INTRAMOLECULAR FORCES CHEMICAL BONDS Chemical bond force that

INTRAMOLECULAR FORCES CHEMICAL BONDS

Chemical bond: force that holds atoms together to form molecules, the attraction between electrons of one atom to the nucleus of another atom The nature of the bond (degree of electron sharing) can be predicted using electronegativity
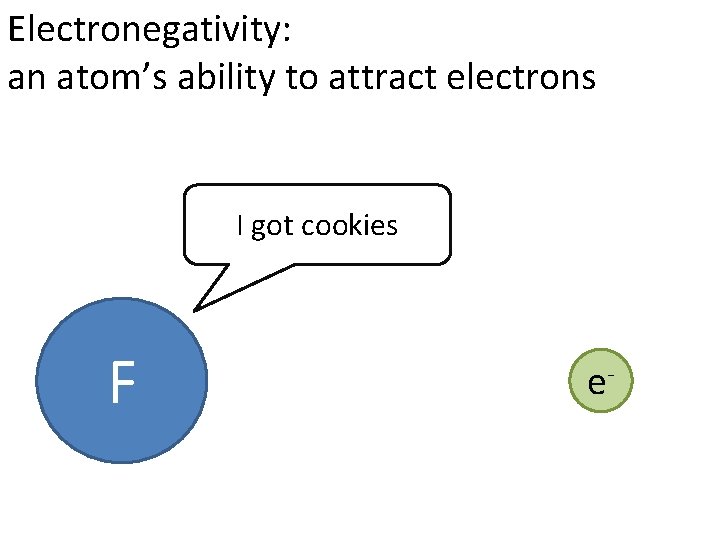
Electronegativity: an atom’s ability to attract electrons I got cookies F e-
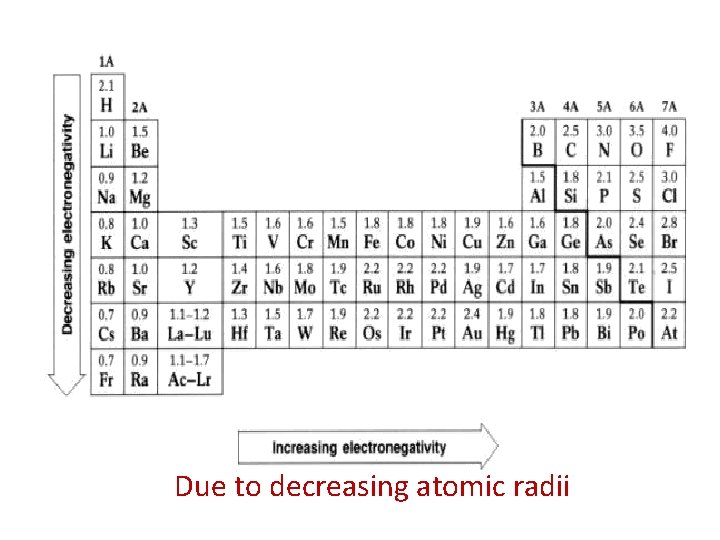
Due to decreasing atomic radii
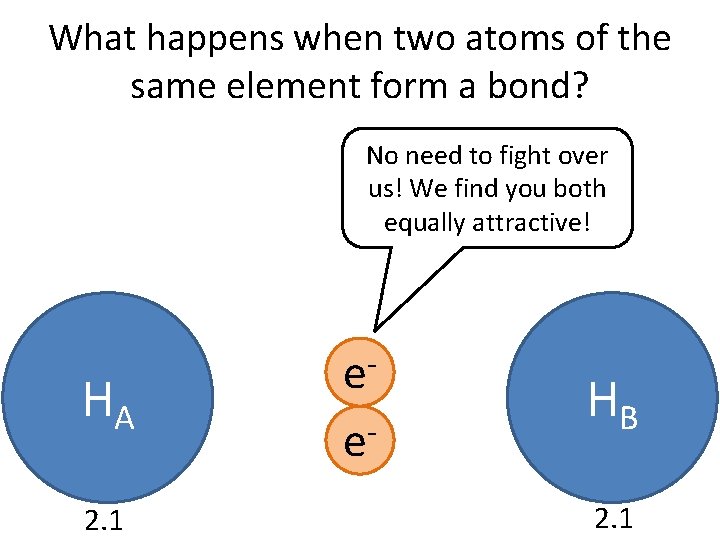
What happens when two atoms of the same element form a bond? No need to fight over us! We find you both equally attractive! HA 2. 1 e e HB 2. 1

Covalent bond • between 2 non metals • a pair of electrons is simultaneously attracted to two nuclei two atoms share the bonding electrons • use the fact that atoms like to have full outer shells to determine just how many electrons are shared • difference in electronegativity is ≤ 0. 2

Covalent bond • between 2 non metals • a pair of electrons is simultaneously attracted to two nuclei two atoms share the bonding electrons • use the fact that atoms like to have full outer shells to determine just how many electrons are shared • difference in electronegativity is ≤ 0. 2

Covalent bond • between 2 non metals • a pair of electrons is simultaneously attracted to two nuclei two atoms share the bonding electrons • use the fact that atoms like to have full outer shells to determine just how many electrons are shared • difference in electronegativity is ≤ 0. 2

Covalent bond • between 2 non metals • a pair of electrons is simultaneously attracted to two nuclei two atoms share the bonding electrons • use the fact that atoms like to have full outer shells to determine just how many electrons are shared • difference in electronegativity is ≤ 0. 2
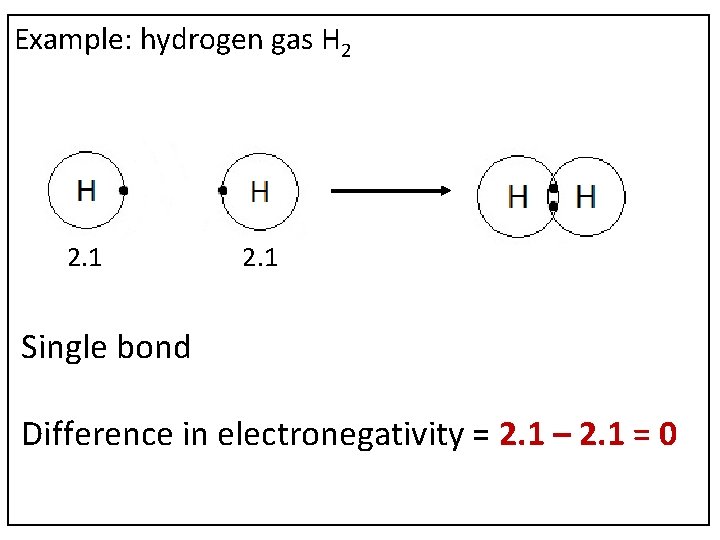
Example: hydrogen gas H 2 2. 1 Single bond Difference in electronegativity = 2. 1 – 2. 1 = 0
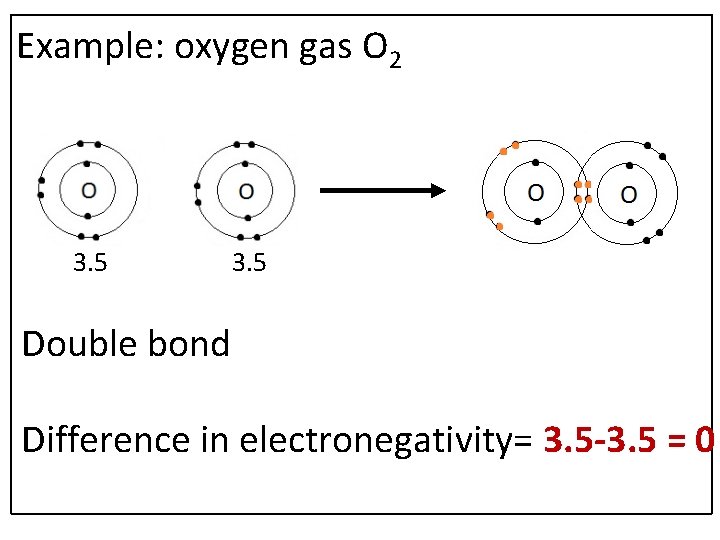
Example: oxygen gas O 2 3. 5 Double bond Difference in electronegativity= 3. 5 -3. 5 = 0

Do you think we can make a triple bond? yep How many electrons will be shared in a triple bond? 6
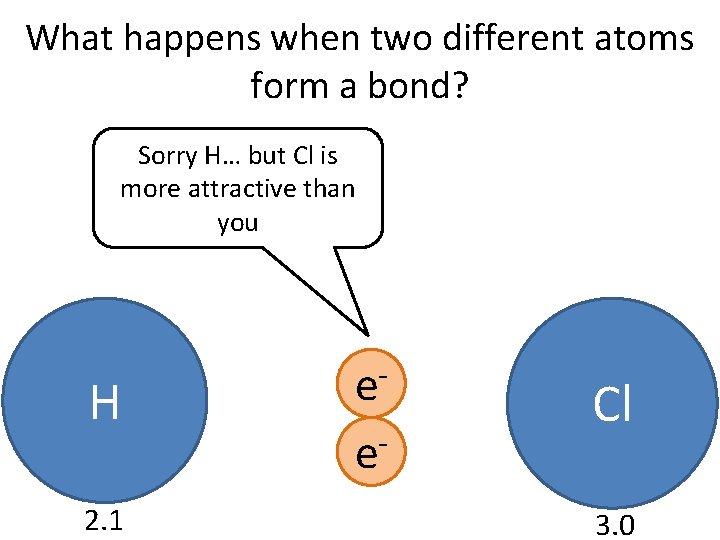
What happens when two different atoms form a bond? Sorry H… but Cl is more attractive than you H 2. 1 ee- Cl 3. 0

Polar covalent bond • between 2 non metals • a pair of electrons is shared, but they are more attracted to one nuclei than the other resulting in a dipole • difference in electronegativity is 0. 3 – 1. 6

Polar covalent bond • between 2 non metals • a pair of electrons is shared, but they are more attracted to one nuclei than the other resulting in a dipole • difference in electronegativity is 0. 3 – 1. 6

Polar covalent bond • between 2 non metals • a pair of electrons is shared, but they are more attracted to one nuclei than the other resulting in a dipole • difference in electronegativity is 0. 3 – 1. 6
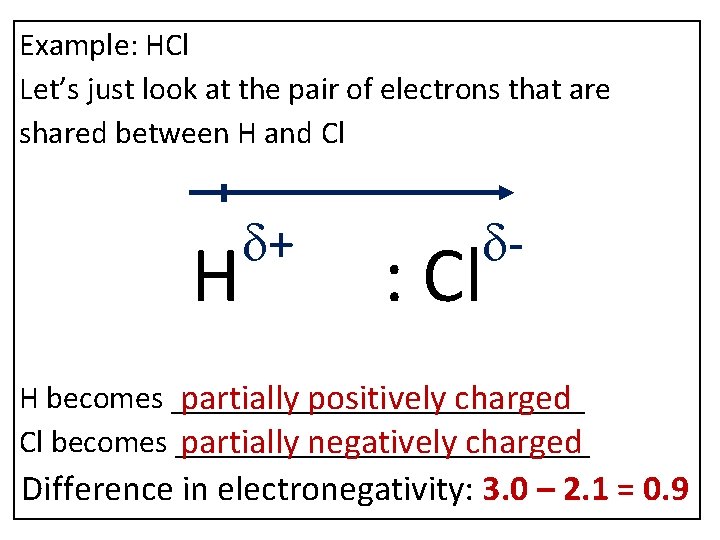
Example: HCl Let’s just look at the pair of electrons that are shared between H and Cl H d+ d- : Cl H becomes _____________ partially positively charged Cl becomes _____________ partially negatively charged Difference in electronegativity: 3. 0 – 2. 1 = 0. 9
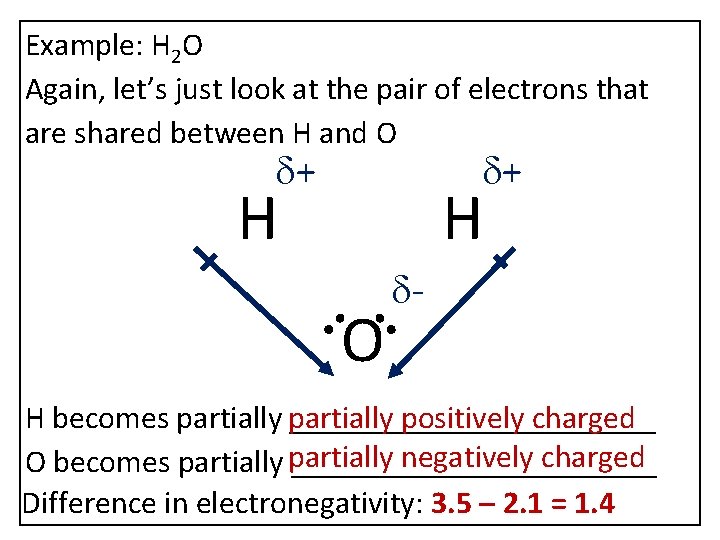
Example: H 2 O Again, let’s just look at the pair of electrons that are shared between H and O d+ d+ H H O d- H becomes partially ____________ positively charged negatively charged O becomes partially ____________ Difference in electronegativity: 3. 5 – 2. 1 = 1. 4

What happens when two atoms of very different electronegativities form a bond?


Ionic bond • between metal and non metal • involves electron transfer
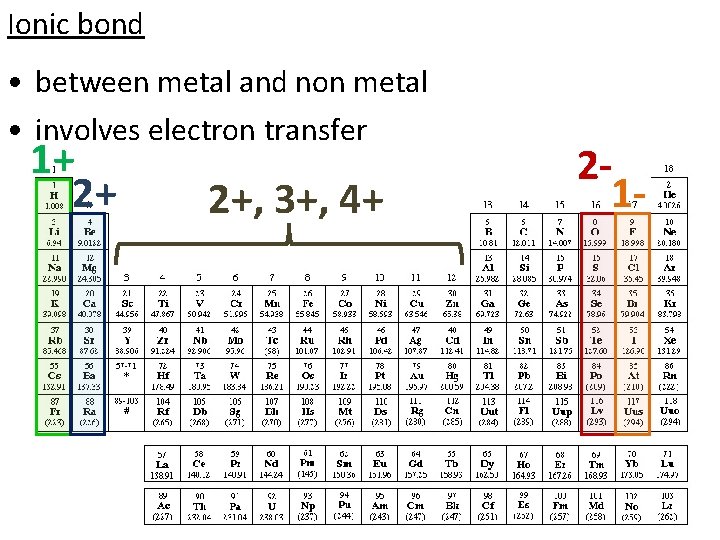
Ionic bond • between metal and non metal • involves electron transfer 1+ 2+ 2+, 3+, 4+ 21 -
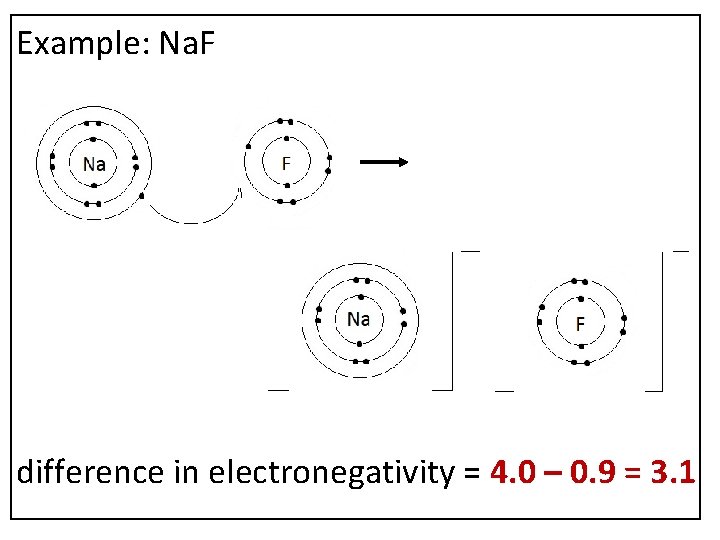
Example: Na. F difference in electronegativity = 4. 0 – 0. 9 = 3. 1

Ionic bond • between metal and non metal • involves electron transfer metal atom (or the atom with the lower electronegativity) loses electrons and becomes positively charged cation non metal atom (or the atom with the higher electronegativity) gains electrons and becomes negatively charged anion • atoms are held together by electrostatic attraction (attraction of opposite charges) • difference in electronegativity is ≥ 1. 7

Atoms bonded covalently are held together by the sharing of electrons. No electron sharing occurs in ionic bonds. What is holding the two atoms together in an ionic bond?

Ionic bond • between metal and non metal • involves electron transfer metal atom (or the atom with the lower electronegativity) loses electrons and becomes positively charged cation non metal atom (or the atom with the higher electronegativity) gains electrons and becomes negatively charged anion • atoms are held together by electrostatic attraction (attraction of opposite charges) • difference in electronegativity is ≥ 1. 7

Ionic bond • between metal and non metal • involves electron transfer metal atom (or the atom with the lower electronegativity) loses electrons and becomes positively charged cation non metal atom (or the atom with the higher electronegativity) gains electrons and becomes negatively charged anion • atoms are held together by electrostatic attraction (attraction of opposite charges) • difference in electronegativity is ≥ 1. 7

- Slides: 28