Intraperitoneal chemotherapy for epithelial ovarian cancer HuaHis Wu





















































- Slides: 74

Intraperitoneal chemotherapy for epithelial ovarian cancer Hua-His Wu, MD OB/GYN, VGH-TPE Mar 5 th, 2007

Epithelial ovarian cancer o Standard therapy n o o o A maximum cytoreductive surgery followed by combination chemotherapy with paclitaxel and carboplatin A chemo-sensitive tumor However, most recur Intraperitoneal spreading Mar 5 th, 2007 H. H. Wu, MD

History of IP C/T o Weisberger 1955 n o Jones 1978 n o Nitrogen mustard intraperitoneally for malignant ascites signicantly greater concentrations of certain chemotherapeutic drugs in the peritoneal cavity than in the blood. SWOG/GOG n n n The first phase III trial since 1980 s, presented in 1996 In favor of IP arm Mar 5 th, 2007 H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD

NCI announcement 2006 o o Encouraging the GO community to consider IP chemotherapy as the standard treatment for optimally debulked advanced ovarian cancer patients Based on a meta-analysis of three US trials and other phase III studies Mar 5 th, 2007 H. H. Wu, MD

However, IP chemotherapy is still regarded as controversial issue. Why Mar 5 th, 2007 H. H. Wu, MD

IP Chemotherapy o Principles o Pharmacology o Clinical aspects o Toxicities and QOL o Future directions Mar 5 th, 2007 H. H. Wu, MD

Principles of IP C/T Mar 5 th, 2007 H. H. Wu, MD

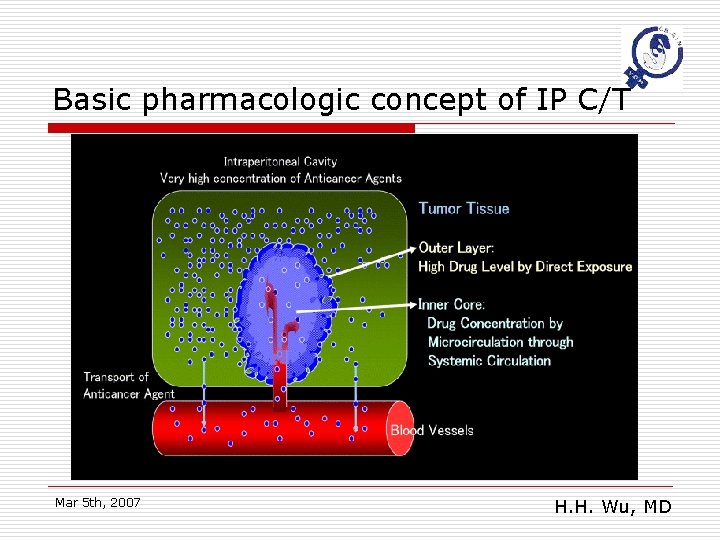
Basic pharmacologic concept of IP C/T Mar 5 th, 2007 H. H. Wu, MD

What is the ideal anticancer agent for IP C/T? o o Very effective systemically against ovarian cancer Penetrate deep into the tumor Stays in the peritoneal cavity for prolonged period Low incidence of systemic adverse effect but providing satisfactory drug concentrations in the inner core of tumor Mar 5 th, 2007 (有效 夠深 留得久) H. H. Wu, MD

Basic concept of IP C/T o o o Penetration Peritoneal dwelling Solute transport model Anatomy of the peritoneum and capillary vessels Resistance to solute transport Mar 5 th, 2007 H. H. Wu, MD

Penetration of anticancer agents o Doxorubicin n o n By osteosarcoma spheroids and autoradiographs (West et al) Limited ability in avascular tumor mass & ≧ 250 μm in dia. Vinblastine & 5 -FU n n o (Ozols et al; Durand et al) Methotrexate n o 4 -6 layers In glioma spheroids (Nederman and Carlsson)) Penetration : 5 -FU > vinblastine Cisplatin n n In mouse model Concentration o o Mar 5 th, 2007 in peripheral: In center : (Los et al) IP > IV IP = IV H. H. Wu, MD

Peritoneal dwelling of anticancer drugs o o Longer stay of anticancer agents Higher drug concentration in the inner core Is a contrary phenomenon Mar 5 th, 2007 H. H. Wu, MD

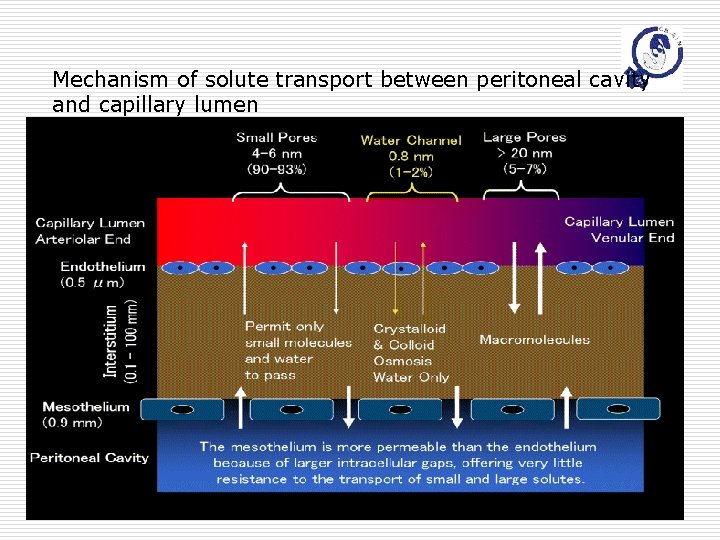
Anatomy of the peritoneum o Primary interface between abdominal cavity & vessels o Parietal peritoneum (10%) & visceral peritoneum (90%) o o The area is approximately to the body surface area (1. 0 -2. 0 cm 2) Components n Mesothelium n Basement membrane n Interstitium n Microcirculation n Visceral lymphatics Mar 5 th, 2007 H. H. Wu, MD

Mesothelium, Interstitium o Mesothelium n n n o Monolayer of flattened cells about 0. 5 mm thick Tight junction ; Gap junction Absence of tight junction in the subdiaphragmatic area directly absorbed into the lymphatic system Interstitium n n The supporting structure Distance varies Mar 5 th, 2007 H. H. Wu, MD

Blood vessels o Visceral peritoneum n n o Parietal peritoneum n o Supplied by celiac and mesentary arteries with venous drainage via the portal vein Rapid firstpass metabolism by the liver Supplied by circumflex iliac, lumbar, intercostal, and epigastric arteries with venous drainage via the systemic circulation. Effective peritoneal surface area n n The density of the number of perfused capillaries The number and the size of pores within the capillaies Mar 5 th, 2007 H. H. Wu, MD

Peritoneal lymphatics o Extensive in the subdiaphragmatic area n n n o o stoma exist, basement membrane absent Little resistance for the solute transport Also present in parietal and visceral peritoneum To maintain the relatively small volume of fluid (50100 ml) Mar 5 th, 2007 H. H. Wu, MD

Mechanism of solute transport between peritoneal cavity and capillary lumen Mar 5 th, 2007 H. H. Wu, MD

Theoretical behaviors of anticancer agents o o Larger molecular weight or water-insoluble anticancer drugs stay longer in the peritoneal cavity Smaller molecular weight or water-soluble can go into the inner core but stay shorter in the cavity Small molecular weight agents that are metabolized in the liver to become active form should not be used for IP C/T. Small molecular weight agents with already active form are suitable for IP C/T Mar 5 th, 2007 H. H. Wu, MD

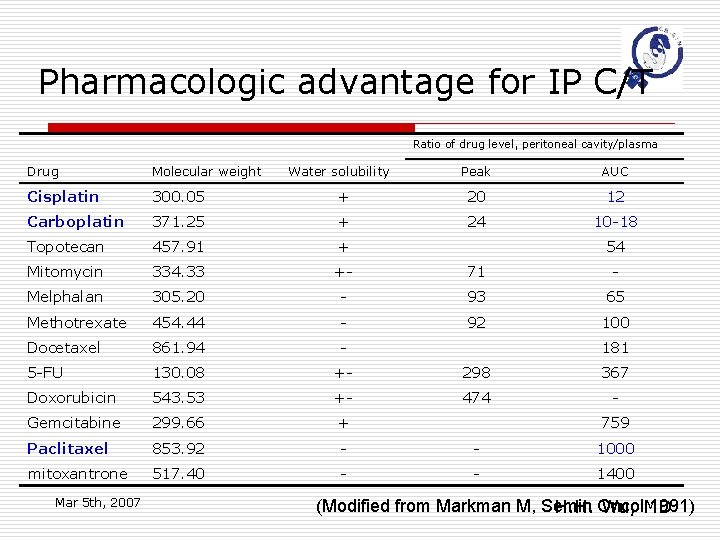
Pharmacologic advantage for IP C/T Ratio of drug level, peritoneal cavity/plasma Drug Molecular weight Cisplatin 300. 05 Carboplatin Peak AUC + 20 12 371. 25 + 24 10 -18 Topotecan 457. 91 + Mitomycin 334. 33 +- 71 - Melphalan 305. 20 - 93 65 Methotrexate 454. 44 - 92 100 Docetaxel 861. 94 - 5 -FU 130. 08 +- 298 367 Doxorubicin 543. 53 +- 474 - Gemcitabine 299. 66 + Paclitaxel 853. 92 - - 1000 mitoxantrone 517. 40 - - 1400 Mar 5 th, 2007 Water solubility 54 181 759 (Modified from Markman M, Semin 1991) H. H. Oncol Wu, MD

Choice of drugs o o If the IP C/T is considered to be a regional therapy paclitaxel, mitoxantrone If the IP C/T is hypothesized as a route of systemic chemotherapy platinum agents Mar 5 th, 2007 H. H. Wu, MD

Pharmacology of IP drugs o Cisplatin o Carboplatin o Paclitaxel Mar 5 th, 2007 H. H. Wu, MD

Cisplatin o o P/V ratio: peak 21; AUC 12 (Howell, 1982) The mode of administration did not affect systemic toxicity (Pretorius, 1981) The amount of drug recovered in the urine and the drug levels within the tissues were similar The peritoneal lining had 2. 5 -8 times higher levels of drug after IP administration IP C/T might increase therapeutic index for small tumors confined to the peritoneal cavity Mar 5 th, 2007 H. H. Wu, MD

Carboplatin o After 4 hrs dwelling, P/V ratio: n Peak: 24; AUC 10 (Elferink, o 1998) Pharmacologic study after IP and IV 2005) n n (Miyagi, 24 -hr free platinum AUC in the serum is identical 24 -hr free platinum AUC in the peritoneal cavity was 17 times higher when which given via IP infusion of carboplatin is feasible not only as an IP regional therapy but also as a more reasonable route for systemic chemotherapy o The recommended dose of IP carboplatin was 400 mg/m 2 (Speyer and Sorich, 1992) Mar 5 th, 2007 H. H. Wu, MD

Paclitaxel o o o Dose-limiting toxicity: severe abdominal pain (when dose ≧ 175 mg/㎡) P/V ratio: peak & AUC : 1000 -fold Paclitaxel persisted in peritoneum for more than 24 -48 h after a single IP instillation (Markman, 1992) o Very slow peritoneal clearance (at dose level ≧ 60 mg/㎡, it can persist more than 1 wk with significant level wkly IP Taxol ) o Low plasma concentration Mar 5 th, 2007 (Francis, 1995) H. H. Wu, MD

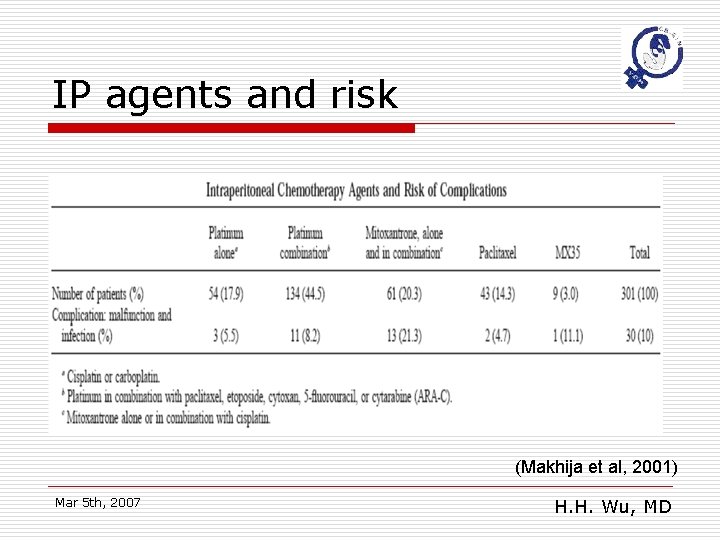
IP agents and risk (Makhija et al, 2001) Mar 5 th, 2007 H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD

Strengths of IP C/T o o Achieve dose intensification (as ‘high-dose’) Treats both intraperitoneal tumor bed and extraperitoneal tumor via systemic recirculation Reaches IP sites that may not be reached by IV route, especially when up to 2 L dialysate are administered Onion skinning effect – IP cisplatin can penetrate as far as 4 mm into surface of IP tumors(by definition, <1 cm in size) and up to 6 repeated administrations Mar 5 th, 2007 H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD

Clinical aspects of IP C/T o Front-line chemotherapy o Consolidation o 2 nd-line chemotherapy Mar 5 th, 2007 H. H. Wu, MD

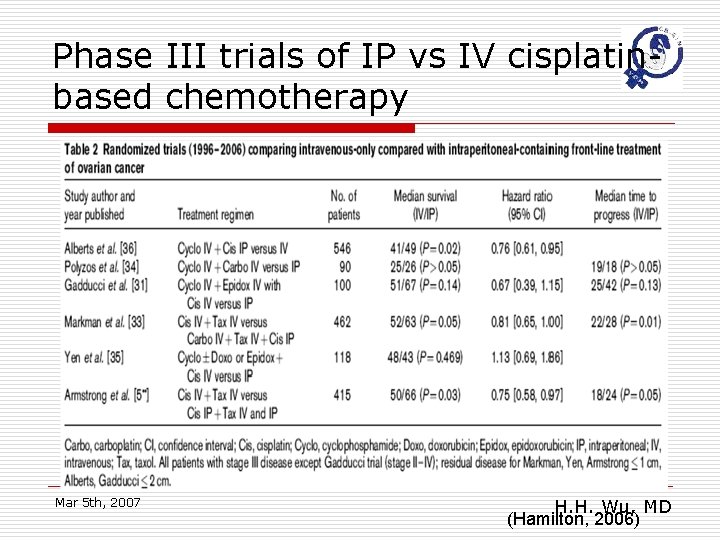
Phase III trials of IP vs IV cisplatinbased chemotherapy Mar 5 th, 2007 H. H. Wu, MD (Hamilton, 2006)

Mar 5 th, 2007 H. H. Wu, MD

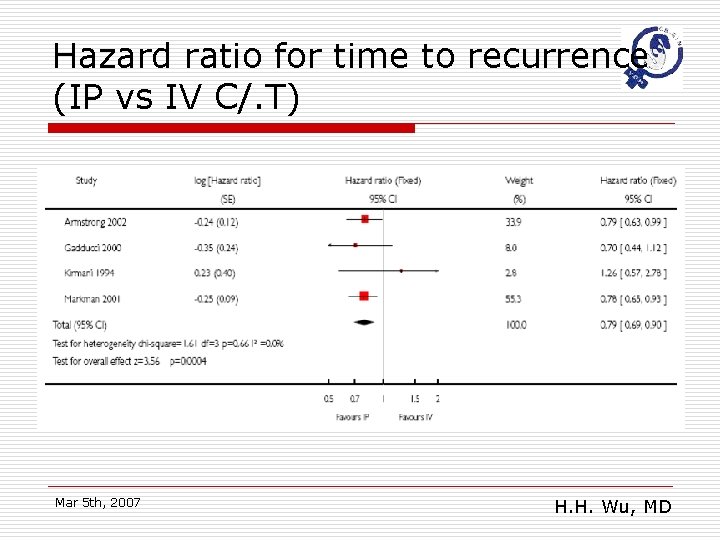
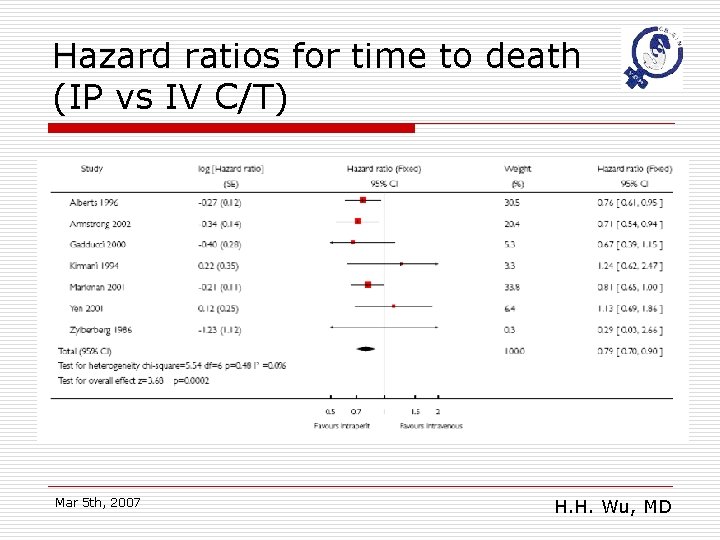
Main results o o o Eight randomized trials studied 1819 women receiving primary treatment for ovarian cancer. Women were less likely to die if they received an intraperitoneal (IP) component to the chemotherapy (hazard ratio (HR) =0. 79; 95% confidence interval (CI): 0. 70 to 0. 90)and the disease free interval (HR =0. 79; 95%CI: 0. 69 to 0. 90) was also significantly prolonged. There may be greater serious toxicity with regard to gastrointestinal effects, effects pain and fever but less ototoxicity with the intraperitoneal than the intravenous route. Mar 5 th, 2007 H. H. Wu, MD

Hazard ratio for time to recurrence (IP vs IV C/. T) Mar 5 th, 2007 H. H. Wu, MD

Hazard ratios for time to death (IP vs IV C/T) Mar 5 th, 2007 H. H. Wu, MD

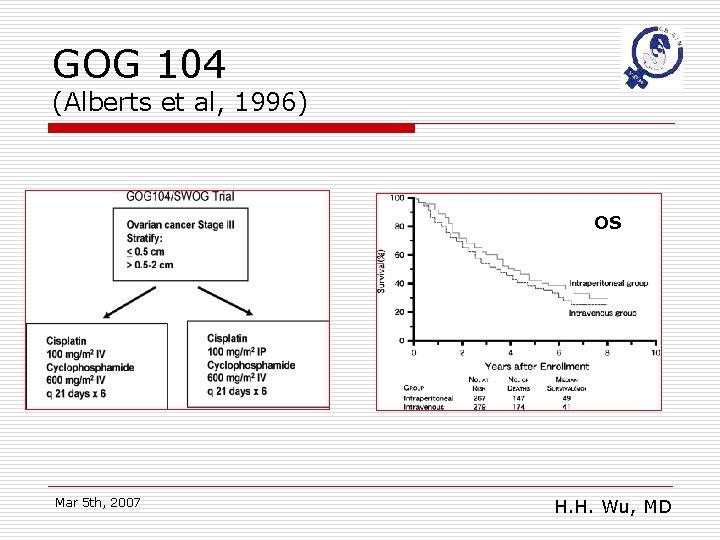
GOG 104 (Alberts et al, 1996) OS Mar 5 th, 2007 H. H. Wu, MD

GOG 104: conclusions o o As compared with IV cisplatin, IP cicplatin significantly improves survival and has significantly lower toxic effects in patients with stage III ovarian cancer and residual tumor mass of 2 cm or less. The only same “dose-intensity” in both arms phase 3 RCT Mar 5 th, 2007 H. H. Wu, MD

Shorts of GOG 104 o GOG 111 n Median survival from 24 months (P+C) to 38 months ( P+T) Mar 5 th, 2007 H. H. Wu, MD

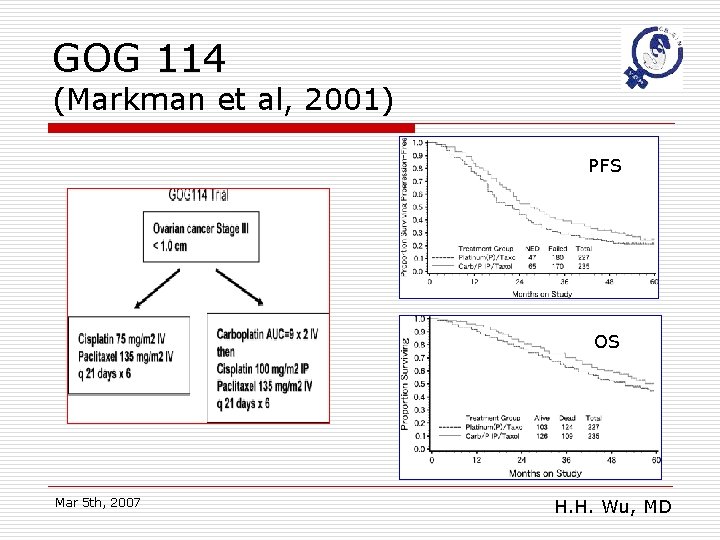
GOG 114 (Markman et al, 2001) PFS OS Mar 5 th, 2007 H. H. Wu, MD

GOG 114: conclusions o o o The 2 nd phase 3 RCT to show IP cisplatin is superior to IV cisplatin in small volume residual advanced ovarian cancer The 1 st phase 3 trial in ovarian cancer to a median survival of >5 years Trial demonstrated that IP cisplatin favorably impacts survival, even through IV paclitaxel is a component of regimen Mar 5 th, 2007 H. H. Wu, MD

Shorts of GOG 114 o More complications in IP arm n n o Neutropenia, thrombocytopenia G-I & metabolic toxicities Carbopltin x 2 cycles ( AUC 9) Mar 5 th, 2007 H. H. Wu, MD

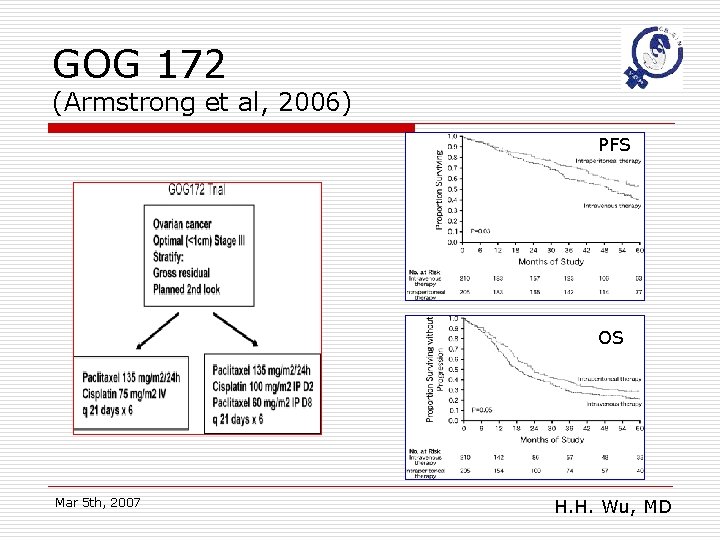
GOG 172 (Armstrong et al, 2006) PFS OS Mar 5 th, 2007 H. H. Wu, MD

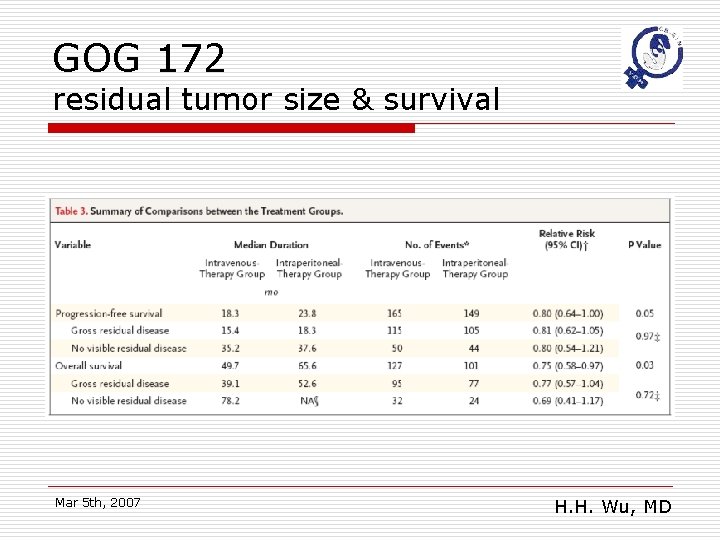
GOG 172 residual tumor size & survival Mar 5 th, 2007 H. H. Wu, MD

GOG 172: conclusions o o Significantly survival benefit in IP arm The 65. 6 months median survival is the longest survival reported to date from a randomized trial in advanced ovarian cancer Mar 5 th, 2007 H. H. Wu, MD

Shorts of GOG 172 o o o The IP regimen uses higher and more frequent dosing than the IV regimen Toxicities were greater on the IP arm Fewer patients on the IP arm were able to complete 6 cycles of therapy Mar 5 th, 2007 H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD

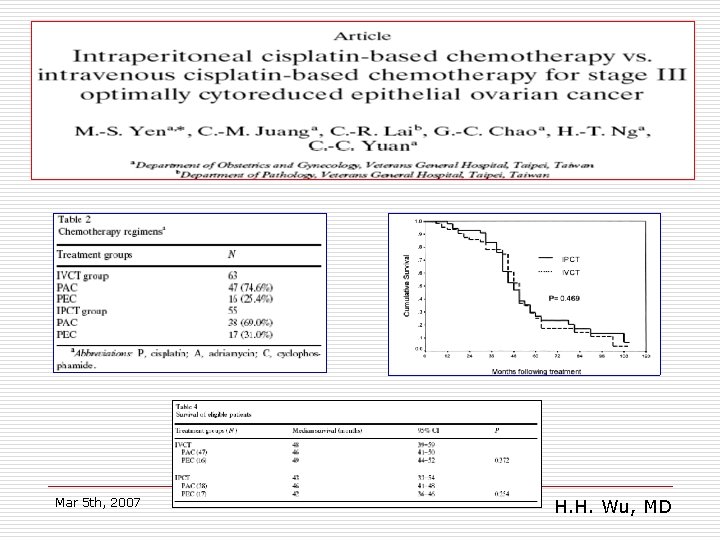
VGH-TPE: conclusions o o Intravenous and intraperitoneal chemotherapy are associated with equivalent survival in patients with minimal residual stage III epithelial ovarian cancer after optimal cytoreductive surgery (<1 m). PEC or PAC regimens Mar 5 th, 2007 H. H. Wu, MD

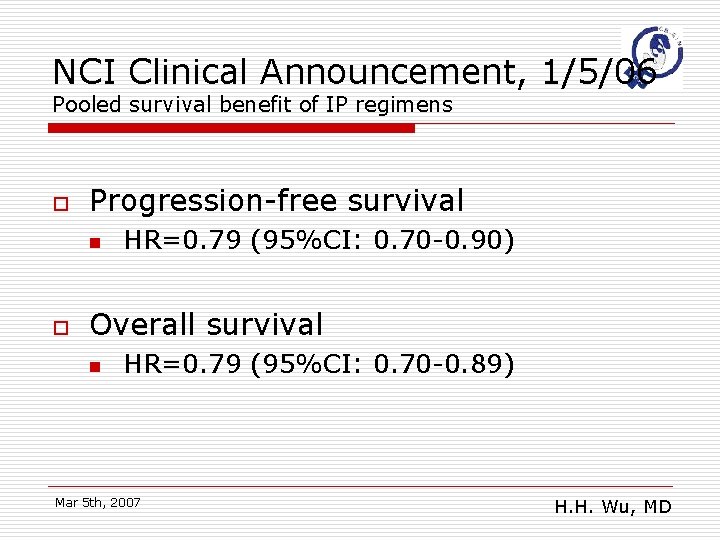
NCI Clinical Announcement, 1/5/06 Pooled survival benefit of IP regimens o Progression-free survival n o HR=0. 79 (95%CI: 0. 70 -0. 90) Overall survival n HR=0. 79 (95%CI: 0. 70 -0. 89) Mar 5 th, 2007 H. H. Wu, MD

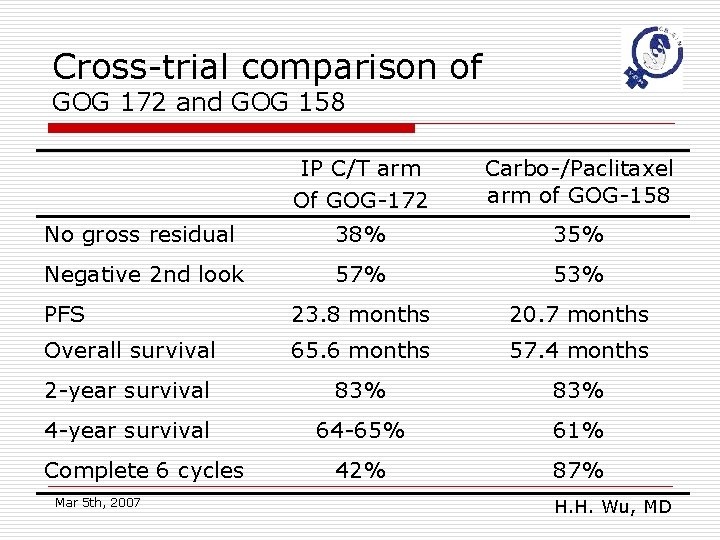
New problems o The role of carboplatin n GOG 158 (non-inferiority test) GOG 114 (moderately high dose IV Carboplatin before IP C/T) Cross-trial GOG 172 vs GOG 158 o How many courses of IP C/T is adequate? o Effect of Dose intensity? n IP regimen uses higher and more frequent dosing schedule than the IV regimen Mar 5 th, 2007 H. H. Wu, MD

Cross-trial comparison of GOG 172 and GOG 158 IP C/T arm Of GOG-172 Carbo-/Paclitaxel arm of GOG-158 No gross residual 38% 35% Negative 2 nd look 57% 53% PFS 23. 8 months 20. 7 months Overall survival 65. 6 months 57. 4 months 2 -year survival 83% 4 -year survival 64 -65% 61% 42% 87% Complete 6 cycles Mar 5 th, 2007 H. H. Wu, MD

GOG 172: eligible patients in IP arm o Although fewer than half the patients assignedto the IP group received six cycles of IP treatment, the group as a whole had a significant improvement in survival as compared with the intravenous group. It is possible that most of the benefit of IP therapy occurs early, during the initial cycles, or that the benefit of IP therapy may be greater if more patients can successfully complete six cycles of treatment. Mar 5 th, 2007 H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD

IP C/T as Consolidation Mar 5 th, 2007 H. H. Wu, MD (Hamilton, 2006)

Potential IP consolidation regimens o Cisplatin alone (50 mg/m 2) o Cisplatin + topotecan o Cisplatin + FUDR Mar 5 th, 2007 H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD

IP C/T as 2 nd-line C/T o Phase I or II studies o IP C/T is safe, feasible, and pharmacokinetically advantageous, but responses varied widely. o Critical factors for response n n o Tumor burden at initial treatment Paltinum sensitivity Few candidates for 2 nd-line IP C/T n n n Mar 5 th, 2007 Those with stage IV, macroscopic, platinum-resistant, or extraperitoneal dz are less likely to be benefit Extensive adhesion 2 nd-look op become rare recurrence is detected by palpable or imageable lesions and symptoms. H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD

More Considerations o Catheter issues o Patient selection o Toxicity and QOL Mar 5 th, 2007 H. H. Wu, MD

Complications of Catheter o o o Blockade Leakage Infection Diarrhea Bowel perforation Fistula formation Mar 5 th, 2007 H. H. Wu, MD

Catheter issues Timing of placement o o o 34% discontinued IP C/T for catheterspecific complications (Walker et al, GO, 2006) Not associated with complication rate Pre-operative counseling, if possible Laparotomy, laparoscopy Close the vaginal cuff Mar 5 th, 2007 H. H. Wu, MD

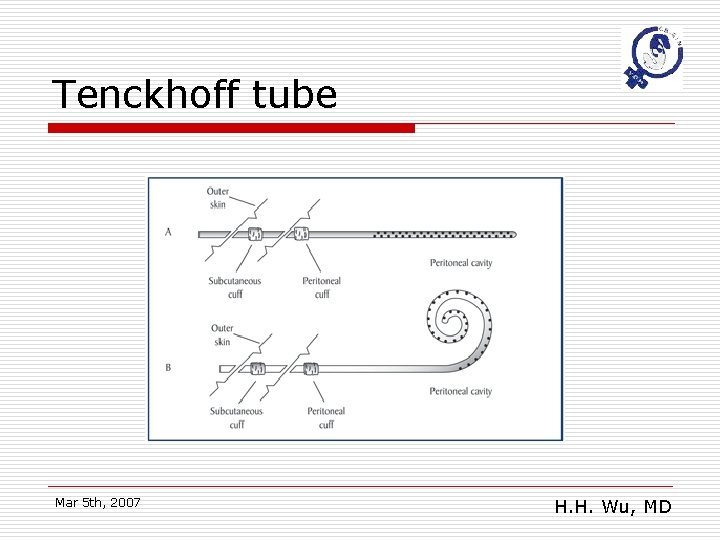
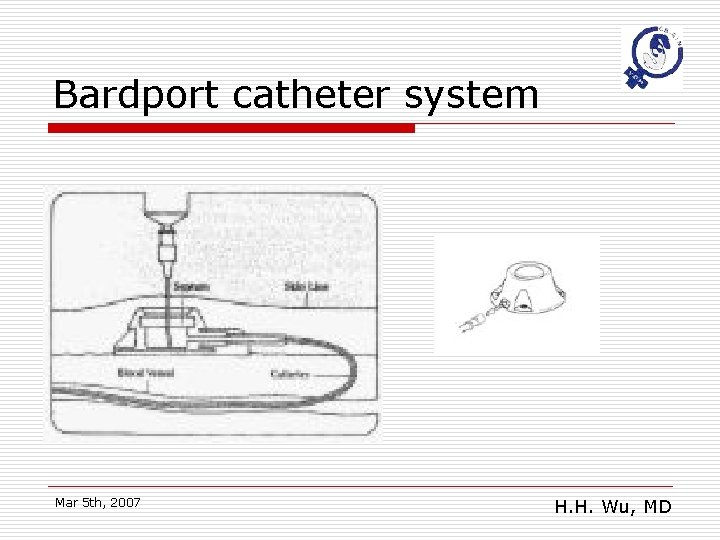
Catheter issues Types of Catheter o o Tenckhoff peritoneal dialysis catheter Subcutaneous port implantation n n o o Port-A-cath Bard. Port peritoneal catheter system JP, CWV catheters Veress needles Mar 5 th, 2007 H. H. Wu, MD

Tenckhoff tube Mar 5 th, 2007 H. H. Wu, MD

Bardport catheter system Mar 5 th, 2007 H. H. Wu, MD

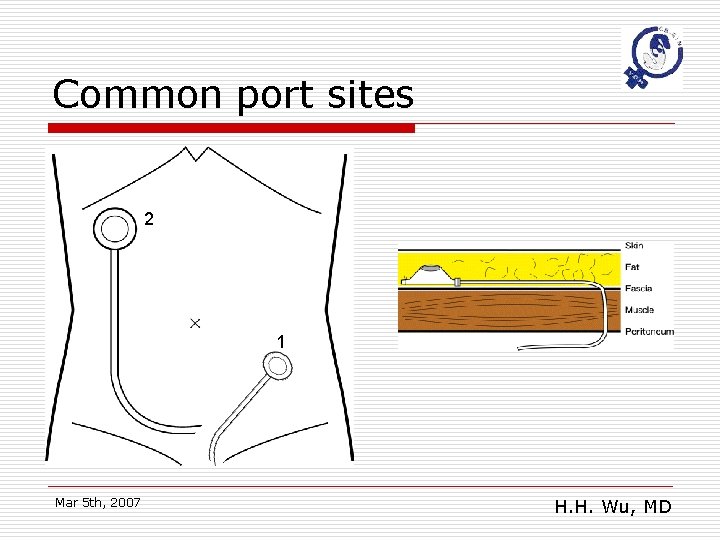
Catheter issues Site of port placement o Goal n n o To minimize patient discomfort, and Facilitate ease of access Port site n n Superior and medial to the iliac crest, or On the inferior thorax, at the midclavicular line, overlying the ribs. Mar 5 th, 2007 H. H. Wu, MD

Common port sites 2 1 Mar 5 th, 2007 H. H. Wu, MD

Patient selection issues o Patient characteristics n o o o Significant peritoneal adhesion Ongoing abdominal infection, or indwelling IP catheter becomes infected or malfunction, will be unable to treated by this route of drug delivery Size of residual tumor masses n o eg. : renal function ; neuropathy (DM – associated) <0. 5 cm, 1 cm, or 2 cm ? Onion skinning Lt colon or rectosigmoid colon resection ? Mar 5 th, 2007 H. H. Wu, MD

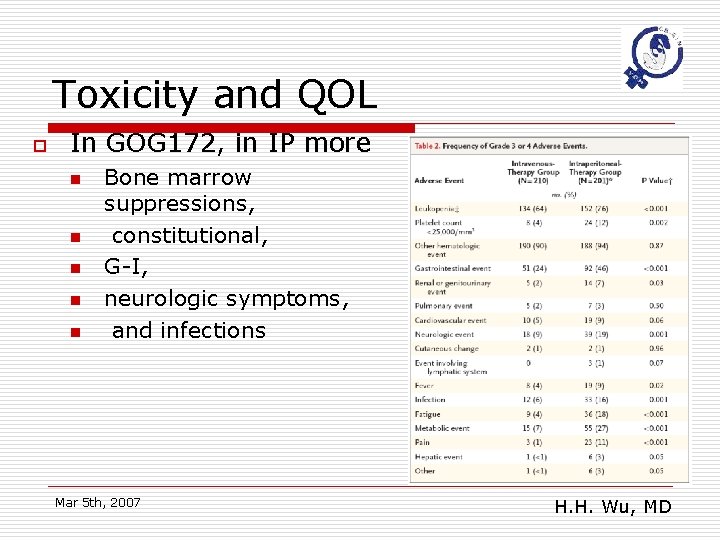
Toxicity and QOL o In GOG 172, in IP more n n n Bone marrow suppressions, constitutional, G-I, neurologic symptoms, and infections Mar 5 th, 2007 H. H. Wu, MD

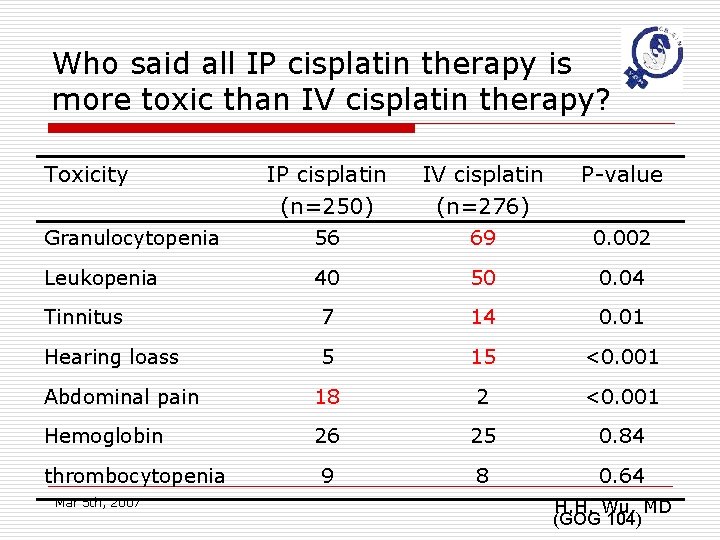
Who said all IP cisplatin therapy is more toxic than IV cisplatin therapy? Toxicity IP cisplatin (n=250) IV cisplatin (n=276) P-value Granulocytopenia 56 69 0. 002 Leukopenia 40 50 0. 04 Tinnitus 7 14 0. 01 Hearing loass 5 15 <0. 001 Abdominal pain 18 2 <0. 001 Hemoglobin 26 25 0. 84 9 8 0. 64 thrombocytopenia Mar 5 th, 2007 H. H. Wu, MD (GOG 104)

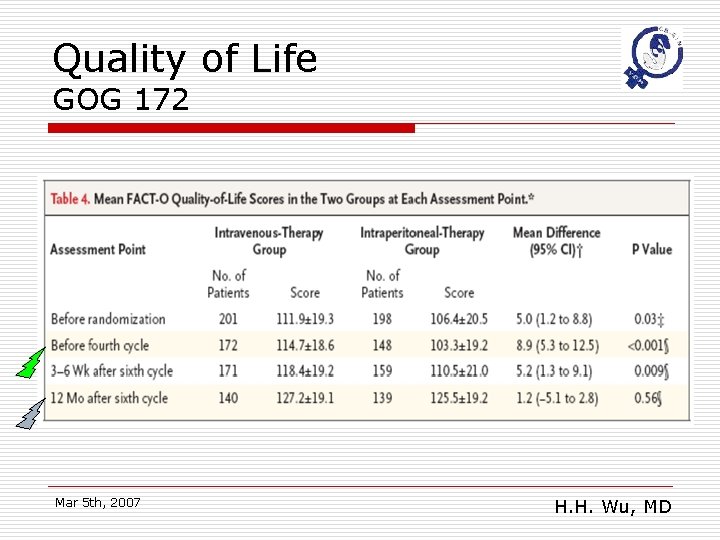
Quality of Life GOG 172 Mar 5 th, 2007 H. H. Wu, MD

How to reduce the toxicities from IP C/T? o IP cisplatin-related toxicites n n o Replacing cisplatin with carboplatin GOG phase I study: IP carboplatin (AUC 6 -7) + IV Taxol (175 mg/m 2, 3 hr) IP Paclitaxel-related toxicities n IV Docetaxel: less neurotoxic than Taxol (SCOTROC trial) n IP Docetaxel no dose-limiting toxicities (Morgan et al) o IP catheter-related toxicities Mar 5 th, 2007 H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD

Conclusion o o IP cisplatin-based C/T has been shown to have a survival benefit over IV cisplatinbased C/T for advance ovarian cancer patients with optimal debulking. However, there a number of unanswered questions that should be resolved before IP C/T becomes truly a standard care in the ovarian cancer. Mar 5 th, 2007 H. H. Wu, MD

Future Directions 1. Is IP administration of carboplatin replacable to IP cisplatin as a less toxic alternative? 2. Is IP administration of paclitaxel necessary or IP administration of docetaxel acceptable? 3. What is the optimal number of IP treatment? 4. What is the optimal timing for the IP catheter placement and what is the optimal type and material? 5. Is IP C/T for ovarian cancer with bulky residual tumor as effective as those for small residual tumor? 6. How effective is IP C/T for retroperitoneal lymph node metastasis? metastasis Mar 5 th, 2007 H. H. Wu, MD

Mar 5 th, 2007 H. H. Wu, MD