Introducing elements compounds and mixtures 06 September 2021

Introducing elements, compounds and mixtures 06 September 2021 Learning question: What is the difference between elements, compounds and mixtures?

Learning objective: • Distinguish between an atom, an element, a compound a mixture

Key words: • Atom • Element • Compound • Mixture
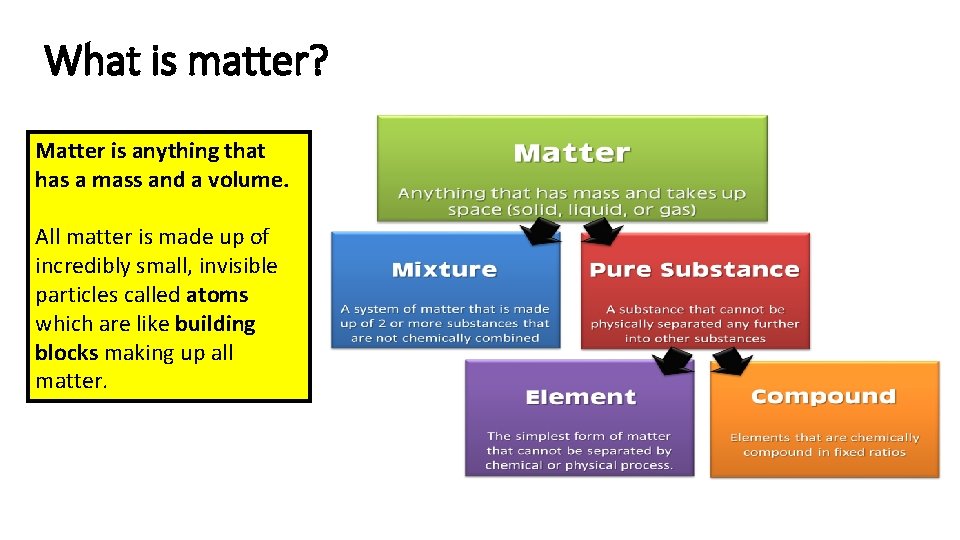
What is matter? Matter is anything that has a mass and a volume. All matter is made up of incredibly small, invisible particles called atoms which are like building blocks making up all matter.

Atoms • Everything is made from atoms, including you. • Atoms are tiny particles that are far too small to see, even with a microscope. If people were the same size as atoms, the entire population of the world would fit into a box about a thousandth of a millimeter across! • We usually image atoms as being like tiny balls or circles, but there is more to them than this.
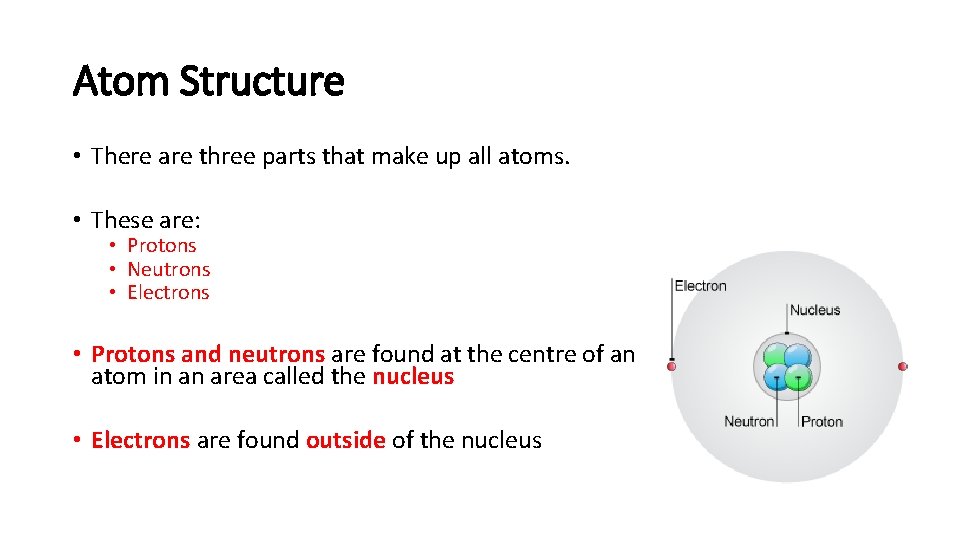
Atom Structure • There are three parts that make up all atoms. • These are: • Protons • Neutrons • Electrons • Protons and neutrons are found at the centre of an atom in an area called the nucleus • Electrons are found outside of the nucleus
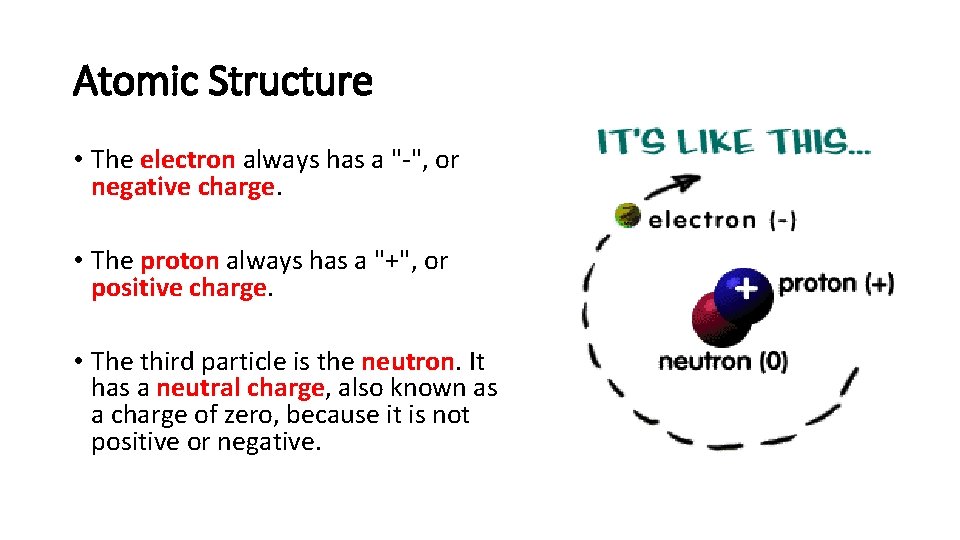
Atomic Structure • The electron always has a "-", or negative charge. • The proton always has a "+", or positive charge. • The third particle is the neutron. It has a neutral charge, also known as a charge of zero, because it is not positive or negative.
Task 1 • Draw and label a picture of an atom without looking at any notes! Make sure you include the charges of each particle.

Elements • Elements are a pure substance made up of only one type of atom. • There are over 118 different elements known to man and each has a special name. E. g. carbon, oxygen and hydrogen are all elements. • Elements have been identified and arranged into a table called the periodic table of elements. (more on this later!)


Compounds • A compound is a substance that contains atoms of two or more different elements, and these atoms are chemically joined together. • For example, water is a compound of hydrogen and oxygen. • Each of its particles contains two hydrogen atoms and one oxygen atom. • There are very many different compounds. The small white circles represent hydrogen atoms The larger red circles represent oxygen atoms
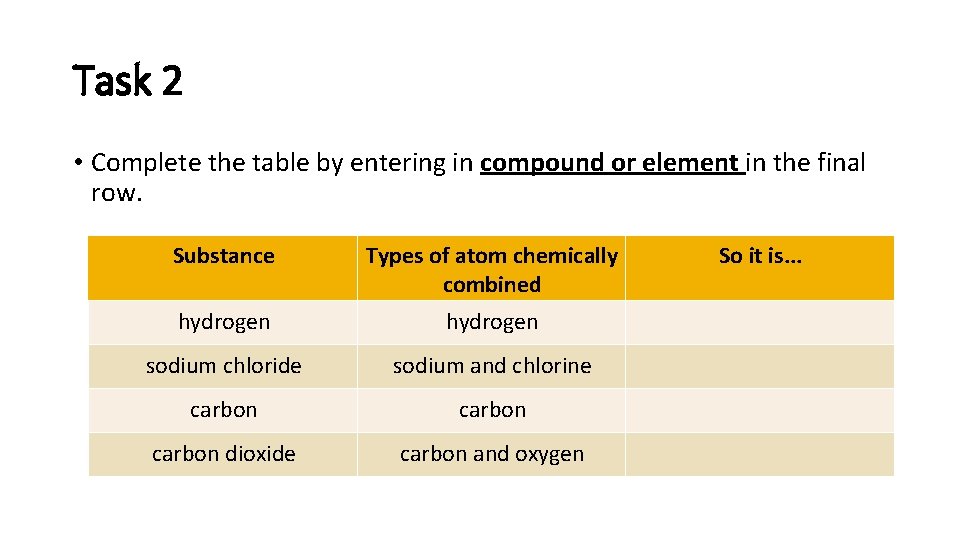
Task 2 • Complete the table by entering in compound or element in the final row. Substance Types of atom chemically combined hydrogen sodium chloride sodium and chlorine carbon dioxide carbon and oxygen So it is. . .

Mixtures • A mixture is made from different substances that are not chemically joined. • For example powdered iron and powdered sulphur mixed together makes a mixture of iron and sulphur. • They can be separated from each other without a chemical reaction, in the way that different coloured lollies can be picked out from a mixed packet and put into separate piles.


Task 3 • Complete the quiz on what you have learned so far. • Remember, you can use your notes to help you, or you can test your understanding by trying it without any support.

Homework • Learn the definitions of the key words for next lesson. • There will be a quiz to see how well you know them! Key words: • Atom • Element • Compound • Mixture
- Slides: 16