intramolecular intermolecular forces Forces of attractionrepulsion between molecules
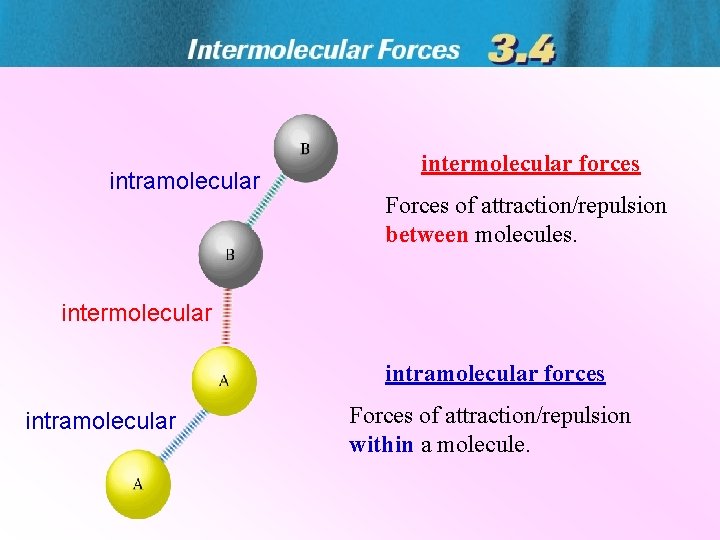
intramolecular intermolecular forces Forces of attraction/repulsion between molecules. intermolecular intramolecular forces intramolecular Forces of attraction/repulsion within a molecule.

Van der Waals Forces This is the name given to the many different weak intermolecular forces. Dipole-Dipole Force J. Van der Waals (1837 – 1923) Negative ends of polar molecules are attracted to positive ends of neighbouring polar molecules. In a liquid, molecules can orient themselves to maximize attraction and minimize repulsion.

London Force (also called: London dispersion) This force is caused by the attraction of electrons to other molecules’ nuclei. It is caused by a brief shift in electrons from one side of an atom/molecule to another, called a momentary dipole. The greater the number of electrons in the atoms of a molecule, the greater the dispersion forces. Fritz London (1900 – 1954)

Using Dipole-Dipole and London Forces to Predict Boiling Points Boiling point is a good relative measure of intermolecular force. The greater the number of electrons in a molecule, the greater the London forces between them, the higher the boiling point.

Isoelectronic molecules have the same number of electrons. Polar molecules will have dipole-dipole forces along with London forces.

Hydrogen Bonding A hydrogen bond is where a hydrogen nucleus (a proton) is shared between electrons on neighbouring molecules. ammonia water • The hydrogen atom must be “unshielded. ” To accomplish this it must be bonded to a highly electronegative atom: nitrogen, oxygen or fluorine. • There must be at least on lone pair of electrons on the atom bonded to the hydrogen.

hydrogen bond (intermolecular) covalent bond (intramolecular) + – covalent bond (intramolecular) Hydrogen bonding is the strongest intermolecular force.
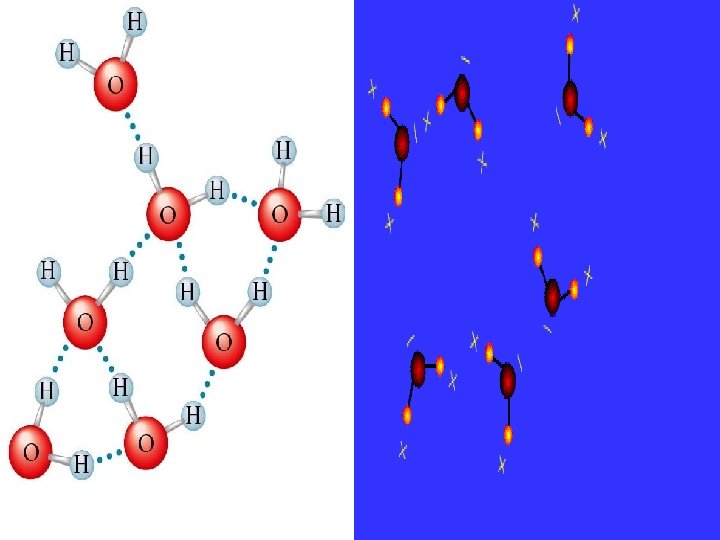

Some Effects of Hydrogen Bonding Hydrogen bonding between water molecules makes water have relatively high melting and boiling points when compared to other substances.

or sucrose Sucrose molecules are held firmly in a crystalline structure as solid and are readily dissolved in water. Water mixed with glycerol is called glycerine, commonly found in soaps. Water molecules form hydrogen bonds with glycerol molecules.
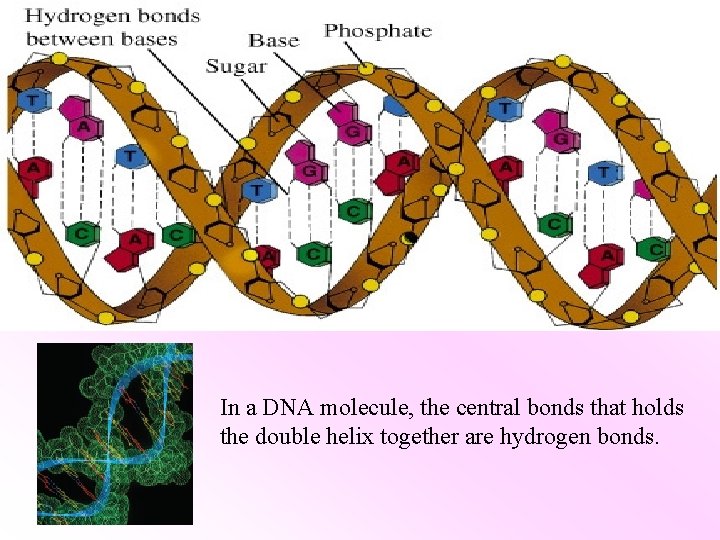
In a DNA molecule, the central bonds that holds the double helix together are hydrogen bonds.

Hydrogen bonding holds water molecules in a hexagonal lattice when it freezes. The large spaces in the lattice makes ice less dense than water.

Physical Properties of Liquids Surface Tension Liquids with high intermolecular forces, especially water, have high surface tension. Molecules right at the surface are only pulled downward and sideways.


Water’s high surface tension causes it to have a concave meniscus. Capillary action is water being pulled up a narrow tube by the intermolecular forces between the water molecules and the tube.

gasoline nail polish remover rubbing alcohol Liquids with small intermolecular forces are volatile: they tend to evaporate quickly.

ü Read pgs. 105 – 117 ü pgs. 109, 116 Practice #’s 1 – 5, 7 – 10 ü pg. 117 – 118 Section 3. 4 Questions #’s 1 – 10
- Slides: 17