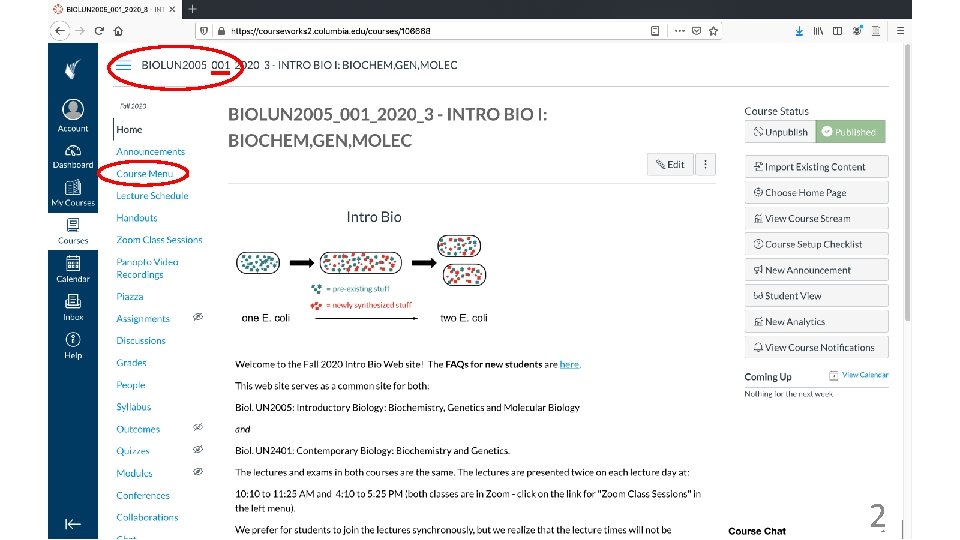
Intro Bio BIOL UN 2005 BIOL UN 2401

Intro Bio BIOL UN 2005 BIOL UN 2401 Dr. Marko Jovanovic mj 2794@columbia. edu Fairchild 913 Dr. Deborah Mowshowitz dbm 2@columbia. edu Mudd 744 D Dr. Mary Ann Price map 2293@columbia. edu Mudd 744 A
2
3
4
5
Advice page 6

Contemporary Biology Lab - UN 2501 • This course, UN 2005/UN 2401 does NOT include a lab • There is a lab course, Contemporary Biology Lab (UN 2501), but it is separate from this course • You do NOT need to take it at the same time as this course, and you may not need to take it at all 7

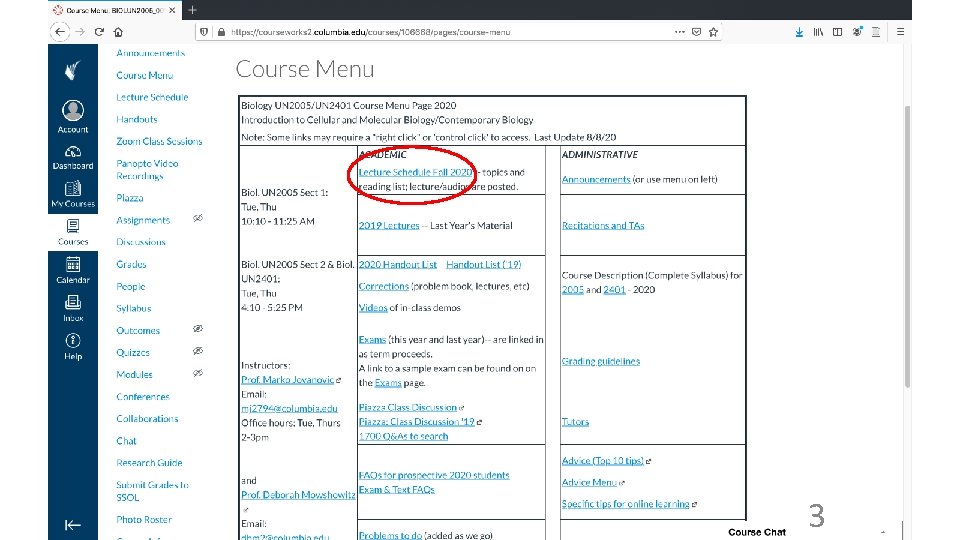
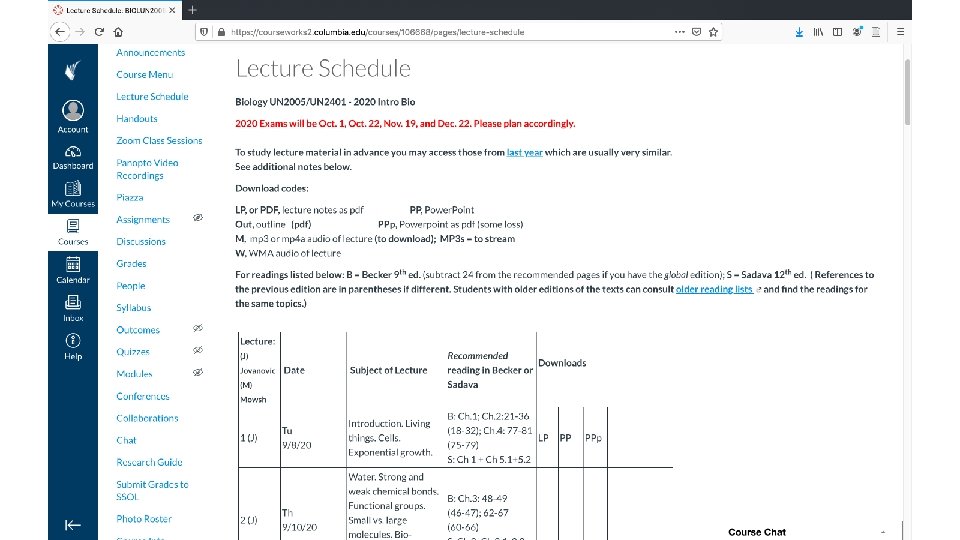
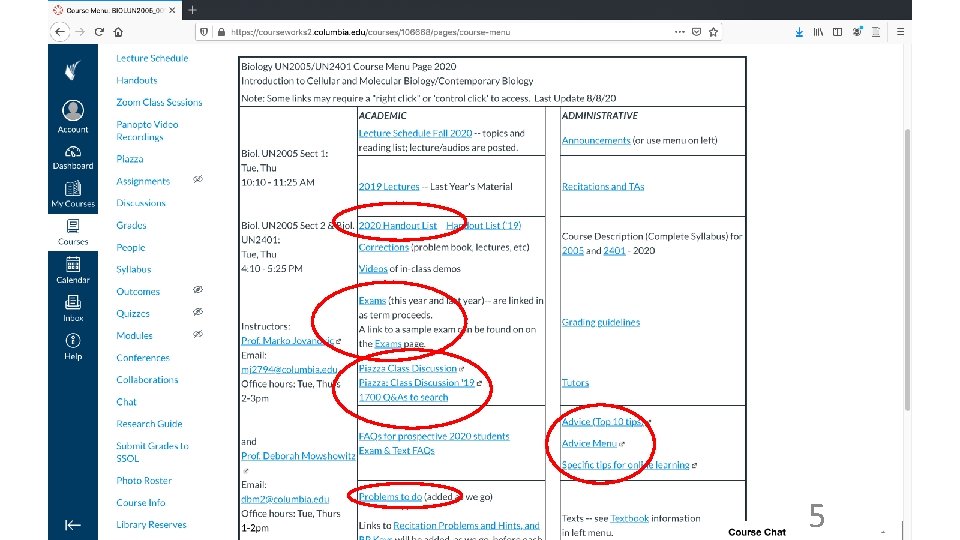
Resources for this course • Web site is required reading (at least twice a week) • Web lecture texts (includes Power. Points; handouts) • Problem book Learner’s Manual (Mowshowitz) • Texts: Becker’s World of the Cell (Hardin); Life (Sadava). Not required… • Recitations • Help (in addition to recitations/TAs): Office hours, Piazza, e-mail questions to instructors (MJ, MAP or DM) • Evening vs. morning lectures (or if you had to miss a lecture online) • Learner’s Manual • Note exam dates and times (drop an exam); note final date 8

Resources for this course, cont. • Learner’s Manual 9

Chemistry and math for this course Chemistry • • • basic atomic structure and bonding ions salts molecular weight stoichiometry chemical equilibria, equilibrium constants p. H etc. ? Math • • exponents logarithms algebra no calculus 10

Course Philosophy How does this work? Does this make sense? vs. Memorization 11

Biology – the study of life Biology – understanding of how living things work Characteristics of living things 1) Structure and composition are complex 2) Metabolism: chemical interaction with the environment 3) Reproduction: duplication of the complex, metabolizing structure 12

Characteristics of living things 1) Structure and composition are complex 2) Metabolism: chemical interaction with the environment 3) Reproduction: duplication of the complex, metabolizing structure 13

Fake plastic tree vs. a real one 1. complexity Artificial: polypropylene plastics polyester 5 dye molecules 7 different distinguishable molecules Real: > 20, 000 distinguishable molecules Each one does a job. 14

Characteristics of living things 1) Structure and composition are complex 2) Metabolism: chemical interaction with the environment 3) Reproduction: duplication of the complex, metabolizing structure 15

2. Chemical interaction with the environment dust H 2 O O 2 CO 2 NO 3 - PO 43 - Artificial Real 16

Characteristics of living things 1) Structure and composition are complex 2) Metabolism: chemical interaction with the environment 3) Reproduction: duplication of the complex, metabolizing structure 17

3. Reproduction cannot reproduce itself can reproduce itself 18

Two approaches to studying biology 1. Focus on the differences to learn about nature Darwin’s finches 19
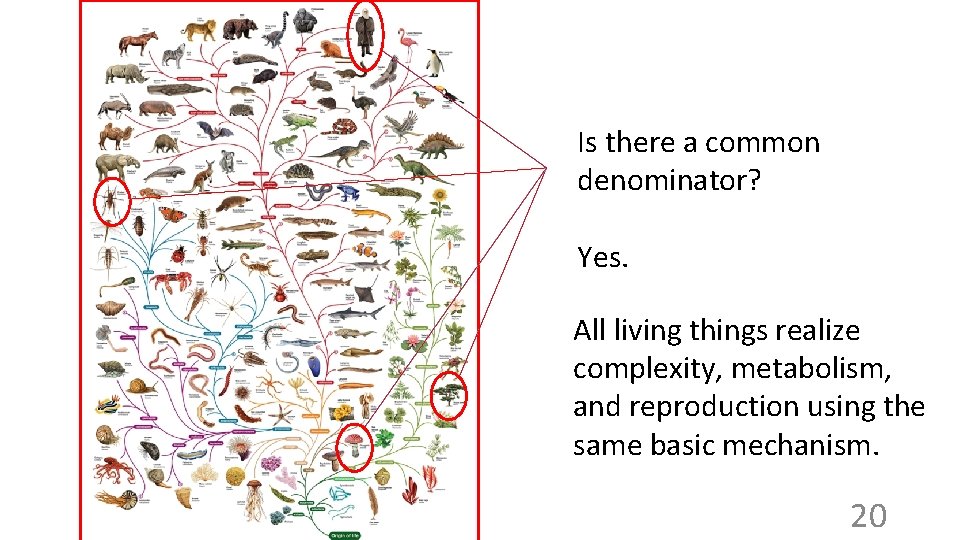
Is there a common denominator? Yes. All living things realize complexity, metabolism, and reproduction using the same basic mechanism. 20
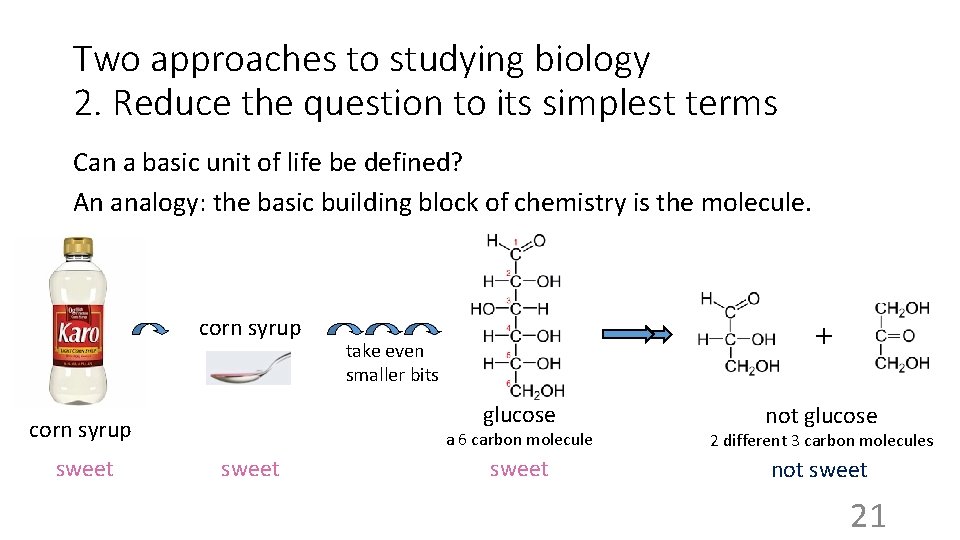
Two approaches to studying biology 2. Reduce the question to its simplest terms Can a basic unit of life be defined? An analogy: the basic building block of chemistry is the molecule. corn syrup glucose corn syrup sweet + take even smaller bits sweet not glucose a 6 carbon molecule 2 different 3 carbon molecules sweet not sweet 21
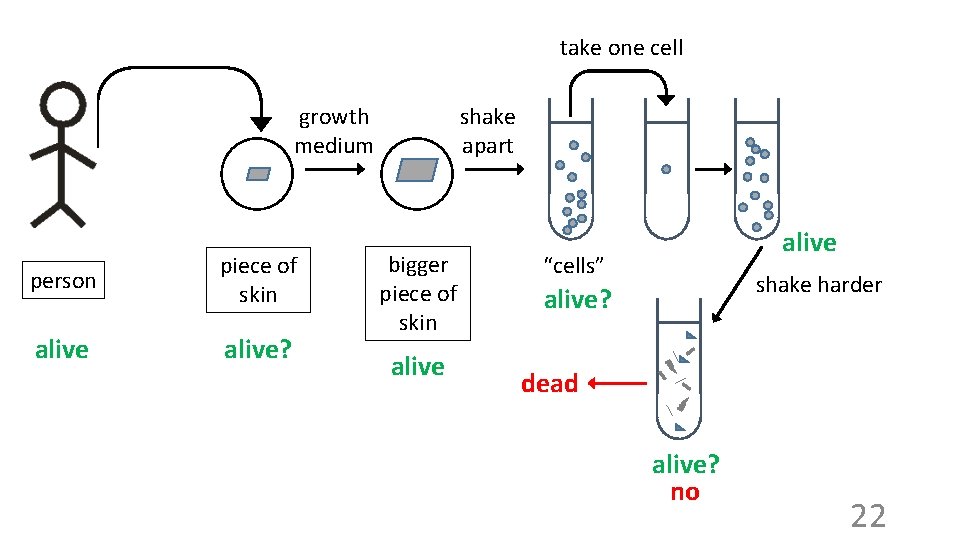
take one cell growth medium person piece of skin alive? shake apart bigger piece of skin alive “cells” shake harder alive? dead alive? no 22
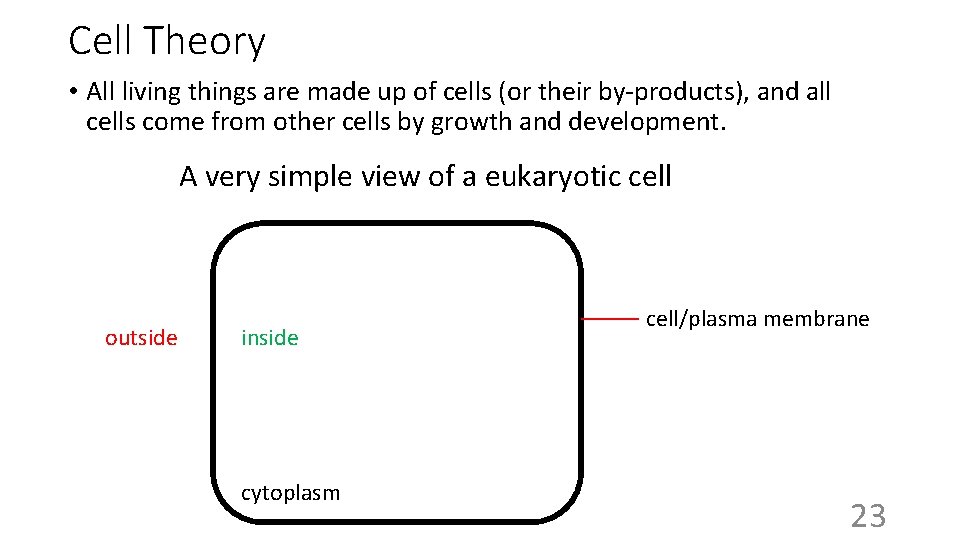
Cell Theory • All living things are made up of cells (or their by-products), and all cells come from other cells by growth and development. A very simple view of a eukaryotic cell outside inside cytoplasm cell/plasma membrane 23
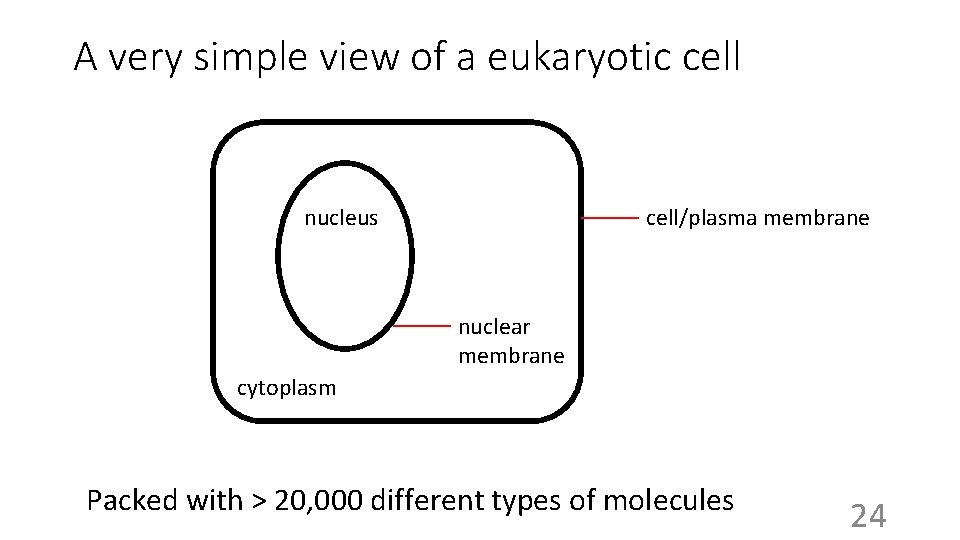
A very simple view of a eukaryotic cell nucleus cell/plasma membrane nuclear membrane cytoplasm Packed with > 20, 000 different types of molecules 24
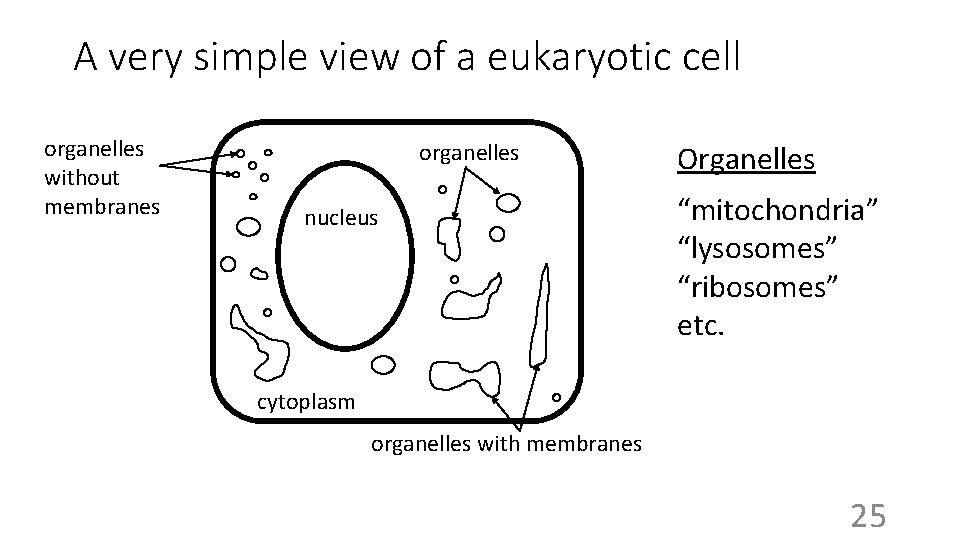
A very simple view of a eukaryotic cell organelles without membranes organelles nucleus Organelles “mitochondria” “lysosomes” “ribosomes” etc. cytoplasm organelles with membranes 25
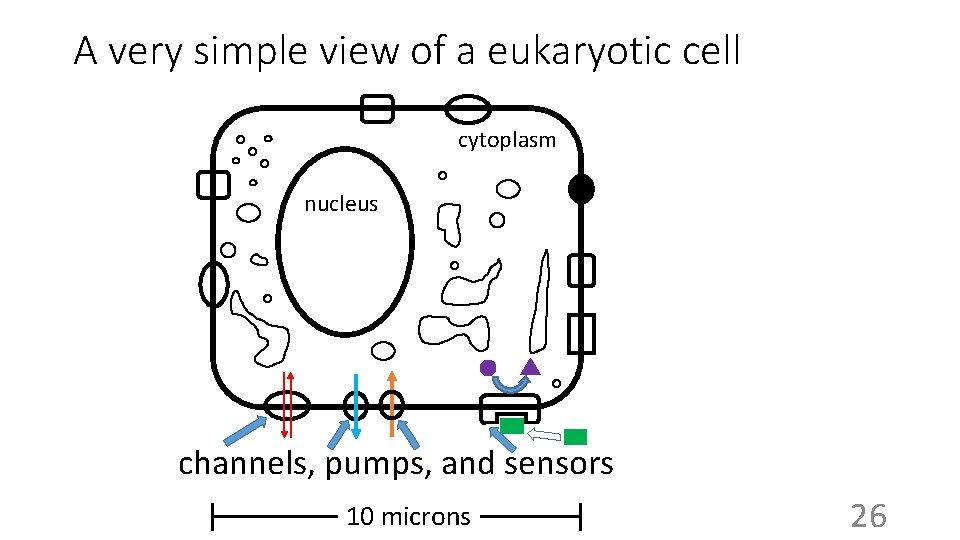
A very simple view of a eukaryotic cell cytoplasm nucleus channels, pumps, and sensors 10 microns 26

Sizes Skin cell ~ 10 micrometers (microns, mm, um, m) • Millimeter (mm) = 1/1000 of a meter (10 -3 meter), 1/10 cm • e. g. , the head of a pin • Micron (m) = 10 -6 meters (1 millionth of a meter, 1/1000 of a mm) • e. g. , smallest cells (~1/1000 volume of a skin cell) • Nanometer (nm) = 10 -9 meters (1 billionth of a meter, 1/1000 of a m) • e. g. , diameter of molecules (e. g. , glucose, 1 nm) • Angstrom (Å, A) = 1/10 of a nm) • e. g. , distance between 2 atoms in a molecule 27
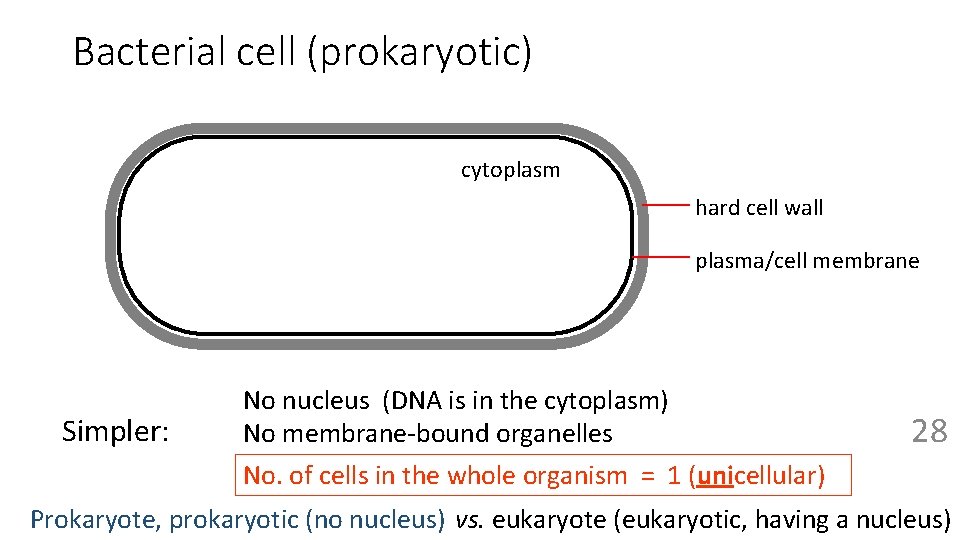
Bacterial cell (prokaryotic) cytoplasm hard cell wall plasma/cell membrane Simpler: No nucleus (DNA is in the cytoplasm) No membrane-bound organelles No. of cells in the whole organism = 1 (unicellular) 28 Prokaryote, prokaryotic (no nucleus) vs. eukaryote (eukaryotic, having a nucleus)
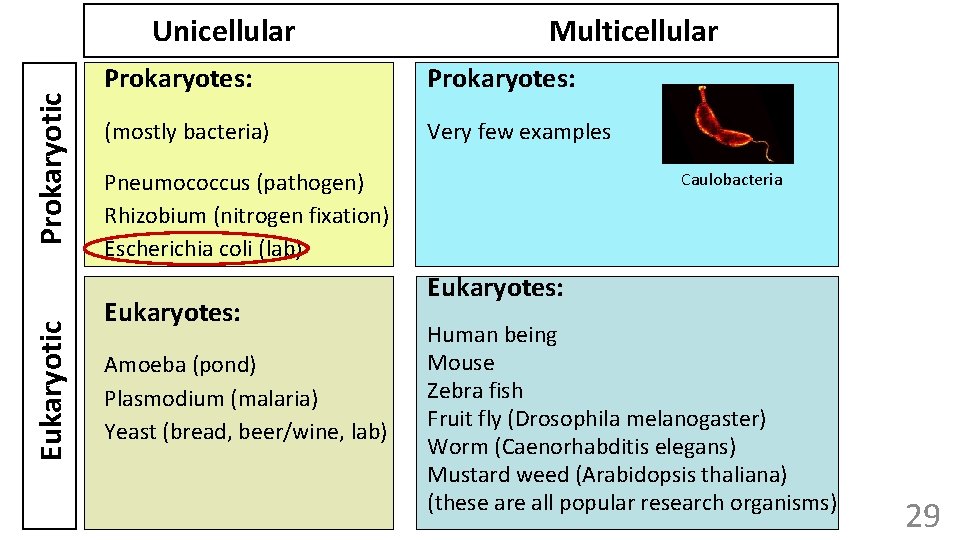
Eukaryotic Prokaryotic Unicellular Multicellular Prokaryotes: (mostly bacteria) Very few examples Pneumococcus (pathogen) Rhizobium (nitrogen fixation) Escherichia coli (lab) Eukaryotes: Amoeba (pond) Plasmodium (malaria) Yeast (bread, beer/wine, lab) Caulobacteria Eukaryotes: Human being Mouse Zebra fish Fruit fly (Drosophila melanogaster) Worm (Caenorhabditis elegans) Mustard weed (Arabidopsis thaliana) (these are all popular research organisms) 29
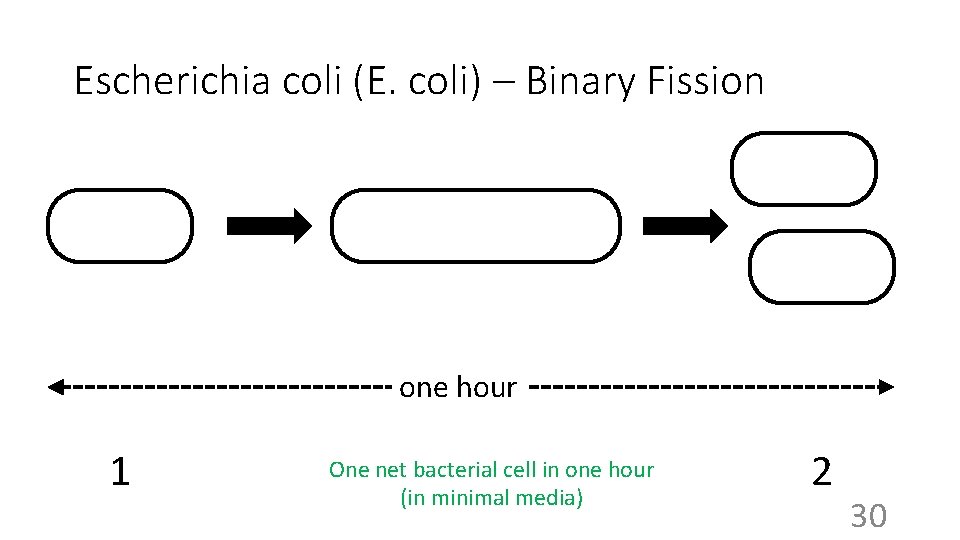
Escherichia coli (E. coli) – Binary Fission one hour 1 One net bacterial cell in one hour (in minimal media) 2 30
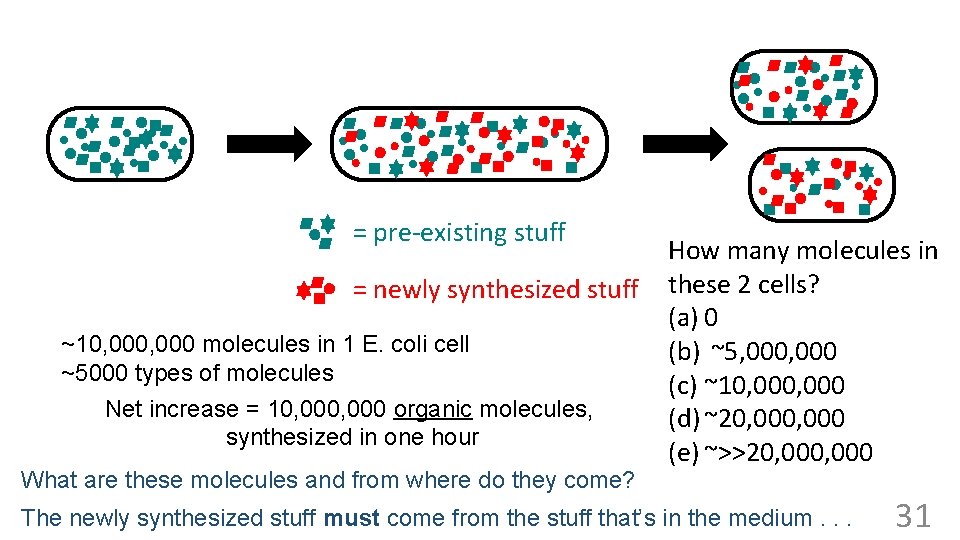
= pre-existing stuff = newly synthesized stuff ~10, 000 molecules in 1 E. coli cell ~5000 types of molecules Net increase = 10, 000 organic molecules, synthesized in one hour What are these molecules and from where do they come? How many molecules in these 2 cells? (a) 0 ~20, 000 molecules (b) ~5, 000 in 2 cells (c) ~10, 000 types of molecules Same (d) ~20, 000 ~5000 (e) ~>>20, 000 The newly synthesized stuff must come from the stuff that’s in the medium. . . 31

A minimal growth medium for E. coli C 6 H 12 O 6 glucose, a sugar C-source: the 5000 types of carbon containing molecules? KH 2 PO 4 potassium phosphate Mg. SO 4 magnesium sulfate NH 4 Cl ammonium chloride H 2 O water +trace elements (e. g. , Zn, Fe, Cu, Se, … ) 32
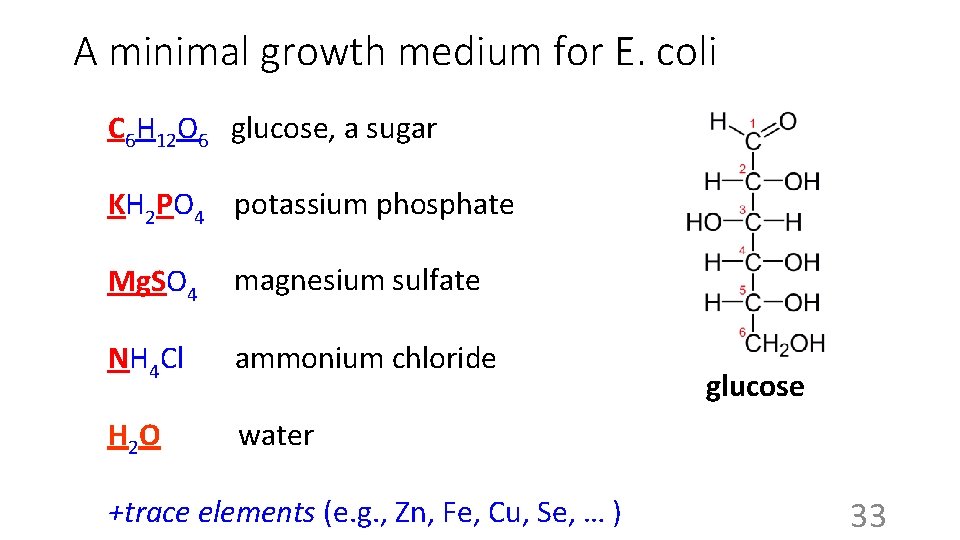
A minimal growth medium for E. coli C 6 H 12 O 6 glucose, a sugar KH 2 PO 4 potassium phosphate Mg. SO 4 magnesium sulfate NH 4 Cl ammonium chloride glucose H 2 O water +trace elements (e. g. , Zn, Fe, Cu, Se, … ) 33
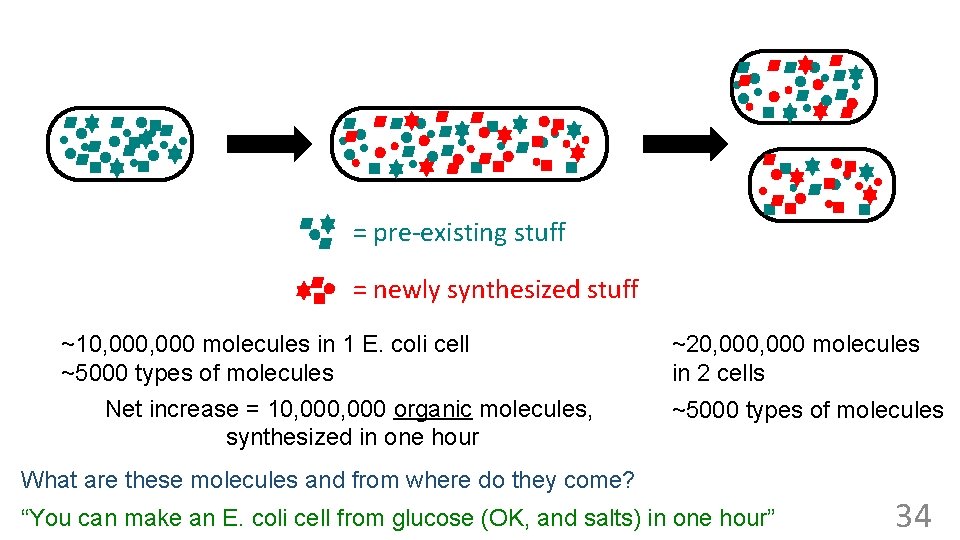
= pre-existing stuff = newly synthesized stuff ~10, 000 molecules in 1 E. coli cell ~5000 types of molecules Net increase = 10, 000 organic molecules, synthesized in one hour ~20, 000 molecules in 2 cells ~5000 types of molecules What are these molecules and from where do they come? “You can make an E. coli cell from glucose (OK, and salts) in one hour” 34
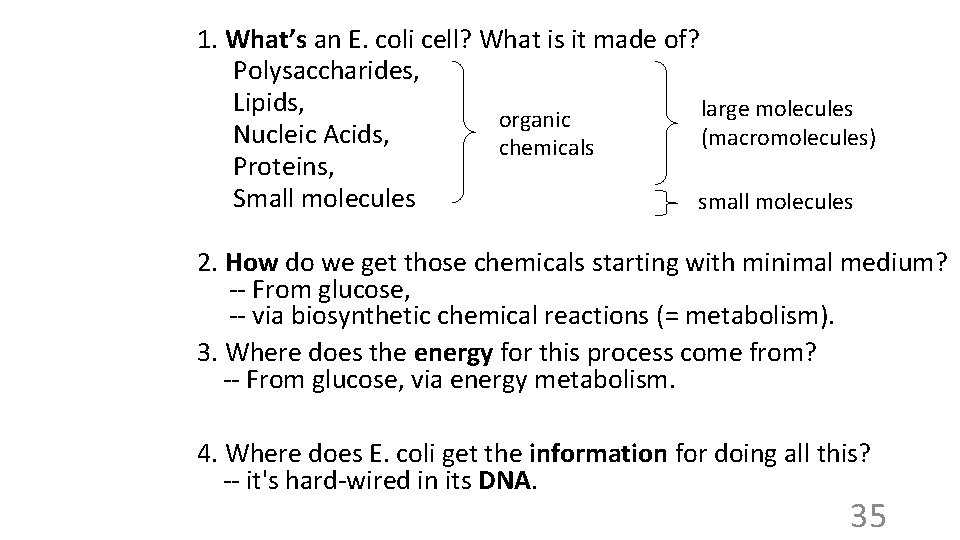
1. What’s an E. coli cell? What is it made of? Polysaccharides, Lipids, large molecules organic Nucleic Acids, (macromolecules) chemicals Proteins, Small molecules small molecules 2. How do we get those chemicals starting with minimal medium? -- From glucose, -- via biosynthetic chemical reactions (= metabolism). 3. Where does the energy for this process come from? -- From glucose, via energy metabolism. 4. Where does E. coli get the information for doing all this? -- it's hard-wired in its DNA. 35
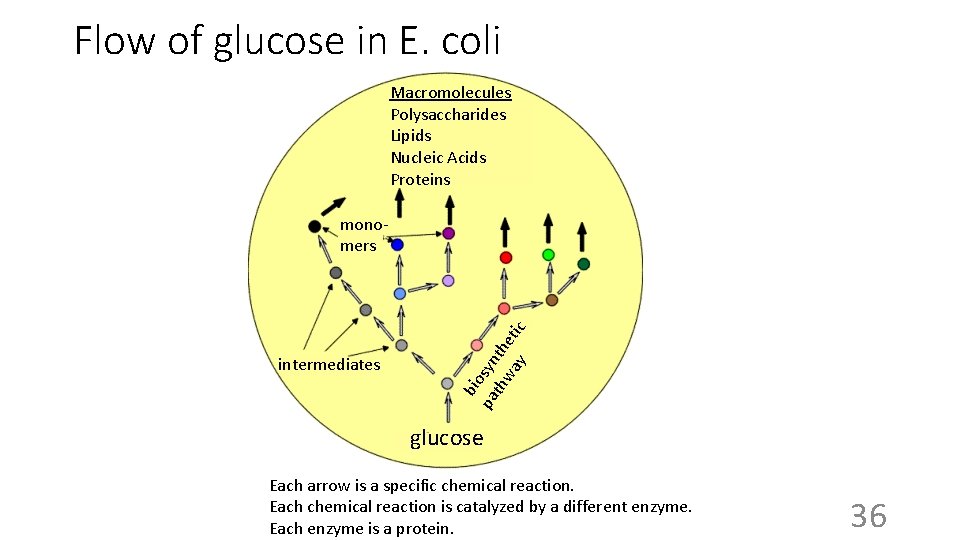
Flow of glucose in E. coli Macromolecules Polysaccharides Lipids Nucleic Acids Proteins intermediates bio pa syn th th wa et y ic monomers glucose Each arrow is a specific chemical reaction. Each chemical reaction is catalyzed by a different enzyme. Each enzyme is a protein. 36
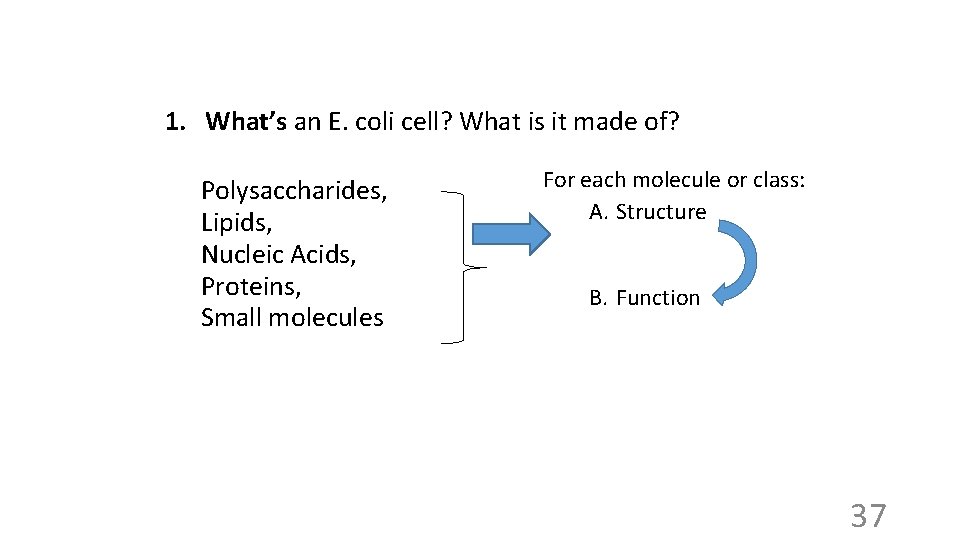
1. What’s an E. coli cell? What is it made of? Polysaccharides, Lipids, Nucleic Acids, Proteins, Small molecules For each molecule or class: A. Structure B. Function 37
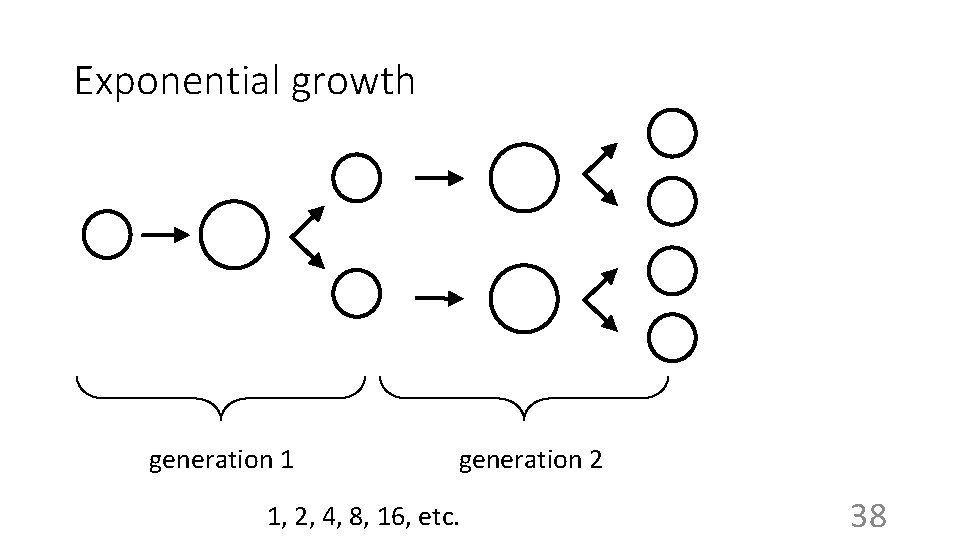
Exponential growth generation 1 generation 2 1, 2, 4, 8, 16, etc. 38

Exponential growth • N = 1 x 2 g • N = 100 x 2 g • N = No x 2 g For a more detailed explanation, see handout on exponential growth. • g = t/t. D where t. D = the doubling time, or generation time. • N = No 2 t/t. D • k= 1/t. D, then N = No 2 kt One can change the base of the exponential relation: N = Noeke t, ln(N/No) = ke t; if N/No=2, t=t. D: ke= ln(2)/t. D =0. 69/t. D N =No 10 k 10 t, log(N/No) =k 10 t; if t=t. D, N/No=2: k 10=log(2)/t. D= 0. 3/t. D 39
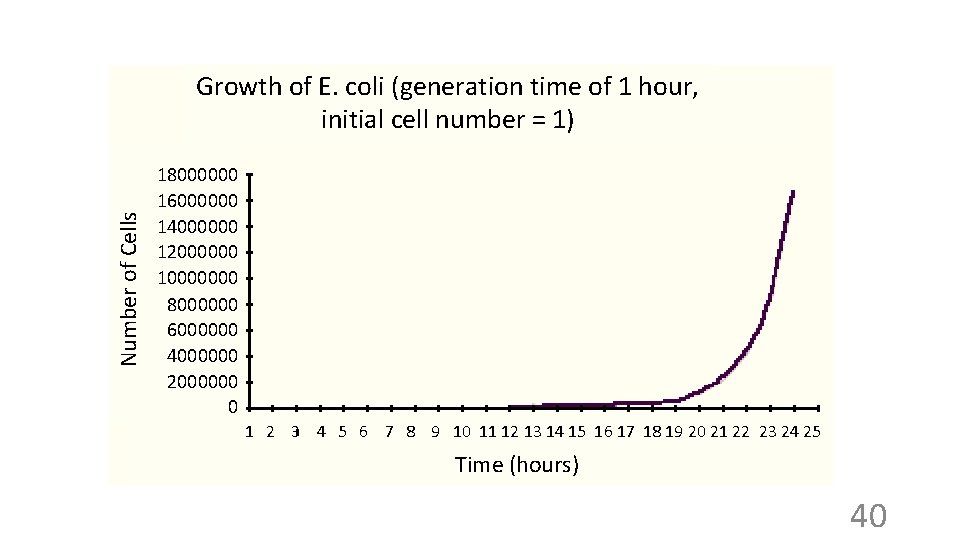
Number of Cells Growth of E. coli (generation time of 1 hour, initial cell number = 1) 18000000 16000000 14000000 12000000 10000000 8000000 6000000 4000000 2000000 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Time (hours) 40
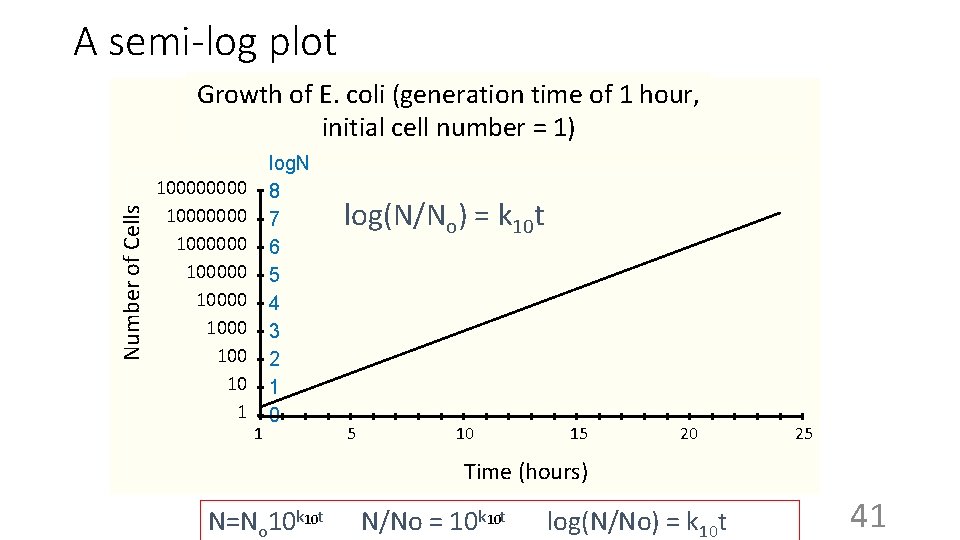
A semi-log plot Number of Cells Growth of E. coli (generation time of 1 hour, initial cell number = 1) 10000000 100000 1000 10 1 1 log. N 8 7 6 5 4 3 2 1 0 log(N/No) = k 10 t 5 10 15 20 25 Time (hours) N=No 10 k 10 t 10 N/No = 10 k t log(N/No) = k 10 t 41

Log of cell number E. coli cultures in real life “stationary” phase “lag” phase Exponential growth: “log” phase (linear on a semi-log plot) Time Note: The cells do not have to be dividing synchronously to generate these curves, and usually are not. We are just seeing the average for the whole population. 42
- Slides: 42