Intrinsic Optical Activity Environmental Perturbations Solvation Effects in
Intrinsic Optical Activity & Environmental Perturbations: Solvation Effects in Chiral Building Blocks Paul M. Lemler, Clayton L. Craft, & Patrick H. Vaccaro Department of Chemistry, Yale University 225 Prospect Street New Haven, CT 06520 -8107 USA
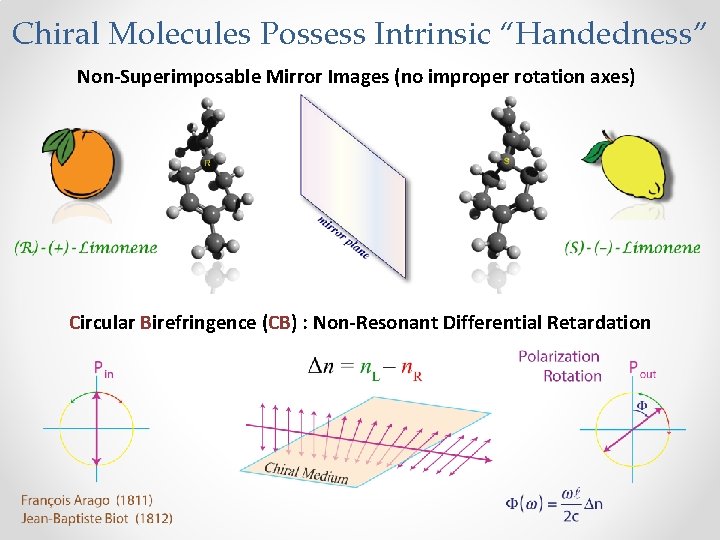
Chiral Molecules Possess Intrinsic “Handedness” Non-Superimposable Mirror Images (no improper rotation axes) Circular Birefringence (CB) : Non-Resonant Differential Retardation
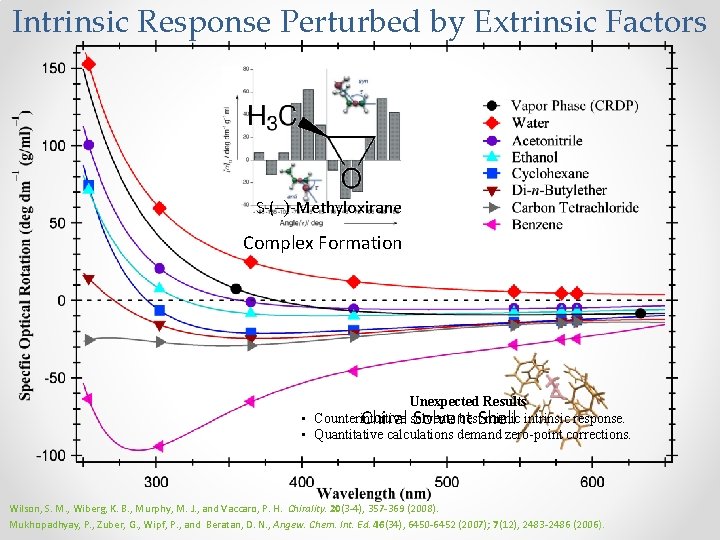
Intrinsic Response Perturbed by Extrinsic Factors S-(–)-Methyloxirane Complex Formation Unexpected Results • Counterintuitive best. Shell mimic intrinsic response. Chiral solvents Solvent • Quantitative calculations demand zero-point corrections. Wilson, S. M. , Wiberg, K. B. , Murphy, M. J. , and Vaccaro, P. H. Chirality. 20(3 -4), 357 -369 (2008). Mukhopadhyay, P. , Zuber, G. , Wipf, P. , and Beratan, D. N. , Angew. Chem. Int. Ed. 46(34), 6450 -6452 (2007); 7(12), 2483 -2486 (2006).
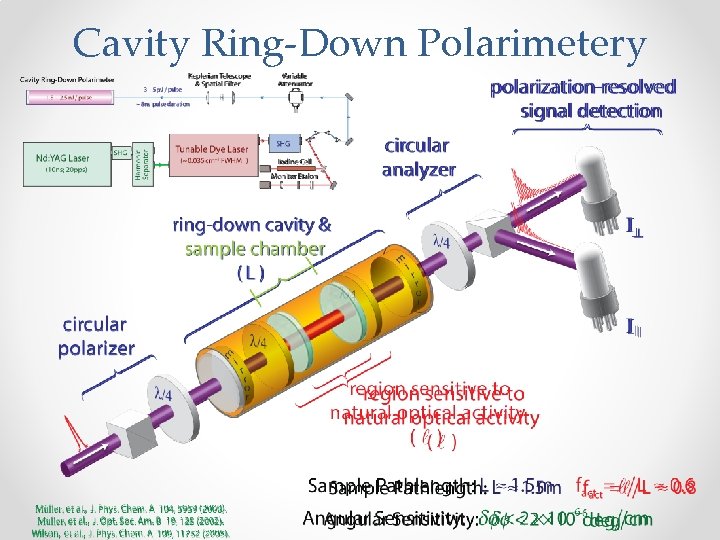
Cavity Ring-Down Polarimetery
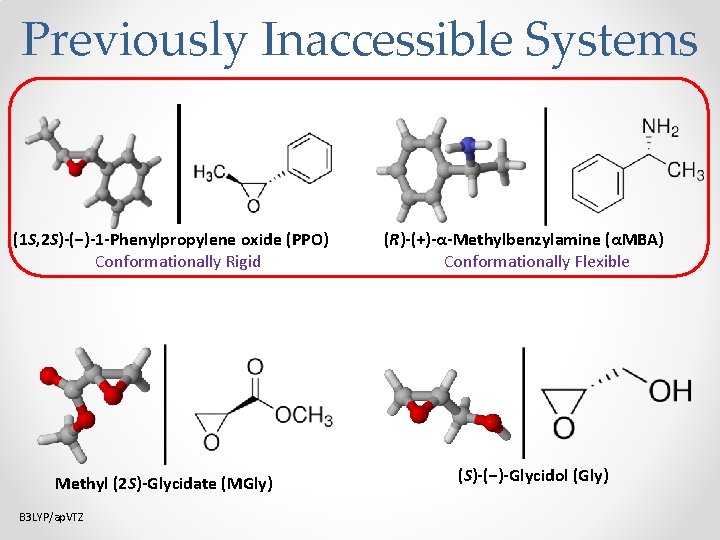
Previously Inaccessible Systems (1 S, 2 S)-(−)-1 -Phenylpropylene oxide (PPO) Conformationally Rigid Methyl (2 S)-Glycidate (MGly) B 3 LYP/ap. VTZ (R)-(+)-α-Methylbenzylamine (αMBA) Conformationally Flexible (S)-(−)-Glycidol (Gly)

Solvation Models Explicit Implicit P olarizable C ontinuum M odel • Solute-solvent system modeled as a supramolecular complex • Solvent modeled as a continuum dielectric (ε) that surrounds the solute cavity • Accurate treatment of solute-solvent interaction in first solvation shell • Neglects important first-shell effects in cybotactic region • Poor description of long-range • Accurate description of long-range interactions and electrostatic polarization electrostatic solute-solvent interactions • Computationally expensive with cost rising dramatically with size of system • Computationally expedient owing to the reduction in effective degrees of freedom

Summary and Conclusions Ø Cavity Ring-Down Polarimetry (CRDP) affords a sensitive means for interrogating dispersive (CB) optical activity under rarefied conditions. Ø Modification of sample vessel has enabled hitherto inaccessible families of chiral molecules with new chemical functionalities to be investigated: αMBA and PPO. Ø Solvation can influence optical activity in pronounced and counterintuitive ways, modifying both magnitude and sign of the response evoked from rigid molecules. Ø Conformational flexibility can modify optical activity grossly, leading to structural isomers that often possess antagonistic physicochemical and chiroptical properties. Ø Additional theoretical & experimental work is needed to understand solution-phase chiroptical response, especially for cases where specific solute-solvent interactions or chiral imprinting of solvation shells plays an important role. Supported by U. S. National Science Foundation
- Slides: 7