use one product approval/certification/ notification for the application. - For the class 2 device, announce the intended use, mechanism of action, performance, test specification, and indication for use of the device that is approved/certified at least three times as a predicate device – homepage - Submission of certification for safety and performance should demonstrate the compatibility with the predicate device. - If you want to get confirmation of substantial equivalence to a predicate device, the data demonstrating the following should be attached. ① Medical device with the same intended use, mechanism of action, raw material, performance, test specification, and indications for use; ② Medical device manufactured by the same manufacturer; and ③ Medical device manufactured by the contracted manufacturer in the manner of design/development/produce by the manufacturing sponsor Source: KFDA Notification No. 2019 -13, Regulations on Approval, Notification, and Review of the Medical Device 5

Regulations Related to Substantial Equivalent Devices u Article 23 (Subject to Technical Document Review, Etc. ) - Medical device seeking approval/certification for manufacture/import - For the class 2 medical device, categorized review based on “equivalence criteria” using “substantial equivalence comparison table” into the new device, modified device, and equivalent device will be conducted. - Exemption from technical document review ① Medical device for export; ② Announced equivalence device; ③ Medical device confirmed as the equivalent device ④ Medical device with minor modification Source: KFDA Notification No. 2019 -13, Regulations on Approval, Notification, and Review of the Medical Device 6

Regulations Related to Substantial Equivalent Devices u Article 28 (Exemption from Review Data) - Class 2 medical device that is categorized as a new device, a modified device, or an equivalent device is exempted from submitting the following data according to the “scope of submitting data such as technical document, etc. ” ① Data on intended use, mechanism of action, development details, and foreign usage status; and ② Only for data confirming performance and safety of the device, rationale for test specification except for certification of analysis, and measurement values - For the class 3 or 4 devices, only the development details, clinical trials, and foreign usage status may be waived. - If biological stability is confirmed according to the notification or announced specification by the Minister of Ministry of Food and Drug Safety, the safety data may be waived. - Data submission is waived when the study itself is theoretically/technologically impossible or meaningless. Source: KFDA Notification No. 2019 -13, Regulations on Approval, Notification, and Review of the Medical Device 7

Regulations Related to Substantial Equivalent Devices u Article 34 (Exemption from Review Data, Etc. , of In-Vitro Diagnostic Medical Device) - For the class 2 in-vitro diagnostic medical device, categorized review based on “equivalence criteria” using “substantial equivalence comparison table for in-vitro diagnostic medical device” into the new device, modified device, and equivalent device will be conducted. - Class 2 in-vitro diagnostic medical device that is categorized as new device, modified device, or equivalent device is exempted from submitting the following data according to the “scope of submitting data such as technical document, etc. ” ① Data on developmental details, measuring principles, and intended use; and ② Data confirming the performance of the device - For the class 3 or 4 devices, the following data according to the “scope of data submission such as technical document, etc. ” may be exempted from submission. ① For Class 3: Data on developmental details, measuring principles, and clinical performance ② For Class 4: Data on developmental details and measuring principles Source: KFDA Notification No. 2019 -13, Regulations on Approval, Notification, and Review of the Medical Device 8
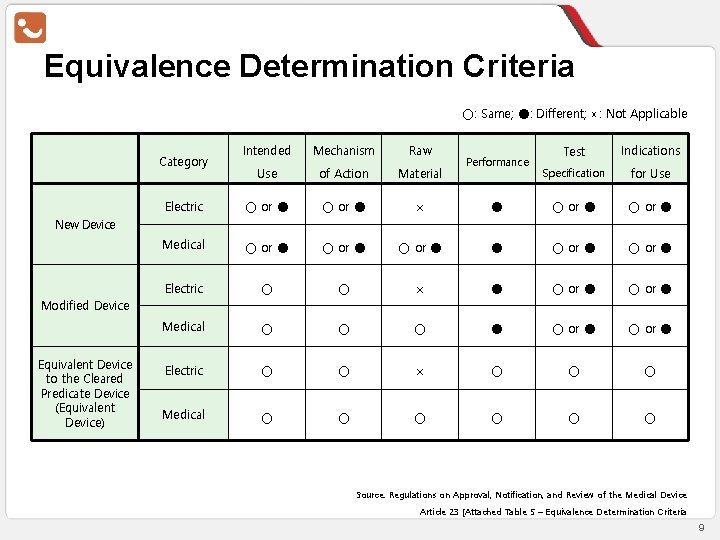
Equivalence Determination Criteria ○: Same; ●: Different; ×: Not Applicable Intended Mechanism Raw Test Indications Use of Action Material Specification for Use Electric ○ or ● × ● ○ or ● Medical ○ or ● ● ○ or ● Electric ○ ○ × ● ○ or ● Medical ○ ○ ○ ● ○ or ● Electric ○ ○ × ○ ○ ○ Medical ○ ○ ○ Category Performance New Device Modified Device Equivalent Device to the Cleared Predicate Device (Equivalent Device) Source: Regulations on Approval, Notification, and Review of the Medical Device Article 23 [Attached Table 5 – Equivalence Determination Criteria 9
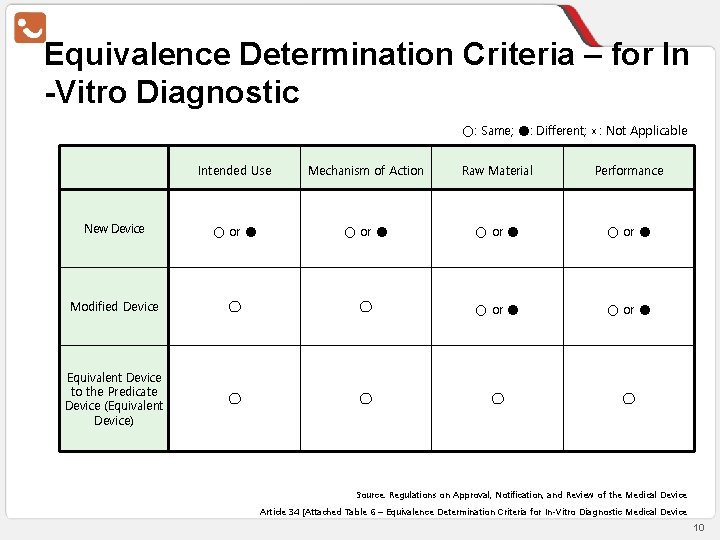
Equivalence Determination Criteria – for In -Vitro Diagnostic ○: Same; ●: Different; ×: Not Applicable Intended Use Mechanism of Action Raw Material Performance New Device ○ or ● Modified Device ◯ ◯ ○ or ● Equivalent Device to the Predicate Device (Equivalent Device) ◯ ◯ Source: Regulations on Approval, Notification, and Review of the Medical Device Article 34 [Attached Table 6 – Equivalence Determination Criteria for In-Vitro Diagnostic Medical Device 10
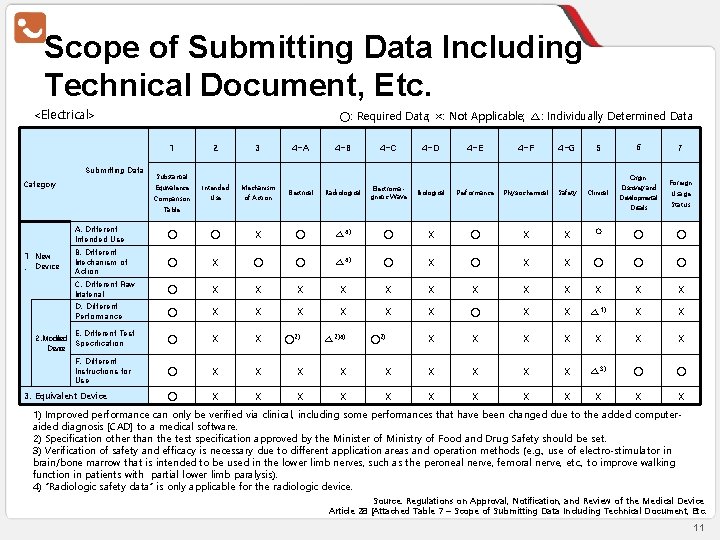
Scope of Submitting Data Including Technical Document, Etc. <Electrical> ○: Required Data; ×: Not Applicable; △: Individually Determined Data 1 Submitting Data Category 2 3 4 -A 4 -B 4 -C 4 -D 4 -E 4 -F 4 -G 5 6 Intended Use Mechanism of Action Electrical Radiological Electromagnetic Wave Biological Performance Physiochemical Safety Clinical Origin∙ Discovery and Developmental Details Substantial Equivalence Comparison Table 1 New. Device A. Different Intended Use ○ ○ X ○ △ 4) ○ X X B. Different Mechanism of Action ○ X ○ ○ △ 4) ○ X C. Different Raw Material ○ X X X X D. Different Performance ○ X X X X ○ X X E. Different Test 2. Modified Specification Device F. Different Instructions for Use 3. Equivalent Device ○2) △ 2)4) 7 Foreign Usage Status ○ ○ ○ X X X ○ X X △ 1) X X X X △ 3) ○ ○ X X X X ○2) 1) Improved performance can only be verified via clinical, including some performances that have been changed due to the added computeraided diagnosis [CAD] to a medical software. 2) Specification other than the test specification approved by the Minister of Ministry of Food and Drug Safety should be set. 3) Verification of safety and efficacy is necessary due to different application areas and operation methods (e. g. , use of electro-stimulator in brain/bone marrow that is intended to be used in the lower limb nerves, such as the peroneal nerve, femoral nerve, etc. , to improve walking function in patients with partial lower limb paralysis). 4) “Radiologic safety data” is only applicable for the radiologic device. Source: Regulations on Approval, Notification, and Review of the Medical Device Article 28 [Attached Table 7 – Scope of Submitting Data Including Technical Document, Etc. 11
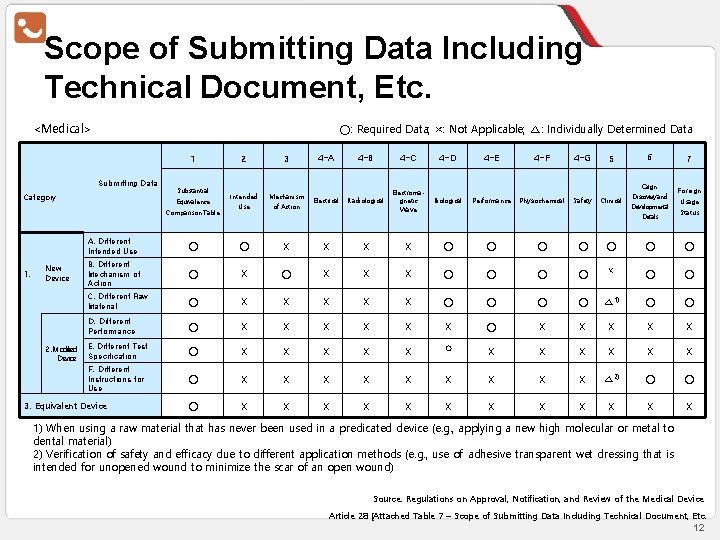
Scope of Submitting Data Including Technical Document, Etc. <Medical> ○: Required Data; ×: Not Applicable; △: Individually Determined Data 1 Submitting Data Category Substantial Equivalence Comparison Table 1. New Device 2. Modified Device 2 3 4 -A 4 -B 4 -C 4 -D 4 -E 4 -F 4 -G 5 6 Intended Use Mechanism of Action Electrical Radiological Electromagnetic Wave Biological Performance Physiochemical Safety Clinical Origin∙ Discovery and Developmental Details 7 Foreign Usage Status A. Different Intended Use ○ ○ X X ○ ○ ○ ○ B. Different Mechanism of Action ○ X X X ○ ○ C. Different Raw Material ○ X X X ○ ○ △ 1) ○ ○ D. Different Performance ○ X X X E. Different Test Specification ○ X X X F. Different Instructions for Use ○ X X X X X △ 2) ○ ○ ○ X X X 3. Equivalent Device ○ 1) When using a raw material that has never been used in a predicated device (e. g. , applying a new high molecular or metal to dental material) 2) Verification of safety and efficacy due to different application methods (e. g. , use of adhesive transparent wet dressing that is intended for unopened wound to minimize the scar of an open wound) Source: Regulations on Approval, Notification, and Review of the Medical Device Article 28 [Attached Table 7 – Scope of Submitting Data Including Technical Document, Etc. 12
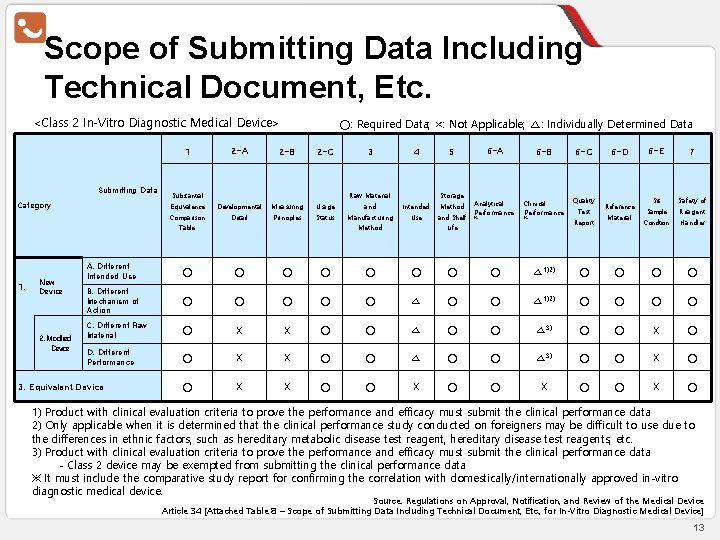
Scope of Submitting Data Including Technical Document, Etc. <Class 2 In-Vitro Diagnostic Medical Device> 2 -A 2 -B 2 -C Equivalence Developmental Measuring Usage Comparison Detail Principles Status 1 Submitting Data Category ○: Required Data; ×: Not Applicable; △: Individually Determined Data Substantial 1. 2. Modified Device 4 5 and Intended Method Manufacturing Use and Shelf Raw Material Table New Device 3 Storage Method 6 -A 6 -B 6 -C Analytical Performance Clinical Performance Quality ※ ※ Test Report Life 6 -D Reference Material 6 -E 7 Set Safety of Sample Reagent Condition Handler A. Different Intended Use ○ ○ ○ ○ △ 1)2) ○ ○ B. Different Mechanism of Action ○ ○ ○ △ 1)2) ○ ○ C. Different Raw Material ○ X X ○ ○ △ 3) ○ ○ X ○ D. Different Performance ○ X X ○ ○ △ 3) ○ ○ X X ○ ○ X ○ 3. Equivalent Device 1) Product with clinical evaluation criteria to prove the performance and efficacy must submit the clinical performance data 2) Only applicable when it is determined that the clinical performance study conducted on foreigners may be difficult to use due to the differences in ethnic factors, such as hereditary metabolic disease test reagent, hereditary disease test reagents, etc. 3) Product with clinical evaluation criteria to prove the performance and efficacy must submit the clinical performance data - Class 2 device may be exempted from submitting the clinical performance data ※ It must include the comparative study report for confirming the correlation with domestically/internationally approved in-vitro diagnostic medical device. Source: Regulations on Approval, Notification, and Review of the Medical Device Article 34 [Attached Table 8 – Scope of Submitting Data Including Technical Document, Etc. , for In-Vitro Diagnostic Medical Device] 13
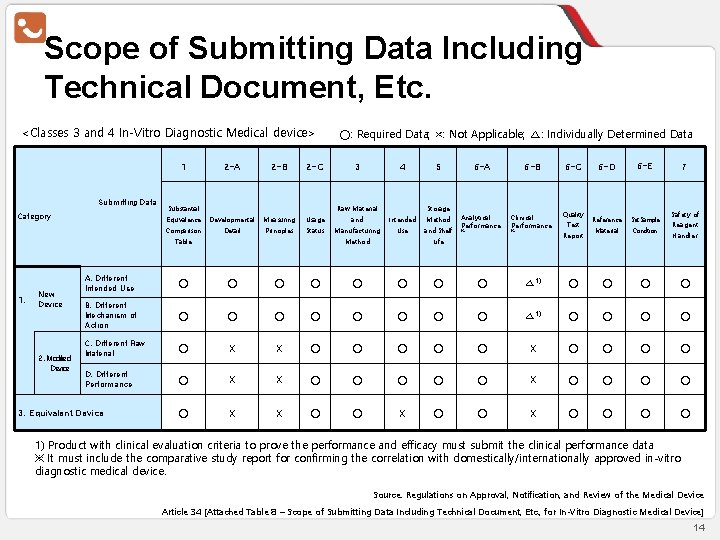
Scope of Submitting Data Including Technical Document, Etc. <Classes 3 and 4 In-Vitro Diagnostic Medical device> 2 -A 2 -B 2 -C Equivalence Developmental Measuring Usage Comparison Detail Principles Status 1 Submitting Data Category Substantial 1. 2. Modified Device 3 4 5 and Intended Method Manufacturing Use and Shelf Raw Material Table New Device ○: Required Data; ×: Not Applicable; △: Individually Determined Data Storage Method 6 -A 6 -B 6 -C Analytical Performance Clinical Performance Quality ※ ※ Test Report Life 6 -D 6 -E Reference Set Sample Material Condition 7 Safety of Reagent Handler A. Different Intended Use ○ ○ ○ ○ △ 1) ○ ○ B. Different Mechanism of Action ○ ○ ○ ○ △ 1) ○ ○ C. Different Raw Material ○ X X ○ ○ ○ X ○ ○ D. Different Performance ○ X X ○ ○ ○ ○ ○ X X ○ ○ 3. Equivalent Device 1) Product with clinical evaluation criteria to prove the performance and efficacy must submit the clinical performance data ※ It must include the comparative study report for confirming the correlation with domestically/internationally approved in-vitro diagnostic medical device. Source: Regulations on Approval, Notification, and Review of the Medical Device Article 34 [Attached Table 8 – Scope of Submitting Data Including Technical Document, Etc. , for In-Vitro Diagnostic Medical Device] 14
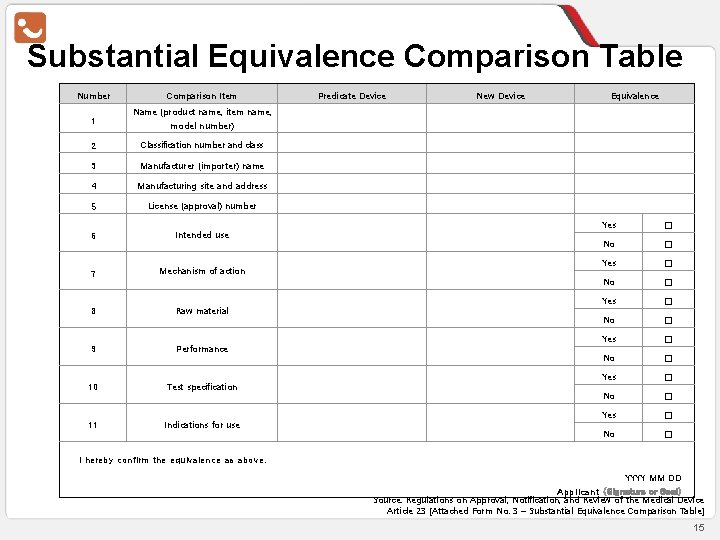
Substantial Equivalence Comparison Table Number 1 Comparison Item Predicate Device New Device Equivalence Name (product name, item name, model number) 2 Classification number and class 3 Manufacturer (importer) name 4 Manufacturing site and address 5 License (approval) number 6 Intended use 7 Mechanism of action 8 Raw material 9 Performance 10 Test specification 11 Indications for use Yes □ No □ Yes □ No □ I hereby confirm the equivalence as above. YYYY MM DD Applicant (Signature or Seal) Source: Regulations on Approval, Notification, and Review of the Medical Device Article 23 [Attached Form No. 3 – Substantial Equivalence Comparison Table] 15
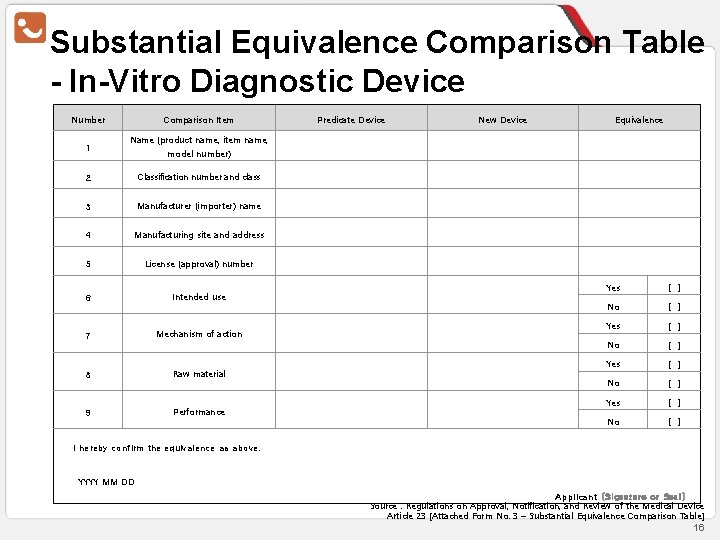
Substantial Equivalence Comparison Table - In-Vitro Diagnostic Device Number 1 Comparison Item Predicate Device New Device Equivalence Name (product name, item name, model number) 2 Classification number and class 3 Manufacturer (importer) name 4 Manufacturing site and address 5 License (approval) number 6 Intended use 7 8 9 Mechanism of action Raw material Performance Yes [] No [] I hereby confirm the equivalence as above. YYYY MM DD Applicant (Signature or Seal) Source : Regulations on Approval, Notification, and Review of the Medical Device Article 23 [Attached Form No. 3 – Substantial Equivalence Comparison Table] 16
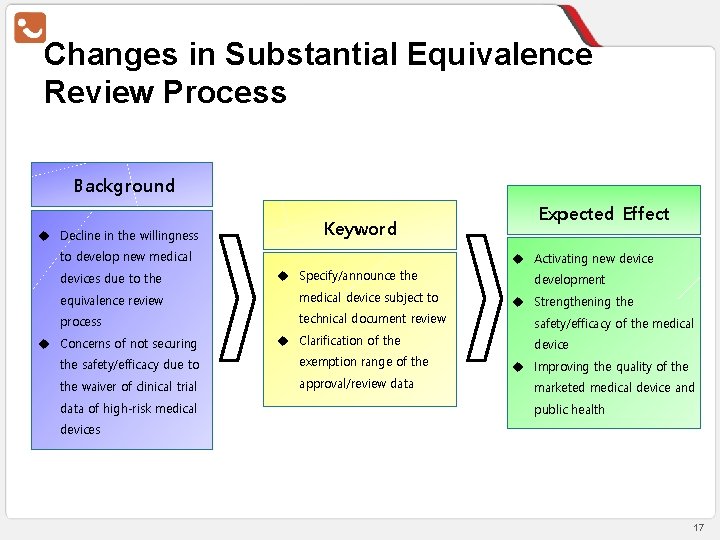
Changes in Substantial Equivalence Review Process Background u Decline in the willingness Keyword to develop new medical devices due to the u Activating new device u Specify/announce the equivalence review medical device subject to process technical document review u Concerns of not securing u Clarification of the safety/efficacy due to exemption range of the waiver of clinical trial approval/review data of high-risk medical Expected Effect development u Strengthening the safety/efficacy of the medical device u Improving the quality of the marketed medical device and public health devices 17

Major Changes u Eliminating the concepts of new device, modified device, and equivalent device, and only limiting to the equivalent device u Equivalent device among class 2 medical devices -> Subject to technical document review : The Minister of Ministry of Food and Drug Safety specifies and announces the medical devices with clinically universal and standard technology from subclasses. u Deletion of categorized review based on the substantial equivalence comparison table and equivalence criteria into new device, modified device, and equivalent device u Re-establish the waiver of review data by class according to the scope of submitting data, such as technical document, etc. -> Review data exemption range : Developmental details, clinical trial, and foreign usage status : Developmental details, measuring principles, and clinical performance study (in-vitro diagnostic device) -> Eligible for exemption : Medical device subject to technical document review : Medical device applicable to the same product group with the licensed device via clinical trial -> Not eligible for exemption : Medical device consisting of raw material that has never been domestically used (only for human contact) : Medical device that requires clinical trial based on the decision of the Minister of Ministry of Food and Drug Safety 18

Major Changes u Waiver of submitting data related to the test specification rationale : If the specification that was announced or approved by the Minister of Ministry of Food and Drug Safety or international specification that is equal or greater is listed. u Re-establish factors of comparative data to the predicate device : Deletion of performance and test specification items : Revision of equivalence comparison table form -> “Equivalence comparison table to the predicate device” u For medical devices other than class 4, clinical data from the medical device with equivalent mechanism of action, performance, or intended use to the applying device may be submitted. u Expansion of papers published in Science Citation Index including SCIE u Clarify the subject of clinical data submission : Newly developed Class 3 that has an essentially different mechanism of action, performance, or intended use : Medical device required for traceability which is implanted to a human body for more than a year : Medical device recognized by the Minister of MFDS as requiring clinical trial 19
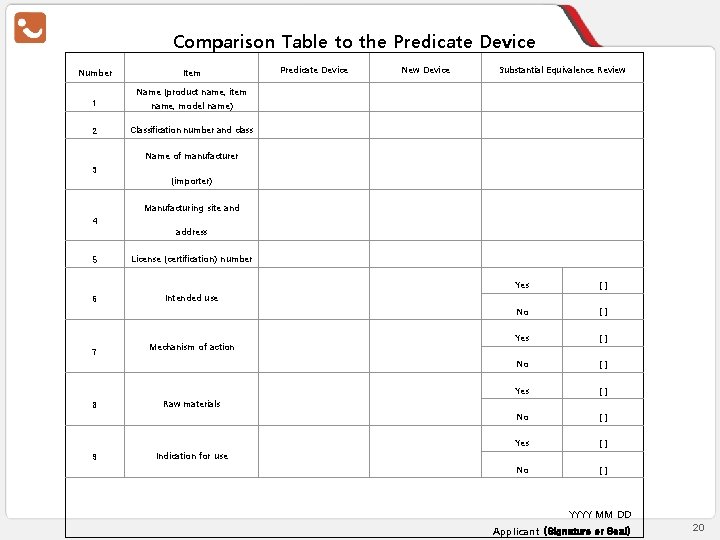
Comparison Table to the Predicate Device Number Item Predicate Device New Device Substantial Equivalence Review Name (product name, item 1 name, model name) 2 Classification number and class Name of manufacturer 3 (importer) Manufacturing site and 4 address 5 License (certification) number 6 Intended use 7 8 9 Mechanism of action Yes [] No [] Raw materials Indication for use YYYY MM DD Applicant (Signature or Seal) 20
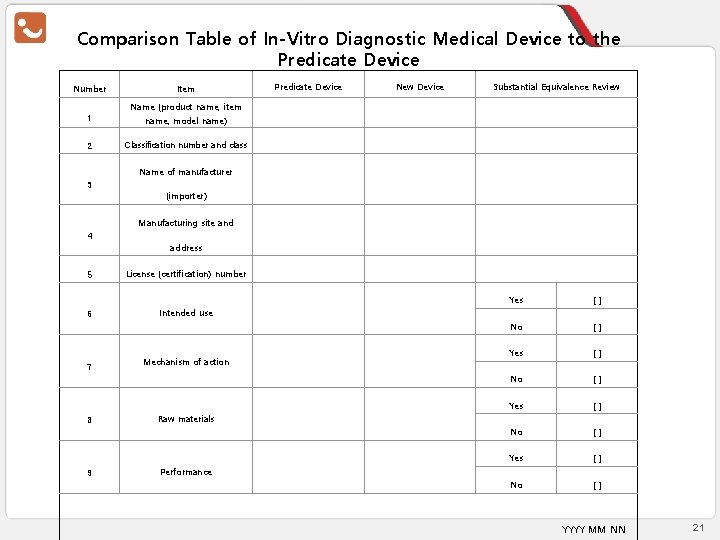
Comparison Table of In-Vitro Diagnostic Medical Device to the Predicate Device Number Item Predicate Device New Device Substantial Equivalence Review Name (product name, item 1 name, model name) 2 Classification number and class Name of manufacturer 3 (importer) Manufacturing site and 4 address 5 6 7 8 9 License (certification) number Yes [] No [] Intended use Mechanism of action Raw materials Performance YYYY MM NN 21

Concerns and Anticipation of Changes Concerns of the Industry u Increasing social cost : Excessive pre-approval cost : Decrease in health care accessibility : The medium and long term regulatory barriers u Disharmony in an international harmony perspective : Establishing substantial equivalence policy : Related literature, non-clinical data, postmarketing clinical evaluation and adverse event report u Difficulty in the clinical trial : Excessive cost u Difficulty in expanding the clinical study data : Meeting clinical study data requirement : Limit to the same product group Counterplans of the Industry u Strengthening the substantial equivalence comparison policy : Submitting technological/clinical/biological equivalent comparison/analysis and demonstrating data in the manner of regulatory advanced countries u Strengthening safety based on the whole cycle safety management system via balances between pre-approval and postapproval management u Considering the introduction of the postapproval clinical evaluation : Clinical evaluation throughout the life cycle : Expansion of the clinical data scope - RWE, clinical evaluation report, etc. - Post-marketing surveillance (PMS) - Modeling and simulation 22
Thank you. 23
- Slides: 23