Intro to Physical Science Unit 1 Science Comes































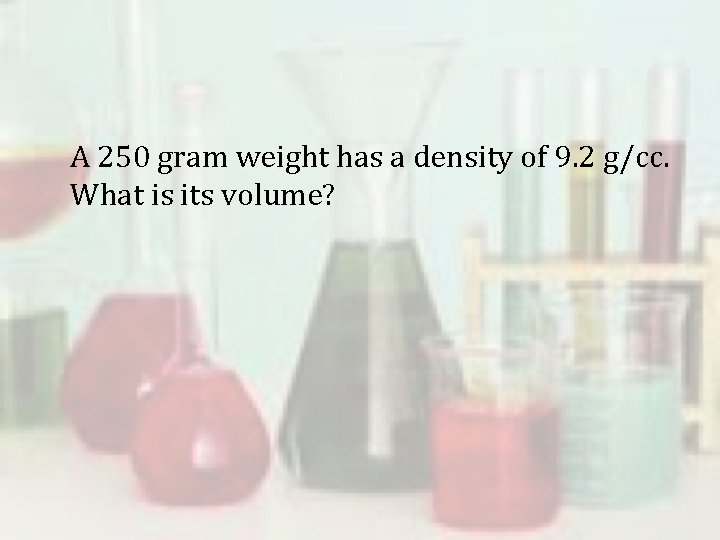
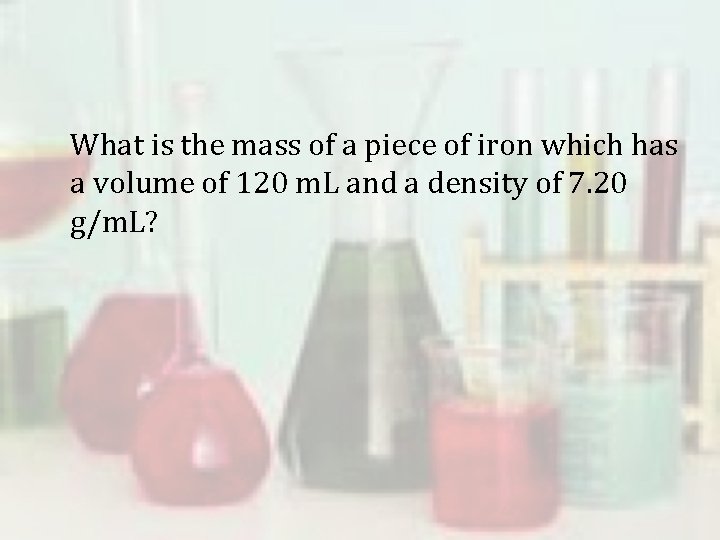












- Slides: 54

Intro to Physical Science Unit 1

Science • Comes from the Latin word that means “having knowledge”. • Science – a way of learning about the natural world that is based on evidence and logic.

How Science Advances • New evidence is usually used to improve earlier ideas rather than entirely replace them. – Different conclusions can be drawn from the same observations, and its not possible to tell which one is correct. – Example: Sun and Earth’s orbit.

Theories and Laws • Scientific theory – a broad explanation that is widely accepted because it is supported by a great deal of evidence. – Example: Kinetic theory of matter

Theories and Laws • Scientific law – a statement describing what always happens under certain conditions in nature. – It answers “how” questions not “why” questions. – Example: Newton’s Law of Gravity – http: //studyjams. scholastic. com/studyjams/ science/scientific-inquiry/scientific-theory-andevid. htm

Physical Science • Physical Science – the study of matter and energy. • Matter is all the “stuff” that exists in the universe. – Examples: tables, you, air • Energy is what gives matter the ability to move and change. – Examples: electricity, heat, light

Physical Science • Physical Science can be divided into two areas: – Chemistry – focuses on matter and energy at the scale of atoms and molecules. – Physics – focuses on matter and energy at all scales, from atoms to outer space.

Chemistry • Chemistry – the study of the structure, properties, and interactions of matter. – Important concepts: • Physical changes – Water freezing • Chemical reactions – Fireworks exploding

Physics • Physics – the study of energy and how it interacts with matter. – Important concepts: • Motion • Forces – magnetism • Forms of energy

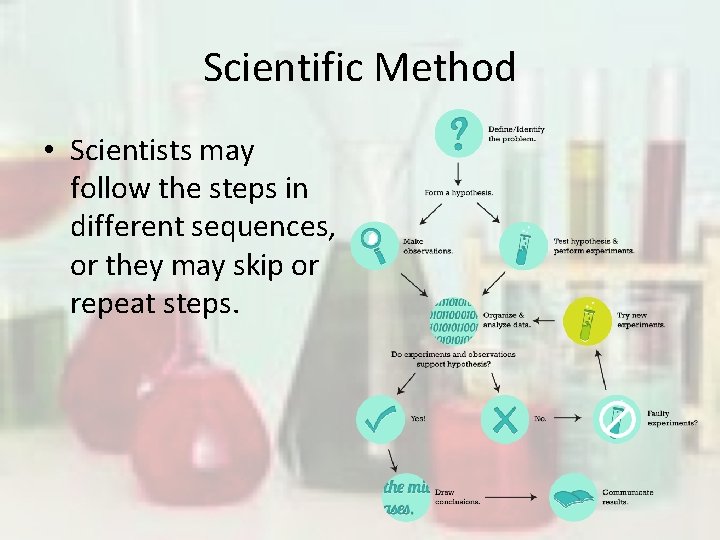
Scientific Method an organized set of investigation procedures 1. 2. 3. 4. 5. 6. State the Problem Form a Hypothesis Test the Hypothesis Analyze Data Draw Conclusions Communicate Results

Scientific Method • Scientists may follow the steps in different sequences, or they may skip or repeat steps.

1. ) State the Problem • Observation – any information that is gathered with the senses. • Questions often arise because of an observation. • The question of “why” or “how” is the problem • Example: Does temperature affect the strength of a magnet

2. ) Forming a Hypothesis • Hypothesis – Educated guess using what you know and what you observe – Must be something that can be tested – Usually “if-then” statements – Example: A magnet is stronger at lower temperatures.

3. ) Testing a Hypothesis • conduct an experiment – organized procedure for testing a hypothesis; tests the effect of one thing on another under controlled conditions • make observations • build a model to help – represents an idea, event, or object to help people better understand it

Variables variable factor that can cause a change in the results of an experiment 2 types independent variable/manipulated variable factor that, as it changes, affects the measure of another variable The factor that is changed by the researcher dependent variable/responding variable factor that changes as a result of changes in the other variables changes

Constants and Controls • Constant – a factor that does not change when other variables change • Control – the standard by which the test results can be compared

4. ) Analyzing the Data • record observations • organizing the data into easy-to-read tables and graphs

5. ) Drawing Conclusions • ask the question: Is your hypothesis supported by the analysis of your data? • for the hypothesis to be considered valid, the experiment must result in the exact same data every time it is repeated

6) Communicating results • Researchers should always communicate their results. • Allows other scientists to give feedback and repeat the investigation. • Replication – getting the same results when an experiment is repeated

Communicating Results • Write articles for peer-reviewed science journals. • Presentations at scientific meetings. • Creating websites • Writing articles for newspapers and magazines

Introduction • Measurement is an important science skill. • Other skills needed: – Keeping records – Doing calculations – Organizing data – Making models – Safety

Measurement Systems • the English system of measurement – United States • The measurement system used by most scientists and most other nations is the metric system – system of measurement based on multiples of ten

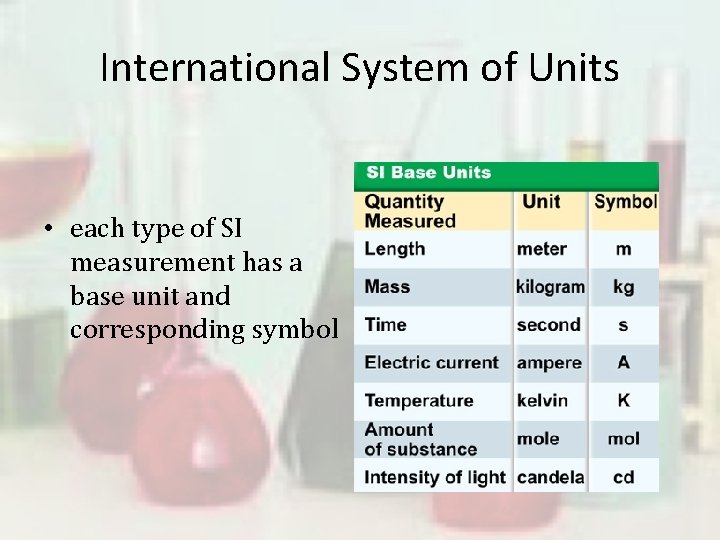
International System of Units • all SI (system international) standards are universally accepted and understood by scientists throughout the world

International System of Units • each type of SI measurement has a base unit and corresponding symbol

Measuring Distance • length is the distance between two points • Base Unit – meter (m) • Tool – metric ruler and meter sticks

• the size of the unit you measure with will depend on the size of the object being measured • Example • What unit would you use to measure the classroom width? • Meter • What unit would you use to measure the length of your pencil? • Centimeter

Measuring Volume (Liquids) • Base Unit – liter (L) • Tool – graduated cylinder – read at eye level at the meniscus • the curvature of a liquid in a container http: //jchemed. chem. wisc. edu/JCESoft/web/ cplsample/Modules/gradcyl/grad 10 m. L. htm http: //morrisonlabs. com/meniscus. htm

Measuring Volume (Solids) • volume – the amount of space occupied by an object • Regular Solid – length x width x height – Base Unit – cm 3 – Tool – ruler • 1 m. L = 1 cm 3

• Irregular Solid – Water displacement method – using a graduated cylinder with water to determine the volume of a irregular solid – Base unit – liter (L) – Tool – graduated cylinder or overflow can and catch pail

Measuring Matter • mass – is a measurement of the quantity of matter in an object. – Base Unit – gram (g) – Tool – balance – https: //youtu. be/C 9 how. XG 7 LUY

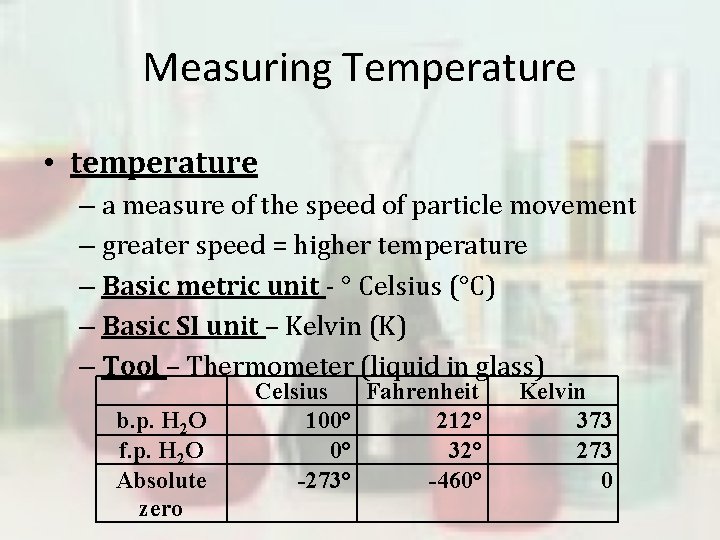
Measuring Temperature • temperature – a measure of the speed of particle movement – greater speed = higher temperature – Basic metric unit - ° Celsius (°C) – Basic SI unit – Kelvin (K) – Tool – Thermometer (liquid in glass) b. p. H 2 O f. p. H 2 O Absolute zero Celsius Fahrenheit 100° 212° 0° 32° -273° -460° Kelvin 373 273 0

Density • Density – mass to volume ratio of a substance • density is the same for the same material regardless of sample size • can be used to identify a material

Density DENSITY = __MASS__ VOLUME D=M V Units g/m. L, g/cm 3, g/cc • 1 m. L = 1 cm 3 • cc =cubic centimeter, same as cm 3 • Density of H 2 O = 1 g/m. L

Density Problems A piece of glass has a volume of 75 cm 3 and weighs 200 grams. What is its density?
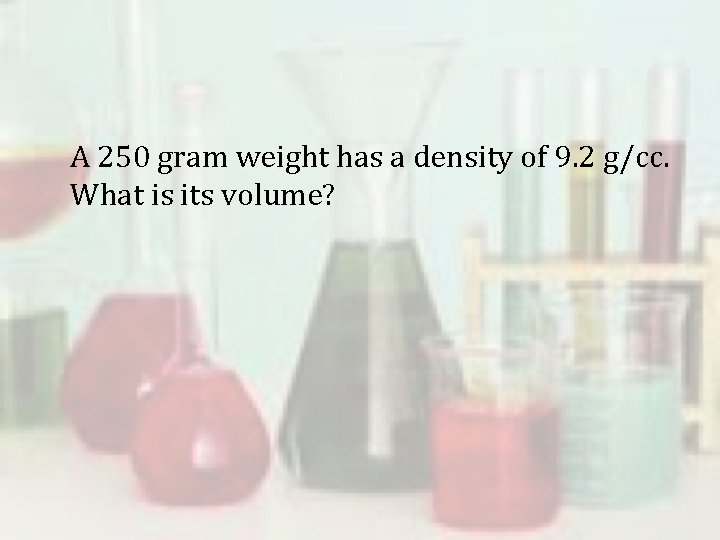
A 250 gram weight has a density of 9. 2 g/cc. What is its volume?
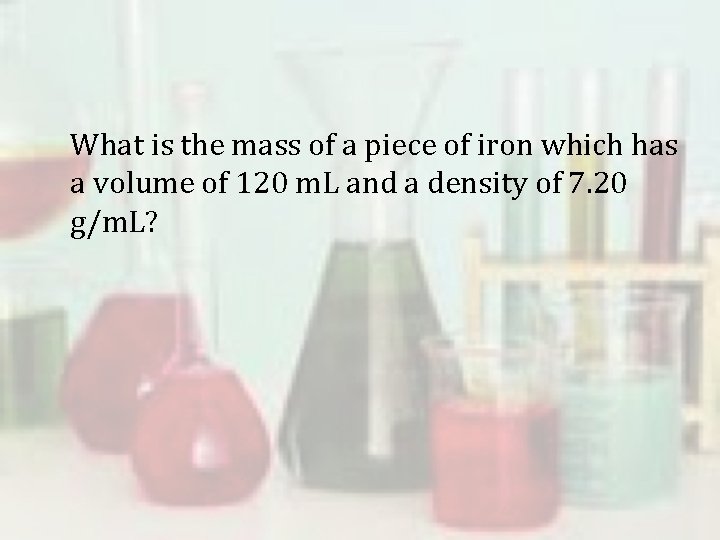
What is the mass of a piece of iron which has a volume of 120 m. L and a density of 7. 20 g/m. L?

Metric Conversions • Since metric is based on the number 10 metric conversions are very simple. • Simply move the decimal place to convert from one unit to another.

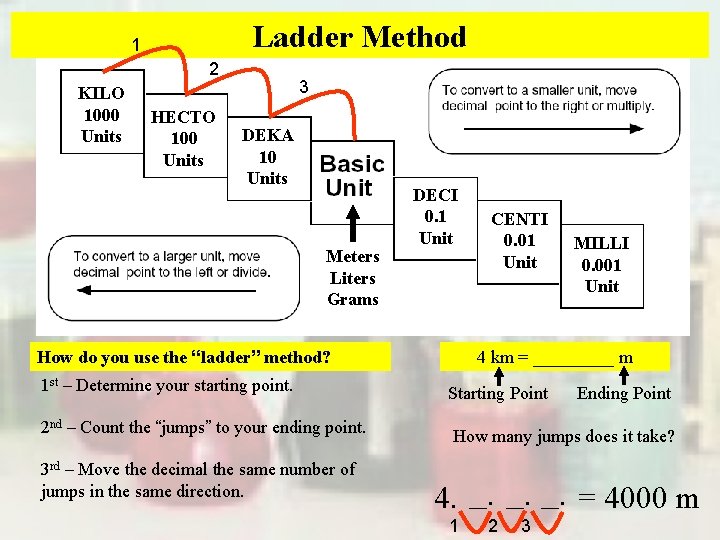
Ladder Method 1 2 KILO 1000 Units HECTO 100 Units 3 DEKA 10 Units Meters Liters Grams DECI 0. 1 Unit How do you use the “ladder” method? 1 st – Determine your starting point. 2 nd – Count the “jumps” to your ending point. 3 rd – Move the decimal the same number of jumps in the same direction. CENTI 0. 01 Unit MILLI 0. 001 Unit 4 km = _____ m Starting Point Ending Point How many jumps does it take? 4. __. __. = 4000 m 1 2 3

Conversion Practice Try these conversions using the ladder method. 1000 mg = _______ g 1 L = _______ m. L 160 cm = _______ mm 14 km = _______ m 109 g = _______ kg 250 m = _______ km Compare using <, >, or =. 56 cm 6 m 7 g 698 mg

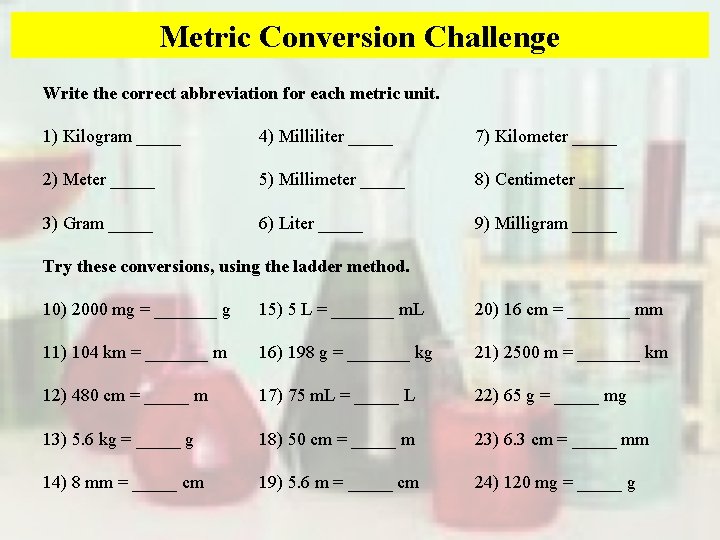
Metric Conversion Challenge Write the correct abbreviation for each metric unit. 1) Kilogram _____ 4) Milliliter _____ 7) Kilometer _____ 2) Meter _____ 5) Millimeter _____ 8) Centimeter _____ 3) Gram _____ 6) Liter _____ 9) Milligram _____ Try these conversions, using the ladder method. 10) 2000 mg = _______ g 15) 5 L = _______ m. L 20) 16 cm = _______ mm 11) 104 km = _______ m 16) 198 g = _______ kg 21) 2500 m = _______ km 12) 480 cm = _____ m 17) 75 m. L = _____ L 22) 65 g = _____ mg 13) 5. 6 kg = _____ g 18) 50 cm = _____ m 23) 6. 3 cm = _____ mm 14) 8 mm = _____ cm 19) 5. 6 m = _____ cm 24) 120 mg = _____ g

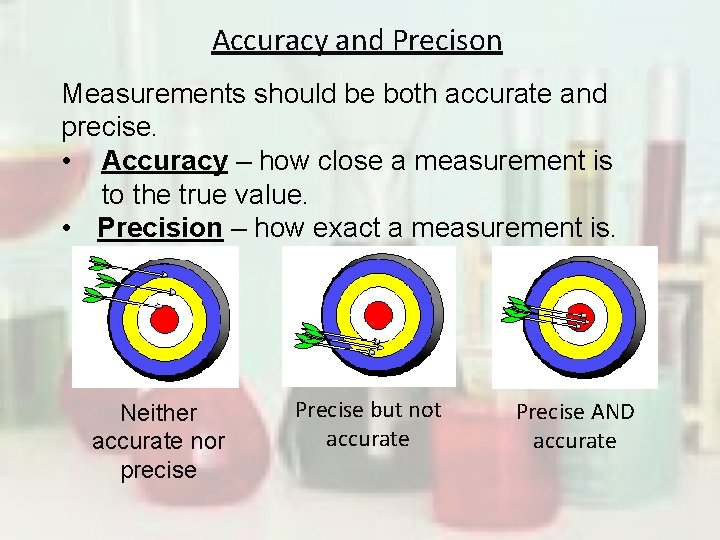
Accuracy and Precison Measurements should be both accurate and precise. • Accuracy – how close a measurement is to the true value. • Precision – how exact a measurement is. Neither accurate nor precise Precise but not accurate Precise AND accurate

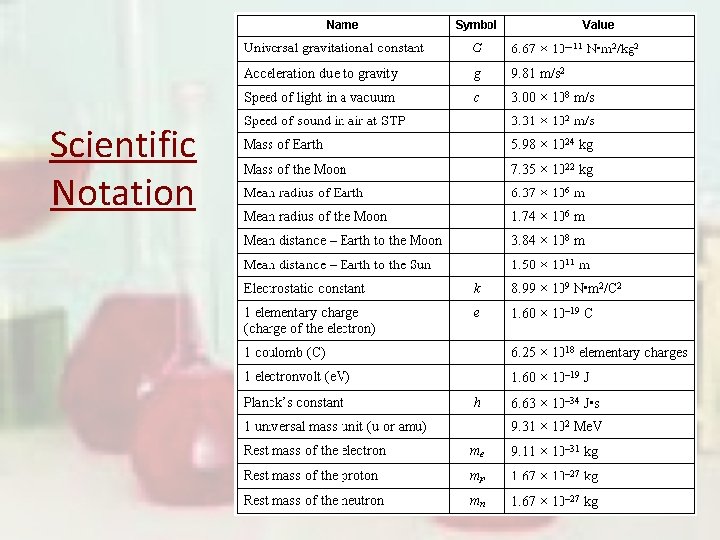
Scientific Notation

Scientific Notation In science, we deal with some very LARGE numbers: 1 mole = 60200000000000 In science, we deal with some very SMALL numbers: Mass of an electron = 0. 000000000000000091 kg

Imagine the difficulty of calculating the mass of 1 mole of electrons! 0. 000000000000000091 kg x 60200000000000 ? ? ? ? ? ? ? ? ?

Scientific Notation: A method of representing very large or very small numbers in the form: M x 10 n Ø M is a number between 1 and 10 Ø n is an integer

2 500 000 9 8 7 6 5 4 3 Step #1: Insert an understood decimal point Step #2: Decide where the decimal must end up so that one number is to its left Step #3: Count how many places you bounce the decimal point Step #4: Re-write in the form M x 10 n 2 . 1

2. 5 x 9 10 The exponent is the number of places we moved the decimal.

0. 0000579 1 2 3 4 5 Step #2: Decide where the decimal must end up so that one number is to its left Step #3: Count how many places you bounce the decimal point Step #4: Re-write in the form M x 10 n

5. 79 x -5 10 The exponent is negative because the number we started with was less than 1.

Descriptive Statistics • Mean – the average value • Range – the total spread of values

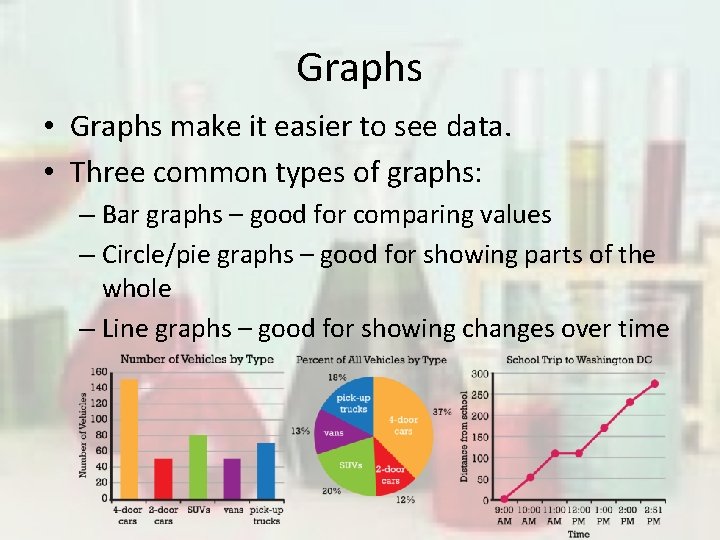
Graphs • Graphs make it easier to see data. • Three common types of graphs: – Bar graphs – good for comparing values – Circle/pie graphs – good for showing parts of the whole – Line graphs – good for showing changes over time

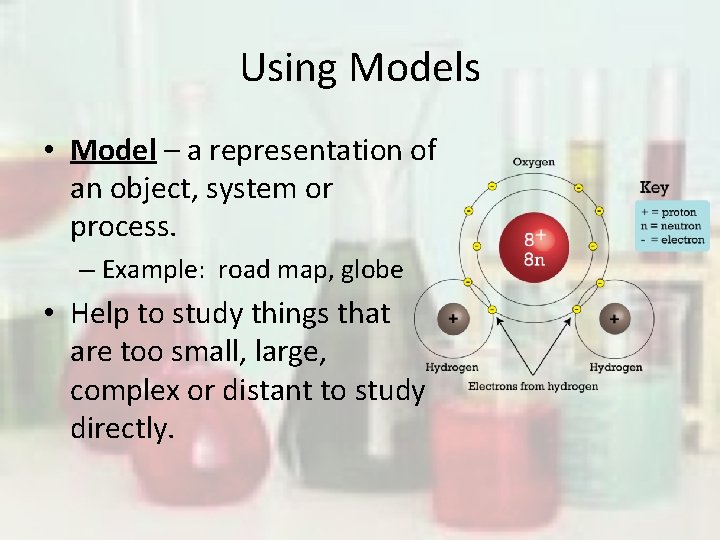
Using Models • Model – a representation of an object, system or process. – Example: road map, globe • Help to study things that are too small, large, complex or distant to study directly.

Safety Symbols

Safety Rules • • Wear safety goggles. Wear appropriate clothing. Tie hair back. Do not eat or drink in lab. Don’t smell things directly (fan vapors). Never perform unauthorized experiments. Wash hand with soap and water before leaving lab.