Intro to Nuclear Chemistry Objective To use atomic
Intro to Nuclear Chemistry Objective – To use atomic mass number to find the abundance of an isotope Mr. Henshaw

Nucleons • Protons and Neutrons • The nucleons are bound together by the strong force.

Isotopes • Atoms of a given element with: same #protons but different # neutrons

17 Cl Isotopes 37 • Chlorine-37 – atomic #: 17 – mass #: 37 – # of protons: 17 37 – # of electrons: 17 17 – # of neutrons: 20 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem Cl
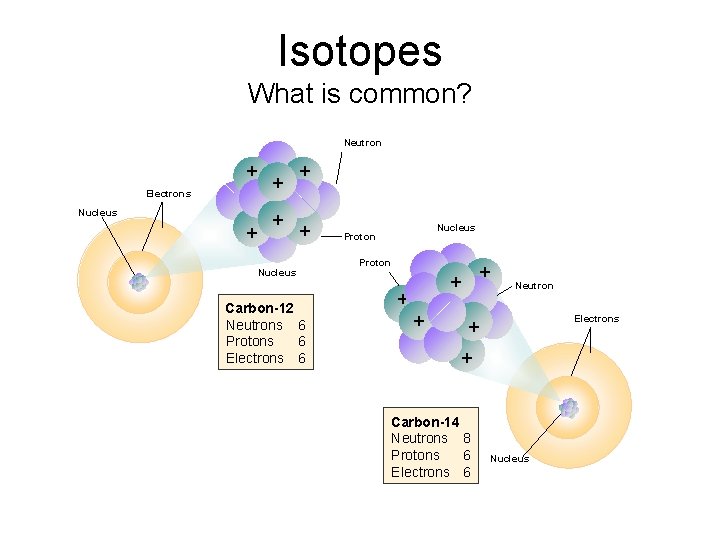
Isotopes What is common? Neutron + Electrons + Nucleus + + Nucleus Carbon-12 Neutrons 6 Protons 6 Electrons 6 Nucleus Proton + + Neutron Electrons + + Carbon-14 Neutrons 8 Protons 6 Electrons 6 Nucleus
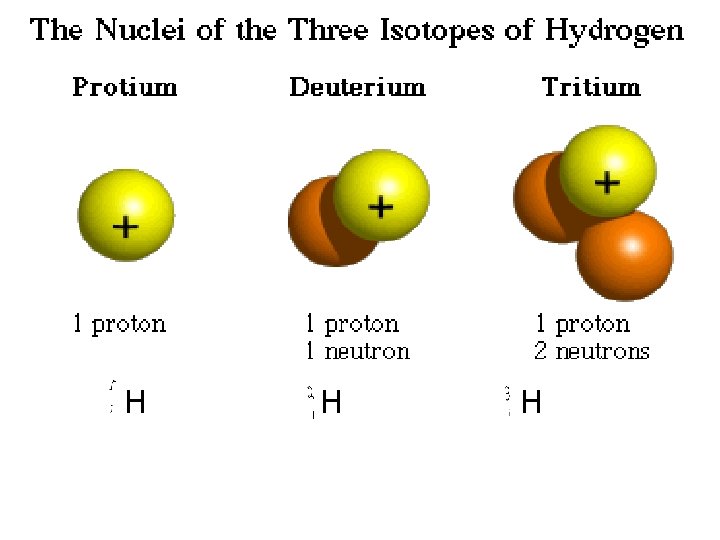
H H

Average Atomic Mass • weighted average of all isotopes • on the Periodic Table • Put %’s in decimal form Avg. (mass)(%) + (mass)(%) Atomic = Mass Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Average Atomic Mass Number Natural Abundance Half-life 85 72. 17% STABLE 87 27. 83% 4. 81× 10+10 years Avg. Atomic = Mass

Marbles in a Jar Periodic Table already gives you the Avg. Atomic Mass ? Isotopes of Marble Mass # (g/mol) Marble - 2 2 Marble - 3 3 1 M A) Which Marble is more Abundant? ______ Marble 2. 22 B) What Percentage of Marble-2 is in the jar? ____ %

Abundance of an Isotope • EX: There are 2 known isotopes of Chlorine. (Chlorine-35 and Chlorine-37). What are their abundances? Avg. Atomic = (35)(. %) + (37)(. %) = 35. 40 amu Mass

Bag of Pennies Isotope year Abundance Penny- 2. 5 >1982 X = Penny -3. 1 <=1982 1 -X = Average Mass of 2. 89 g Pennies There are 100 pennies in the bag. How many of them are minted in 1982 or before?

Isotopes of Carbon
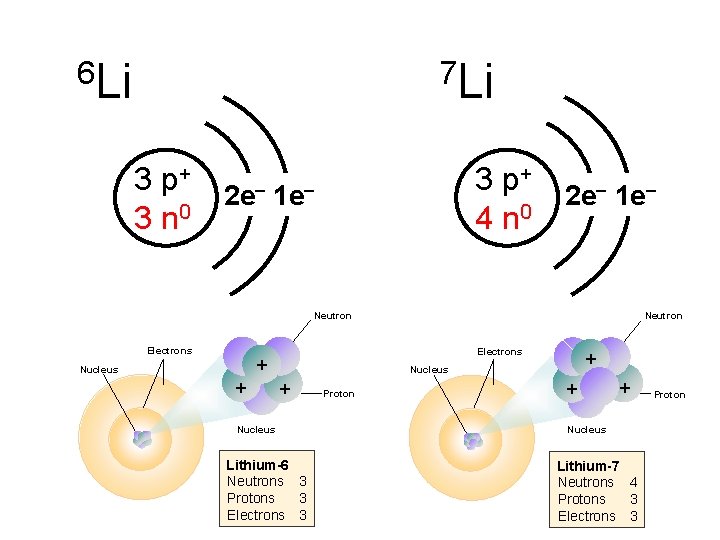
6 Li 7 Li 3 p+ 3 n 0 3 p+ 4 n 0 2 e– 1 e– Neutron Electrons + Electrons + Nucleus Neutron + Nucleus + + Proton Nucleus Lithium-6 Neutrons 3 Protons 3 Electrons 3 + Nucleus Lithium-7 Neutrons 4 Protons 3 Electrons 3 Proton
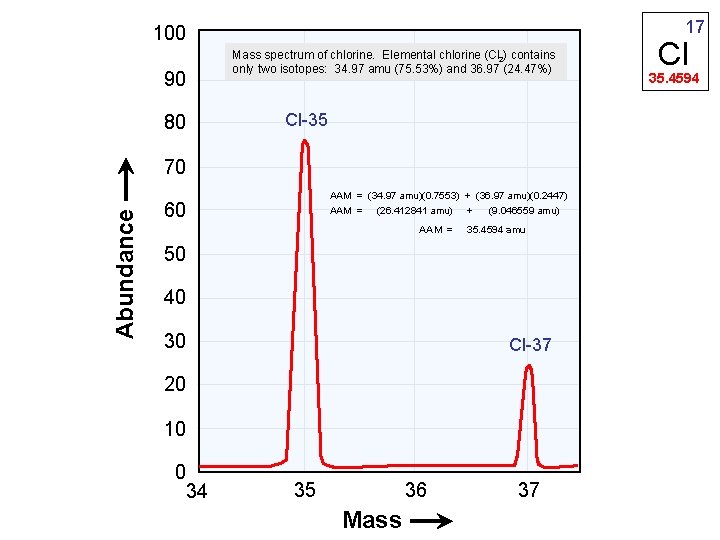
17 100 Mass spectrum of chlorine. Elemental chlorine (Cl 2) contains only two isotopes: 34. 97 amu (75. 53%) and 36. 97 (24. 47%) 90 Cl-35 80 Abundance 70 AAM = (34. 97 amu)(0. 7553) + (36. 97 amu)(0. 2447) AAM = (26. 412841 amu) + (9. 046559 amu) 60 AAM = 35. 4594 amu 50 40 30 Cl-37 20 10 0 34 36 35 Mass 37 Cl 35. 4594
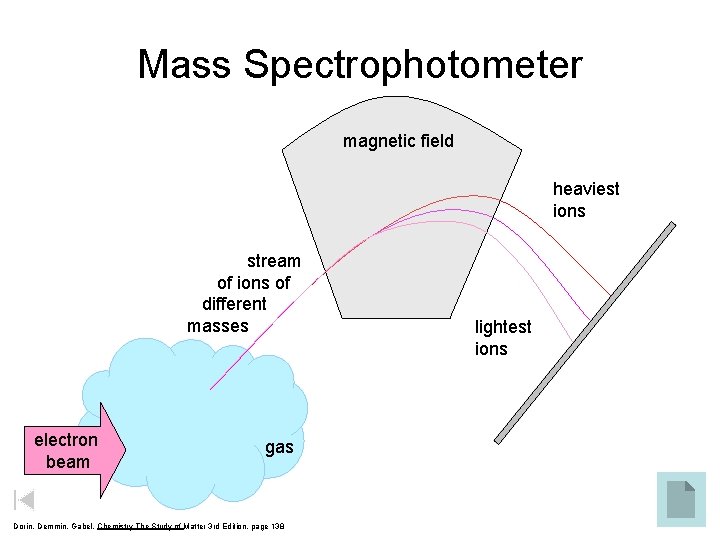
Mass Spectrophotometer magnetic field heaviest ions stream of ions of different masses electron beam gas Dorin, Demmin, Gabel, Chemistry The Study of Matter 3 rd Edition, page 138 lightest ions
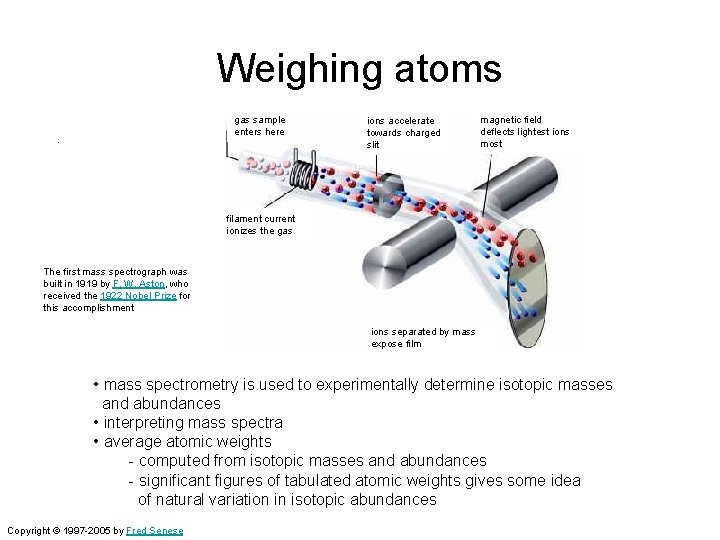
Weighing atoms. gas sample enters here ions accelerate towards charged slit magnetic field deflects lightest ions most filament current ionizes the gas The first mass spectrograph was built in 1919 by F. W. Aston, who received the 1922 Nobel Prize for this accomplishment ions separated by mass expose film • mass spectrometry is used to experimentally determine isotopic masses and abundances • interpreting mass spectra • average atomic weights - computed from isotopic masses and abundances - significant figures of tabulated atomic weights gives some idea of natural variation in isotopic abundances Copyright © 1997 -2005 by Fred Senese
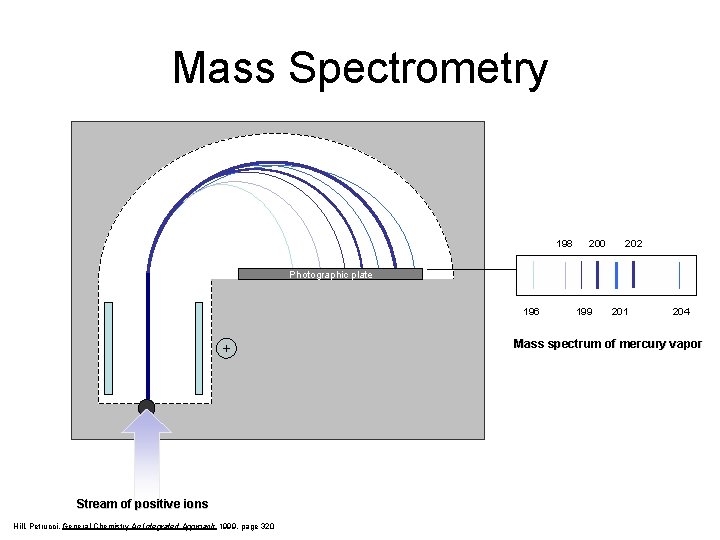
Mass Spectrometry 198 200 202 Photographic plate 196 199 201 204 - + Stream of positive ions Hill, Petrucci, General Chemistry An Integrated Approach 1999, page 320 Mass spectrum of mercury vapor
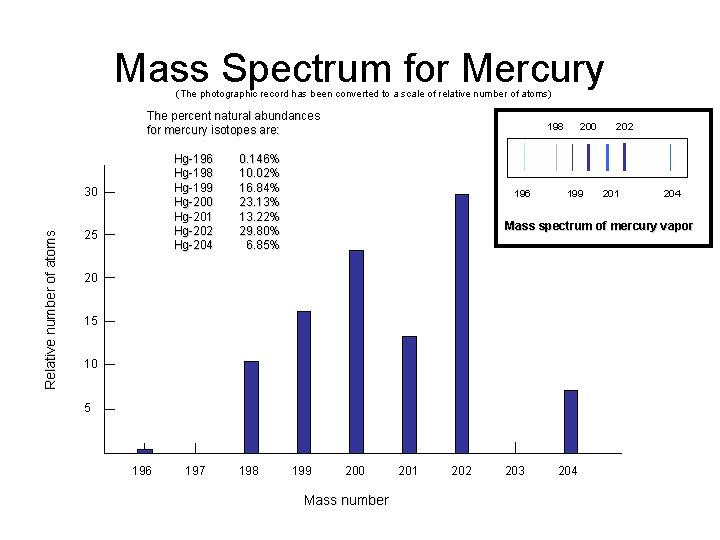
Mass Spectrum for Mercury (The photographic record has been converted to a scale of relative number of atoms) The percent natural abundances for mercury isotopes are: Relative number of atoms 30 25 Hg-196 0. 146% Hg-198 10. 02% Hg-199 16. 84% Hg-200 23. 13% Hg-201 13. 22% Hg-202 29. 80% Hg-204 6. 85% 198 200 202 196 199 201 204 Mass spectrum of mercury vapor 20 15 10 5 196 197 198 199 200 201 202 203 204 Mass number
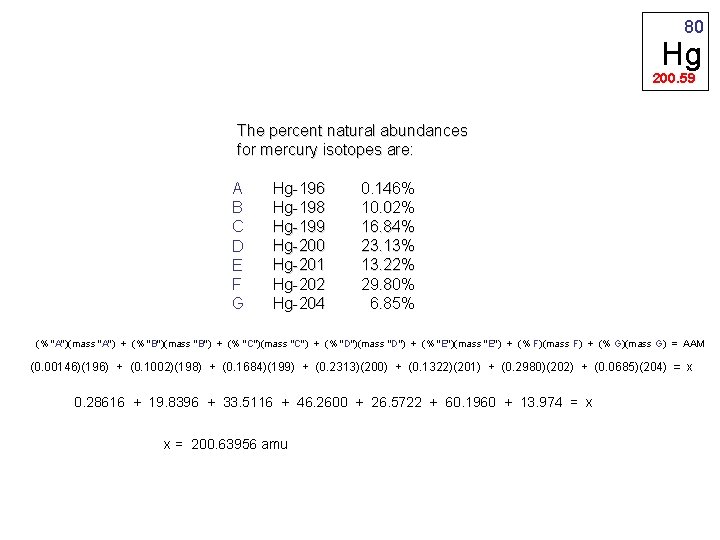
80 Hg 200. 59 The percent natural abundances for mercury isotopes are: A Hg-196 0. 146% B Hg-198 10. 02% C Hg-199 16. 84% D Hg-200 23. 13% E Hg-201 13. 22% F Hg-202 29. 80% G Hg-204 6. 85% (% "A")(mass "A") + (% "B")(mass "B") + (% "C")(mass "C") + (% "D")(mass "D") + (% "E")(mass "E") + (% F)(mass F) + (% G)(mass G) = AAM (0. 00146)(196) + (0. 1002)(198) + (0. 1684)(199) + (0. 2313)(200) + (0. 1322)(201) + (0. 2980)(202) + (0. 0685)(204) = x 0. 28616 + 19. 8396 + 33. 5116 + 46. 2600 + 26. 5722 + 60. 1960 + 13. 974 = x x = 200. 63956 amu

92 Separation of Isotopes U 238 Natural uranium, atomic weight = 238. 029 g/mol Density is 19 g/cm 3. Melting point 1000 o. C. Two main isotopes: 238 92 235 92 U U 99. 3% 0. 7% (238 amu) x (0. 993) + (235 amu) x (0. 007) 236. 334 amu + 1. 645 amu Because isotopes are chemically identical (same electronic structure), they cannot be separated by chemistry. So Physics separates them by diffusion or centrifuge (mass spectrograph is too slow)… 237. 979 amu

17 Cl 35. 453 • Assume you have only two atoms of chlorine. • One atom has a mass of 35 amu (Cl-35) • The other atom has a mass of 36 amu (Cl-36) • What is the average mass of these two isotopes? 35. 5 amu • Looking at the average atomic mass printed on the periodic table. . . approximately what percentage is Cl-35 and Cl-36? 55% Cl-35 and 45% Cl-36 is a good approximation

17 Cl 35. 453 Using our estimated % abundance data 55% Cl-35 and 45% Cl-36 calculate an average atomic mass for chlorine. Average Atomic Mass = (% abundance of isotope "A")(mass "A") + (% "B")(mass "B") AAM = (% abundance of isotope Cl-35)(mass Cl-35) + (% abundance of Cl-36)(mass Cl-36) AAM = (0. 55)(35 amu) + (0. 45)(36 amu) AAM = (19. 25 amu) + (16. 2 amu) AAM = 35. 45 amu

Isotopes Dalton was wrong. Atoms of the same element can have different numbers of neutrons different mass numbers called isotopes C-12 vs. C-14 California WEB

Naming Isotopes • Put the mass number after the name of the element • carbon- 12 • carbon -14 • uranium-235 California WEB
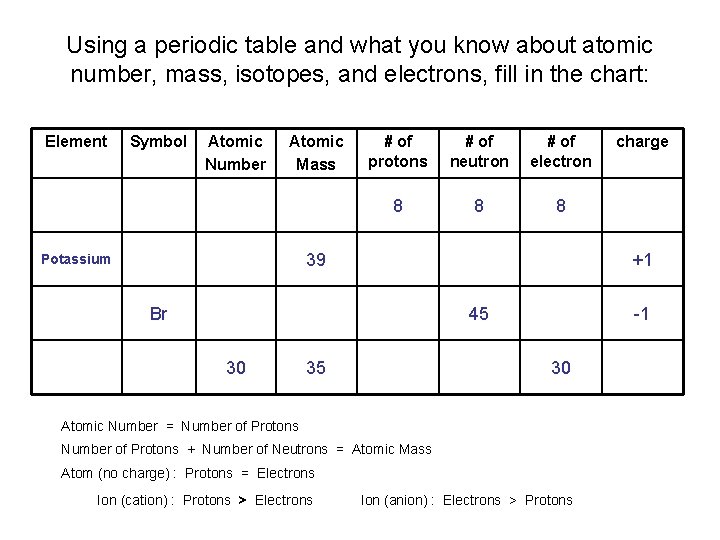
Using a periodic table and what you know about atomic number, mass, isotopes, and electrons, fill in the chart: Element Symbol Atomic Number Atomic Mass # of protons # of neutron # of electron 8 8 8 39 Potassium +1 Br 45 30 35 -1 30 Atomic Number = Number of Protons + Number of Neutrons = Atomic Mass Atom (no charge) : Protons = Electrons Ion (cation) : Protons > Electrons charge Ion (anion) : Electrons > Protons
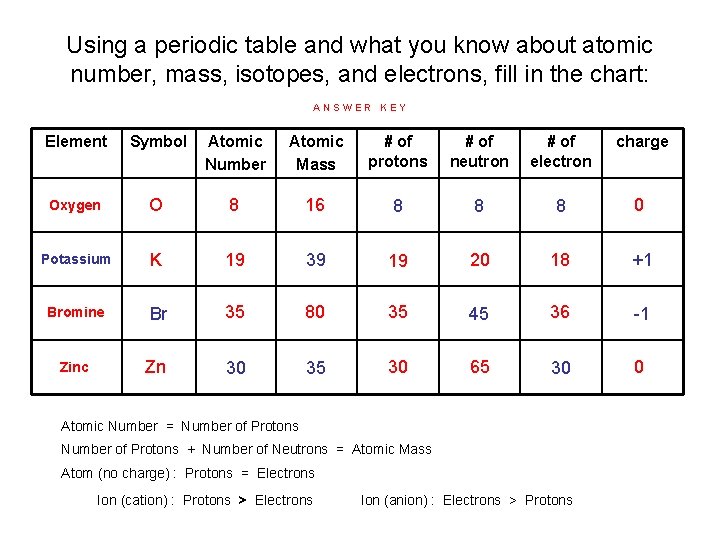
Using a periodic table and what you know about atomic number, mass, isotopes, and electrons, fill in the chart: ANSWER KEY Element Symbol Atomic Number Atomic Mass # of protons # of neutron # of electron charge Oxygen O 8 16 8 8 8 0 Potassium K 19 39 19 20 18 +1 Bromine Br 35 80 35 45 36 -1 Zinc Zn 30 35 30 65 30 0 Atomic Number = Number of Protons + Number of Neutrons = Atomic Mass Atom (no charge) : Protons = Electrons Ion (cation) : Protons > Electrons Ion (anion) : Electrons > Protons

Atomic Mass • • • How heavy is an atom of oxygen? There are different kinds of oxygen atoms. More concerned with average atomic mass. Based on abundance of each element in nature. Don’t use grams because the numbers would be too small

Measuring Atomic Mass • Unit is the Atomic Mass Unit (amu) • One twelfth the mass of a carbon-12 atom. • Each isotope has its own atomic mass we need the average from percent abundance. (1 amu) carbon atom (1 amu) (12 amu) (1 amu)

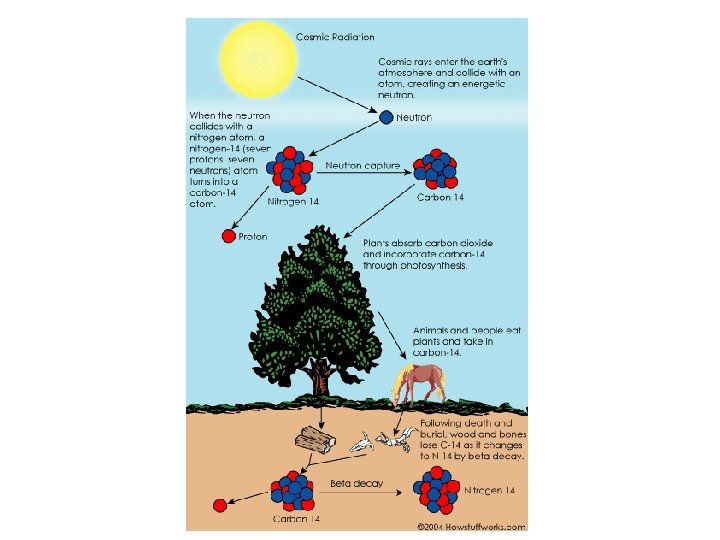
Mass spectrums reflect the abundance of naturally occurring isotopes. Natural Abundance of Common Elements Hydrogen 1 H = 99. 985% 2 H = 0. 015% Carbon 12 C = 98. 90% 13 C = 1. 10% Nitrogen 14 N = 99. 63% 15 N = 0. 37% Oxygen 16 O = 99. 762% 17 O = 0. 038% Sulfur 32 S = 95. 02% 33 S = 0. 75% 34 S = 4. 21% 36 S = 0. 02% Chlorine 35 Cl = 75. 77% 37 Cl = 24. 23% Bromine 79 Br = 50. 69% 81 Br = 49. 31% 18 O = 0. 200%

For example…. Methane • For carbon 1 in approximately 90 atoms are carbon-13 • The rest are carbon-12 the isotope that is 98. 9% abundant. • So, for approximately 90 methane molecules… 1 carbon is carbon-13
C-13 Where’s Waldo?
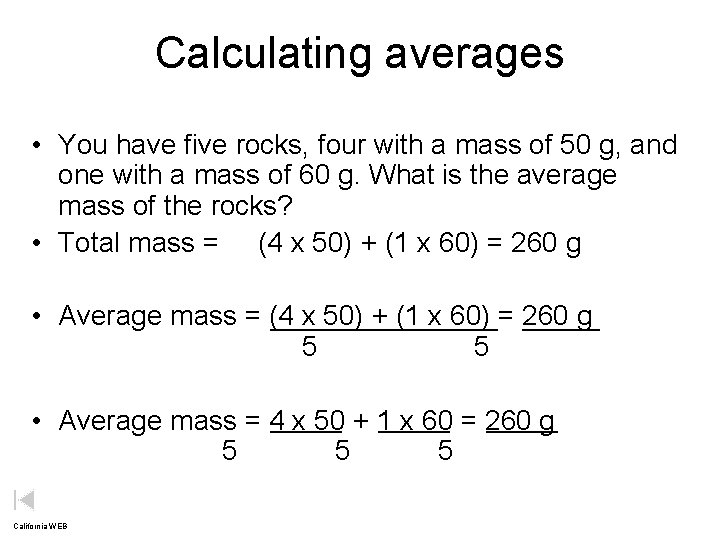
Calculating averages • You have five rocks, four with a mass of 50 g, and one with a mass of 60 g. What is the average mass of the rocks? • Total mass = (4 x 50) + (1 x 60) = 260 g • Average mass = (4 x 50) + (1 x 60) = 260 g 5 • Average mass = 4 x 50 + 1 x 60 = 260 g 5 5 5 California WEB
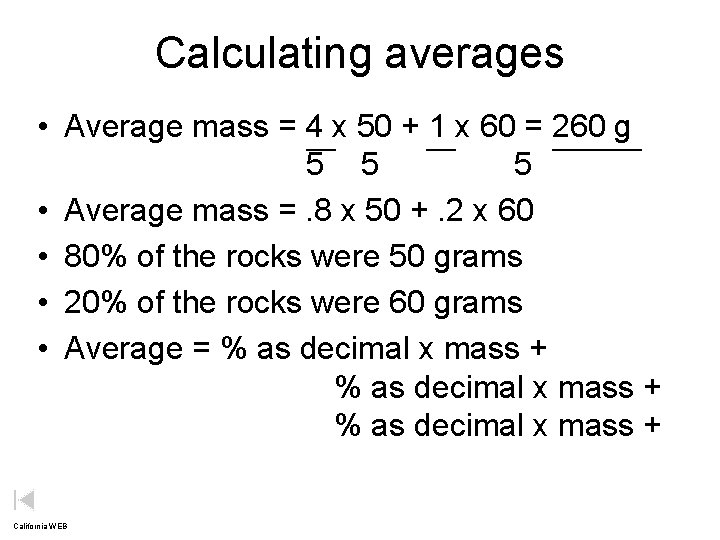
Calculating averages • Average mass = 4 x 50 + 1 x 60 = 260 g 5 5 5 • Average mass =. 8 x 50 +. 2 x 60 • 80% of the rocks were 50 grams • 20% of the rocks were 60 grams • Average = % as decimal x mass + California WEB

Isotopes • Because of the existence of isotopes, the mass of a collection of atoms has an average value. • Average mass = ATOMIC WEIGHT • Boron is 20% B-10 and 80% B-11. That is, B-11 is 80 percent abundant on earth. • For boron atomic weight = 0. 20 (10 amu) + 0. 80 (11 amu) = 10. 8 amu

Periodic Table • Dmitri Mendeleev developed the modern periodic table. • Argued that element properties are periodic functions of their atomic weights. • We now know that element properties are periodic functions of their ATOMIC NUMBERS.
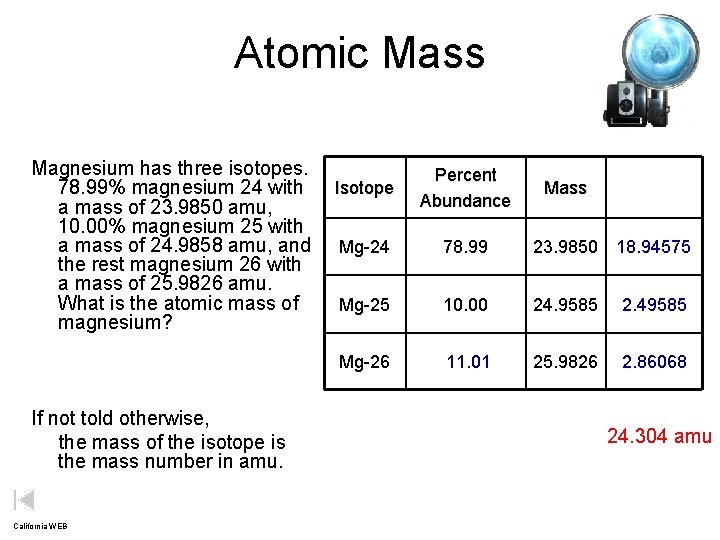
Atomic Mass Magnesium has three isotopes. 78. 99% magnesium 24 with Isotope a mass of 23. 9850 amu, 10. 00% magnesium 25 with a mass of 24. 9858 amu, and Mg-24 the rest magnesium 26 with a mass of 25. 9826 amu. What is the atomic mass of Mg-25 magnesium? Mg-26 If not told otherwise, the mass of the isotope is the mass number in amu. California WEB Percent Abundance Mass 78. 99 23. 9850 18. 94575 10. 00 24. 9585 2. 49585 11. 01 25. 9826 2. 86068 24. 304 amu

Atomic Mass Calculate the atomic mass of copper if copper has two isotopes. 69. 1% has a mass of 62. 93 amu and the rest has a mass of Percent 64. 93 amu. Isotope Mass Abundance Cu-63 69. 1 62. 93 43. 48463 Cu-65 30. 9 64. 93 20. 06337 63. 548 29 Cu 63. 548
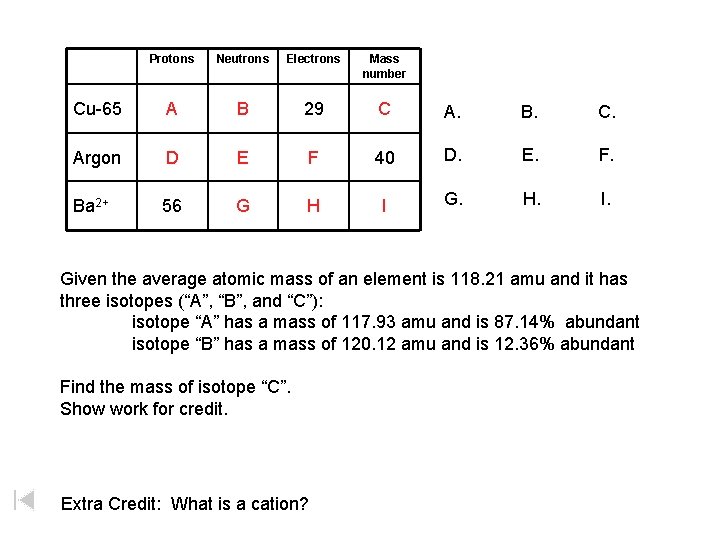
Protons Neutrons Electrons Mass number Cu-65 A B 29 C A. B. C. Argon D E F 40 D. E. F. Ba 2+ 56 G H I G. H. I. Given the average atomic mass of an element is 118. 21 amu and it has three isotopes (“A”, “B”, and “C”): isotope “A” has a mass of 117. 93 amu and is 87. 14% abundant isotope “B” has a mass of 120. 12 amu and is 12. 36% abundant Find the mass of isotope “C”. Show work for credit. Extra Credit: What is a cation?

Protons Neutrons Electrons Mass number Cu-65 A = 29 B = 36 29 C = 65 Argon D = 18 E = 22 F = 18 40 Ba 2+ 56 G = 81 H = 54 I = 137 Given the average atomic mass of an element is 118. 21 amu and it has three isotopes (“A”, “B”, and “C”): isotope “A” has a mass of 117. 93 amu and is 87. 14% abundant isotope “B” has a mass of 120. 12 amu and is 12. 36% abundant Find the mass of isotope “C”. 119. 7932 amu Show work for credit. Extra Credit: What is a cation? A positively charged atom. An atom that has lost a(n) electron(s).

Given the average atomic mass of an element is 118. 21 amu and it has three isotopes (“A”, “B”, and “C”): isotope “A” has a mass of 117. 93 amu and is 87. 14% abundant isotope “B” has a mass of 120. 12 amu and is 12. 36% abundant Find the mass of isotope “C”. Show work for credit.

3 Main Types of Radioactive Decay • Alpha a • Beta b • Gamma g

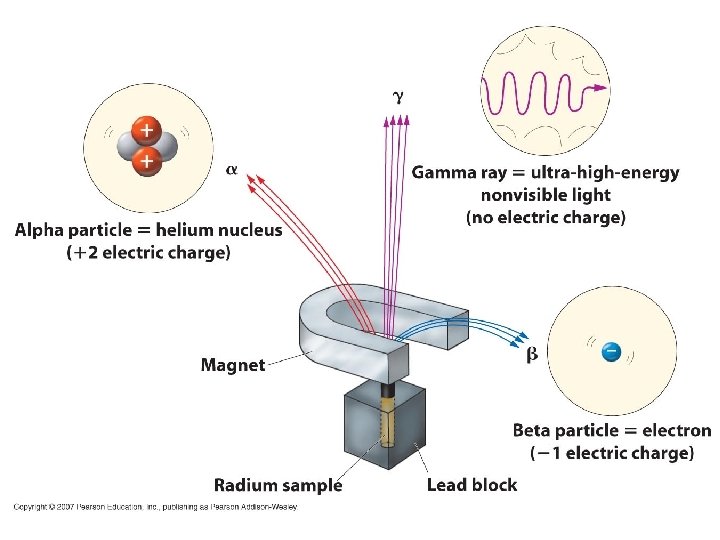
Alpha Decay Emission of alpha particles a : • • • helium nuclei two protons and two neutrons charge +2 e can travel a few inches through air can be stopped by a sheet of paper, clothing.
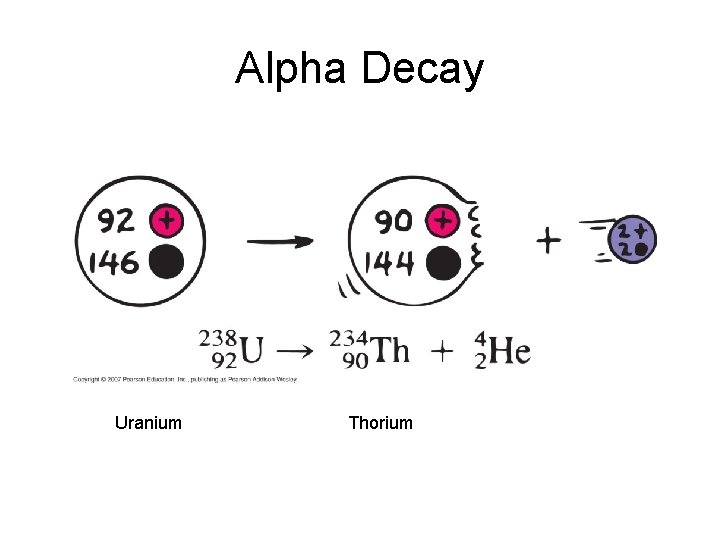
Alpha Decay Uranium Thorium
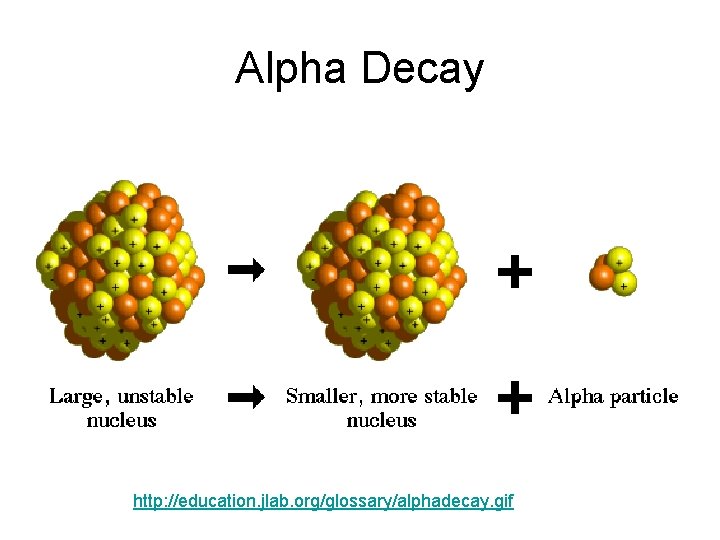
Alpha Decay http: //education. jlab. org/glossary/alphadecay. gif

Beta Decay • Beta particles b: electrons ejected from the nucleus when neutrons decay ( n -> p+ +b- ) • Beta particles have the same charge and mass as "normal" electrons.

Beta Decay • Beta particles b: electrons ejected from the nucleus when neutrons decay n -> p+ +b • Beta particles have the same charge and mass as "normal" electrons. • Can be stopped by aluminum foil or a block of wood.
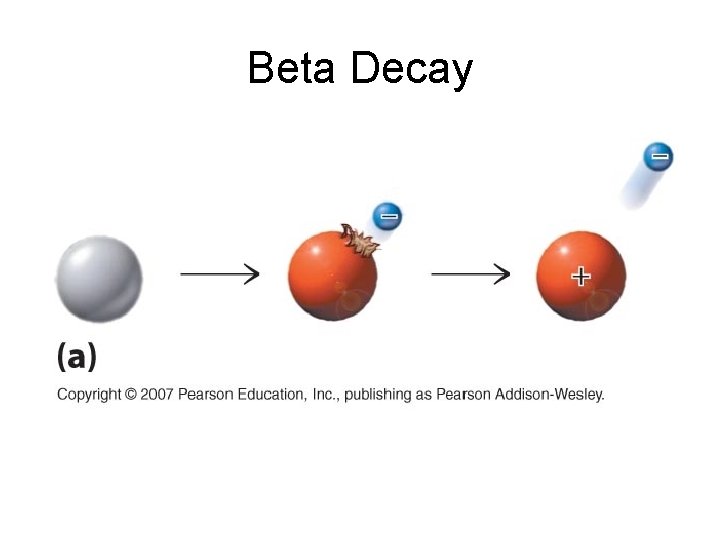
Beta Decay
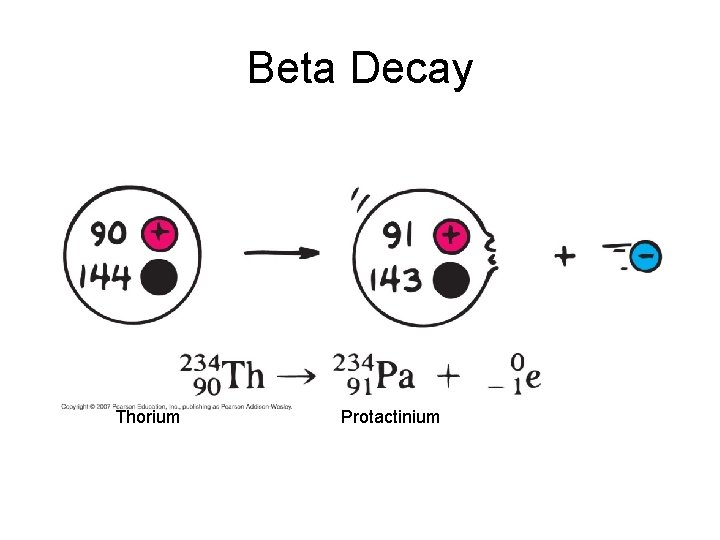
Beta Decay Thorium Protactinium

Gamma Decay • Gamma radiation g : electromagnetic energy that is released. • Gamma rays are electromagnetic waves. • They have no mass. • Gamma radiation has no charge. – Most Penetrating, can be stopped by 1 m thick concrete or a several cm thick sheet of lead.

Examples of Radioactive Decay Alpha Decay Po Pb + He Beta Decay p n + e n p + e C N + e Gamma Decay Ni + g (excited nucleus)
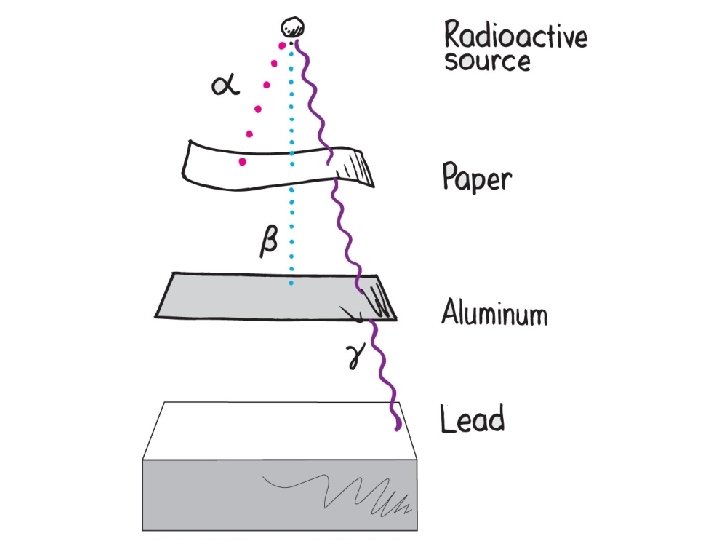

Which is more penetrating? Why?


Part II • Nuclear Stability • Half-Life

Nuclear Stability • Depends on the neutron to proton ratio.
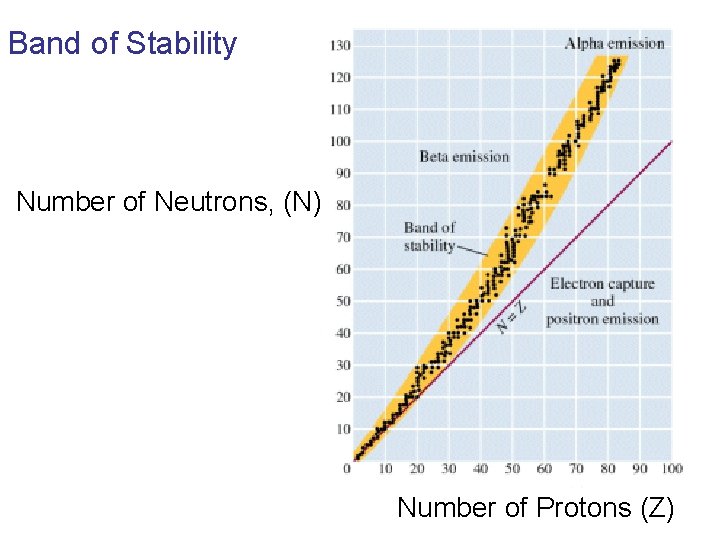
Band of Stability Number of Neutrons, (N) Number of Protons (Z)

What happens to an unstable nucleus? • They will undergo decay • The type of decay depends on the reason for the instability

What type of decay will happen if the nucleus contains too many neutrons? • Beta Decay

Example: 14 6 14 0 7 -1 C N + e In N-14 the ratio of neutrons to protons is 1: 1

• Nuclei with atomic number > 83 are radioactive

Radioactive Half-Life (t 1/2 ): • The time for half of the radioactive nuclei in a given sample to undergo decay.

Common Radioactive Isotopes Isotope Half-Life Radiation Emitted Carbon-14 5, 730 years b, g Radon-222 3. 8 days a Uranium-235 7. 0 x 108 years a, g Uranium-238 4. 46 x 109 years a

Radioactive Half-Life • After one half life there is 1/2 of original sample left. • After two half-lives, there will be 1/2 of the 1/2 = 1/4 the original sample.
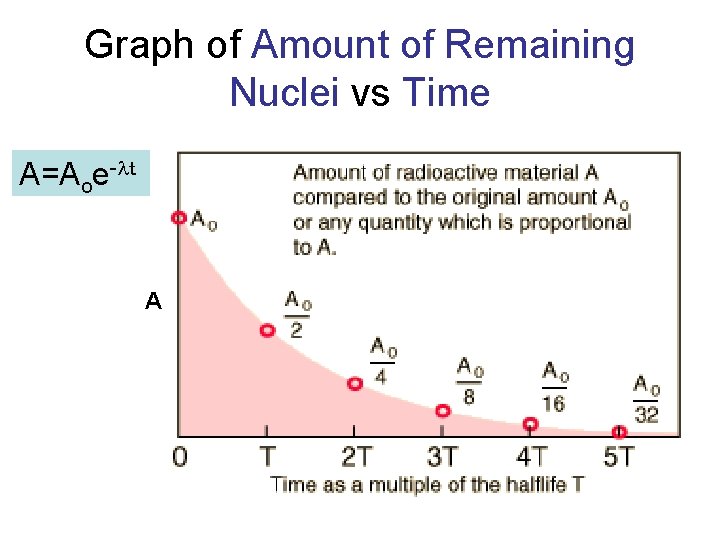
Graph of Amount of Remaining Nuclei vs Time A=Aoe-lt A

Example You have 100 g of radioactive C-14. The half -life of C-14 is 5730 years. • How many grams are left after one halflife? Answer: 50 g • How many grams are left after two halflives?

Problem A sample of 3 x 107 Radon atoms are trapped in a basement that is sealed. The half-life of Radon is 3. 83 days. How many radon atoms are left after 31 days? answer: 1. 2 x 105 atoms
- Slides: 68