Intraluminal Coronary Reentry Bridgepoint Medical Crossboss and Stingray

Intraluminal Coronary Reentry Bridgepoint Medical Crossboss and Stingray Systems Craig A. Thompson, M. D. , MMSc. Director, Cardiovascular Catheterization and Intervention Yale University School of Medicine/Yale New Haven Hospital Consultant (Hon) Heart Hospital, London and London Chest Hospital Author : John K. Forrest, M. D Updated : June 2009 CRT 2012, Washington, DC

Craig A. Thompson, MD § Consulting Fees: §Abbott Vascular §Terumo Cardiovascular Systems Group §Ownership Interest (Stocks, Stock Options, or other Ownership Interest): §Bridgepoint

YALE SCHOOL OF MEDICINE Antegrade Dissection Reentry Techniques CTO Lumen Distal cap STAR reentry CTO Distal cap random Lumen LAST redirection CTO Distal cap Lumen Bridgepoint reentry CRT 2012, Washington, DC

YALE SCHOOL OF MEDICINE Why Develop Limited Subadventitial Tracking (La. ST)? • Bailout for complex cases – Of cases viewed by highly experienced operators as suitable for retrograde, ~20% of time collaterals cannot be crossed – Bailout for failed connection after successful retrograde collateral crossing – Treatment option for patients not suitable for retrograde at all and refractory to conventional approach • Preceded availability of dedicated reentry tools CRT 2012, Washington, DC
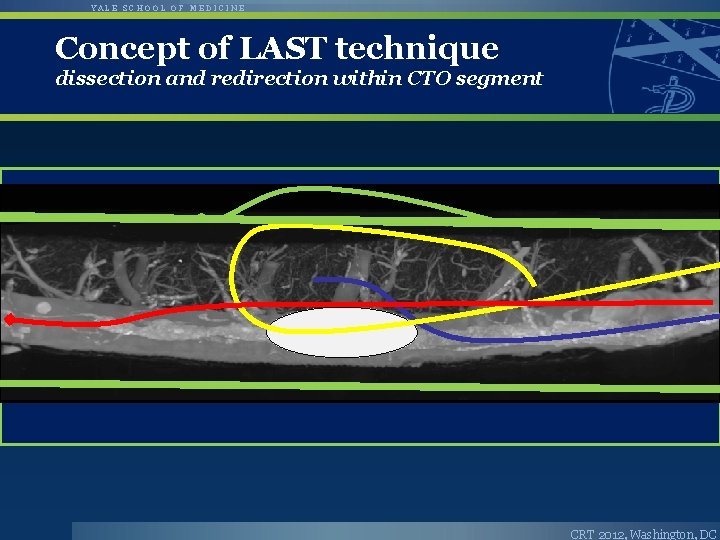
YALE SCHOOL OF MEDICINE Concept of LAST technique dissection and redirection within CTO segment CRT 2012, Washington, DC
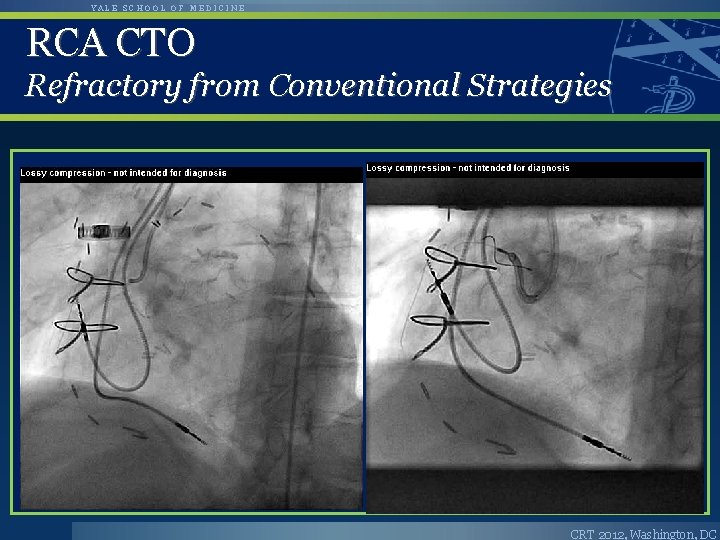
YALE SCHOOL OF MEDICINE RCA CTO Refractory from Conventional Strategies CRT 2012, Washington, DC
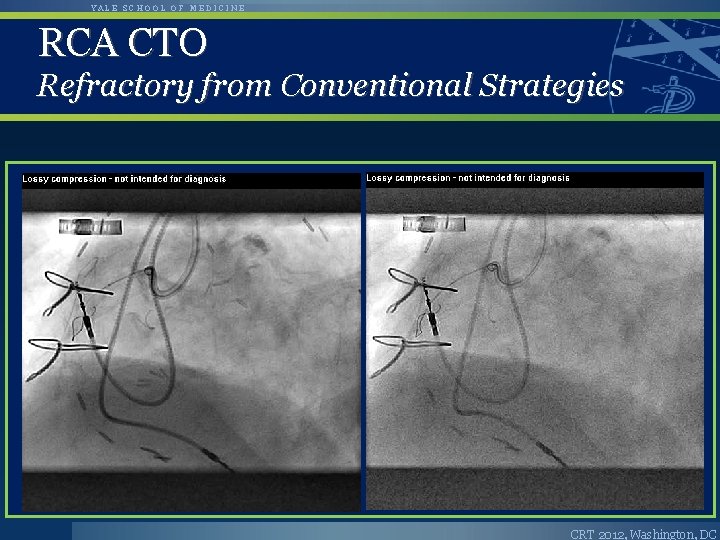
YALE SCHOOL OF MEDICINE RCA CTO Refractory from Conventional Strategies CRT 2012, Washington, DC
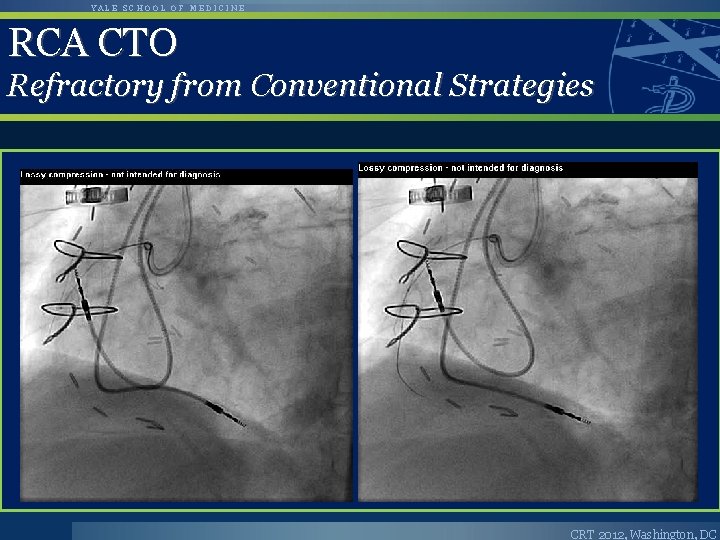
YALE SCHOOL OF MEDICINE RCA CTO Refractory from Conventional Strategies CRT 2012, Washington, DC
YALE SCHOOL OF MEDICINE RCA CTO Refractory from Conventional Strategies CRT 2012, Washington, DC

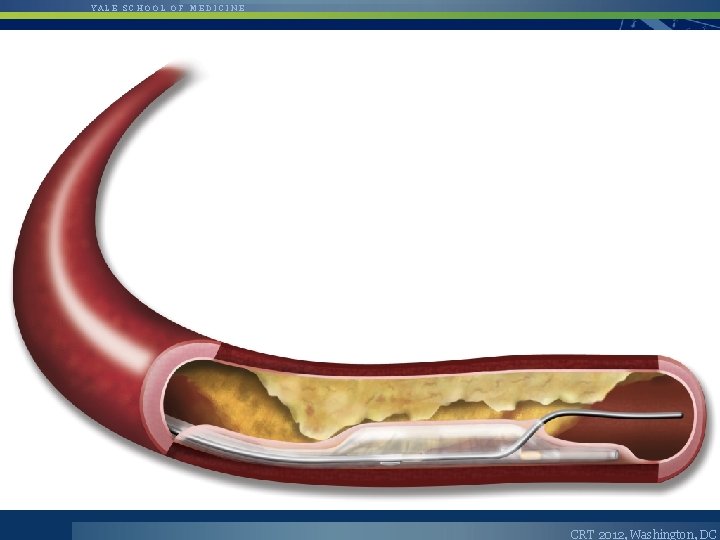
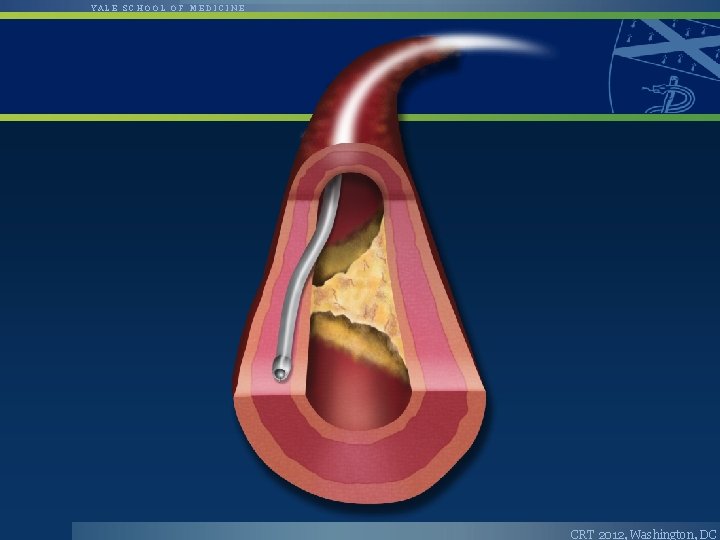
YALE SCHOOL OF MEDICINE The Cross. Boss™ CTO Catheter • • • Cross. Boss is designed to quickly and safely deliver a guidewire via true lumen or subintimal pathways Multi-wire coiled shaft Tracks via FAST Spin Technique – Highly torqueable coiled-wire shaft – FAST Spin reduces push required to cross CTO • • Atraumatic distal tip advanced across a CTO ahead of the guidewire OTW 0. 014” guidewire compatible CRT 2012, Washington, DC
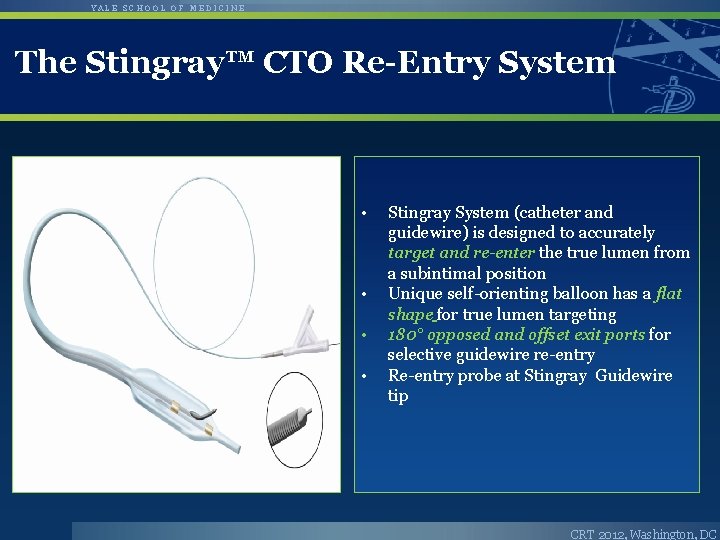
YALE SCHOOL OF MEDICINE The Stingray™ CTO Re-Entry System • • Stingray System (catheter and guidewire) is designed to accurately target and re-enter the true lumen from a subintimal position Unique self-orienting balloon has a flat shape for true lumen targeting 180° opposed and offset exit ports for selective guidewire re-entry Re-entry probe at Stingray Guidewire tip CRT 2012, Washington, DC

YALE SCHOOL OF MEDICINE Bridgepoint Crossing and Reentry CRT 2012, Washington, DC

YALE SCHOOL OF MEDICINE FAST-CTOs Overview • FAST-CTOs: Facilitated Antegrade Steering Technique in Chronic Total Occlusions • Objective: demonstrate safety and effectiveness of Bridge. Point Medical CTO System compared to historical controls • Bridge. Point Medical CTO System – Cross. Boss CTO Catheter – Stingray CTO Re-Entry System • Stingray CTO Orienting Balloon Catheter • Stingray CTO Re-Entry Guidewire CRT 2012, Washington, DC

YALE SCHOOL OF MEDICINE FAST-CTOs Study Design • 149 Patients, 20 Centers • Multicenter, non-randomized, IDE study • Historically-controlled – Control literature consisted of similarly designed CTO device trials with similar technical success and safety measures CRT 2012, Washington, DC
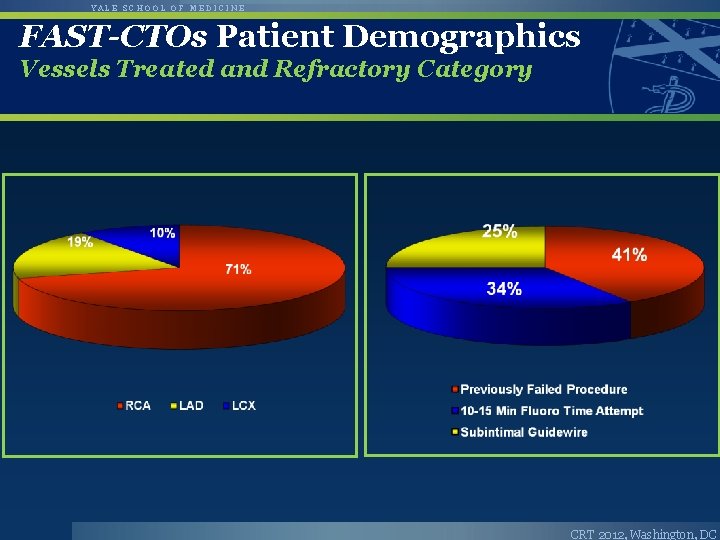
YALE SCHOOL OF MEDICINE FAST-CTOs Patient Demographics Vessels Treated and Refractory Category CRT 2012, Washington, DC
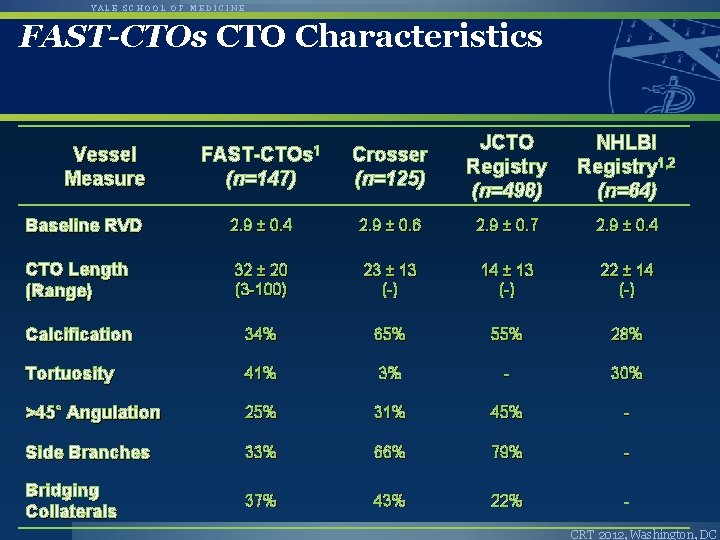
YALE SCHOOL OF MEDICINE FAST-CTOs CTO Characteristics FAST-CTOs 1 (n=147) Crosser (n=125) JCTO Registry (n=498) NHLBI Registry 1, 2 (n=64) Baseline RVD 2. 9 ± 0. 4 2. 9 ± 0. 6 2. 9 ± 0. 7 2. 9 ± 0. 4 CTO Length (Range) 32 ± 20 (3 -100) 23 ± 13 (-) 14 ± 13 (-) 22 ± 14 (-) Calcification 34% 65% 55% 28% Tortuosity 41% 3% - 30% >45˚ Angulation 25% 31% 45% - Side Branches 33% 66% 79% - Bridging Collaterals 37% 43% 22% - Vessel Measure CRT 2012, Washington, DC
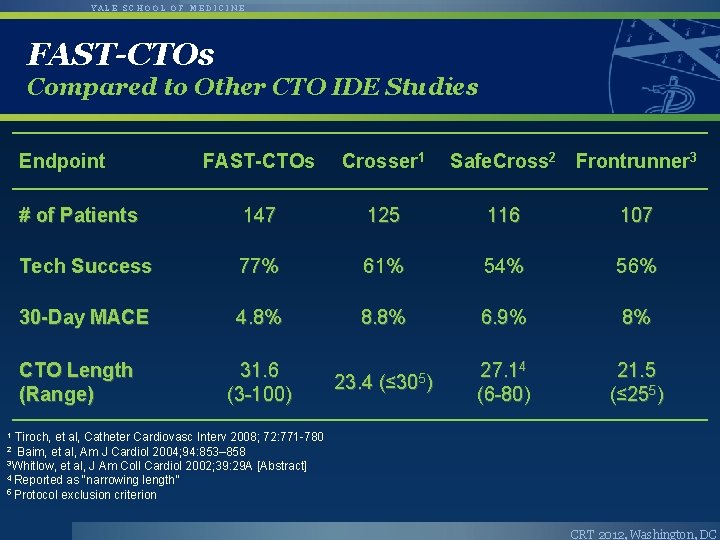
YALE SCHOOL OF MEDICINE FAST-CTOs Compared to Other CTO IDE Studies Endpoint FAST-CTOs Crosser 1 # of Patients 147 125 116 107 Tech Success 77% 61% 54% 56% 30 -Day MACE 4. 8% 8. 8% 6. 9% 8% 31. 6 (3 -100) 23. 4 (≤ 305) 27. 14 (6 -80) 21. 5 (≤ 255) CTO Length (Range) Safe. Cross 2 Frontrunner 3 1 Tiroch, et al, Catheter Cardiovasc Interv 2008; 72: 771 -780 2 Baim, et al, Am J Cardiol 2004; 94: 853– 858 3 Whitlow, et al, J Am Coll Cardiol 2002; 39: 29 A [Abstract] 4 Reported as “narrowing length” 5 Protocol exclusion criterion CRT 2012, Washington, DC
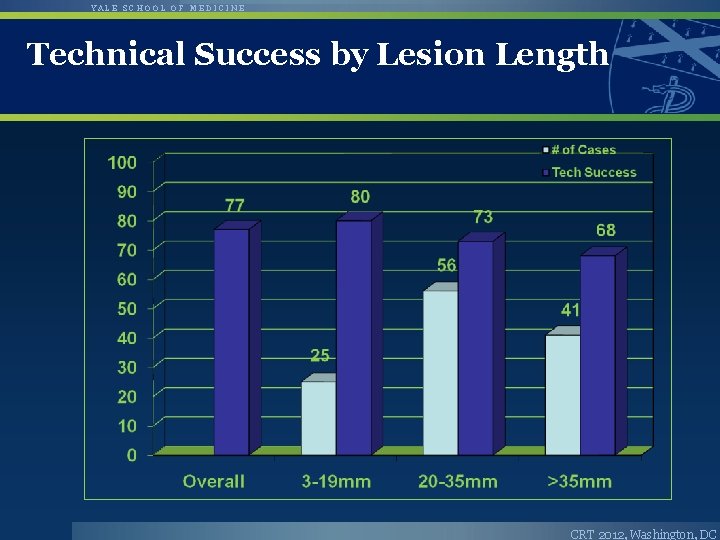
YALE SCHOOL OF MEDICINE Technical Success by Lesion Length CRT 2012, Washington, DC

YALE SCHOOL OF MEDICINE FAST-CTOs Results Procedure/Fluoroscopy Time FASTCTOs (n=147) min Historical Control (n=661) min JCTO Registry (n=498) min Crosser (n=125) min Proc Time Mean ± SD Median (Range) 105 ± 54 96 (17 -332) 146 - - 108 - Fluoro Time Mean ± SD Median (Range) 44 ± 25 41 (5 -163) 53 - 45 (1 -301) 44 - Measure CRT 2012, Washington, DC
YALE SCHOOL OF MEDICINE CRT 2012, Washington, DC
YALE SCHOOL OF MEDICINE CRT 2012, Washington, DC
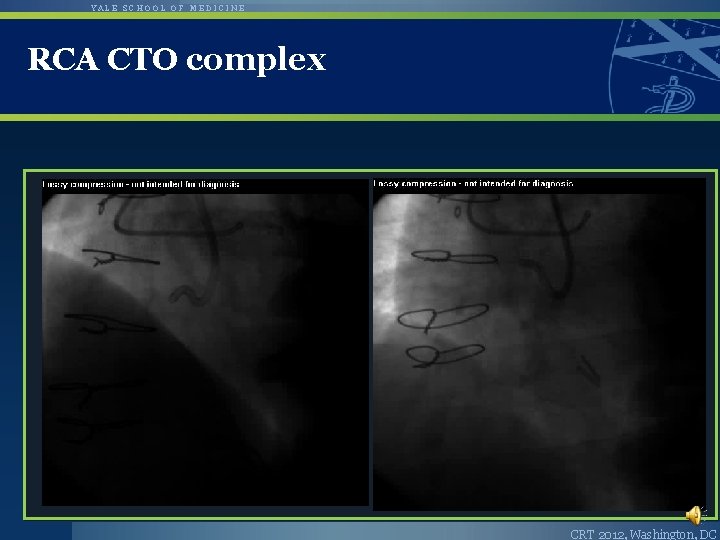
YALE SCHOOL OF MEDICINE RCA CTO complex CRT 2012, Washington, DC
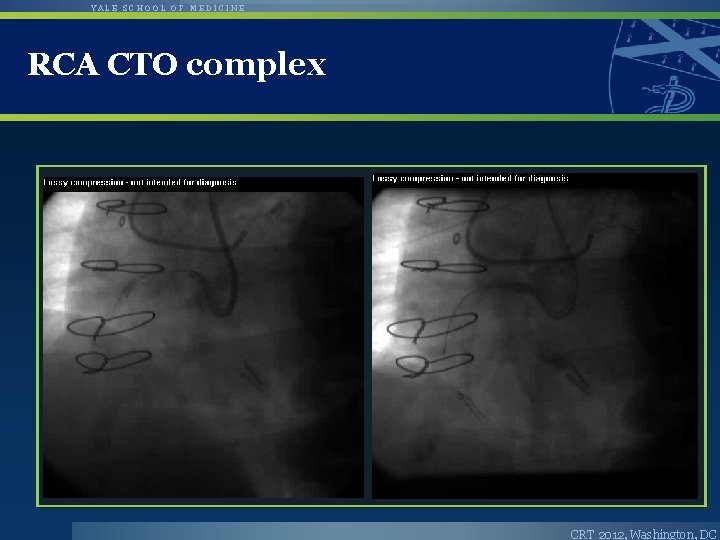
YALE SCHOOL OF MEDICINE RCA CTO complex CRT 2012, Washington, DC
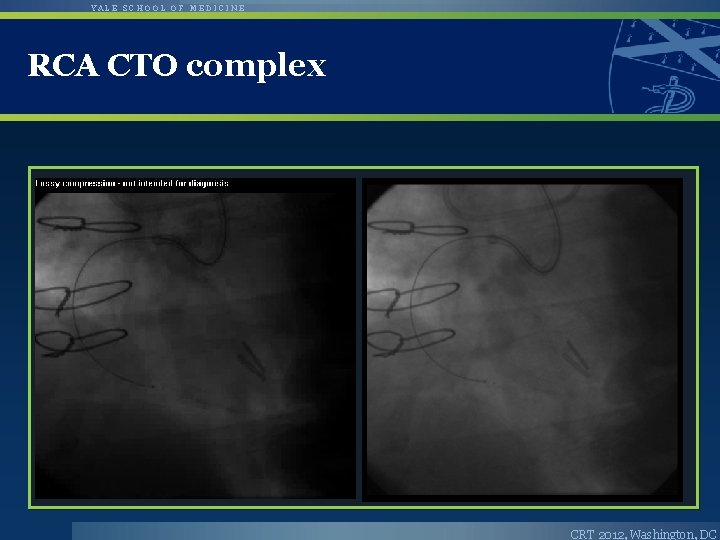
YALE SCHOOL OF MEDICINE RCA CTO complex CRT 2012, Washington, DC
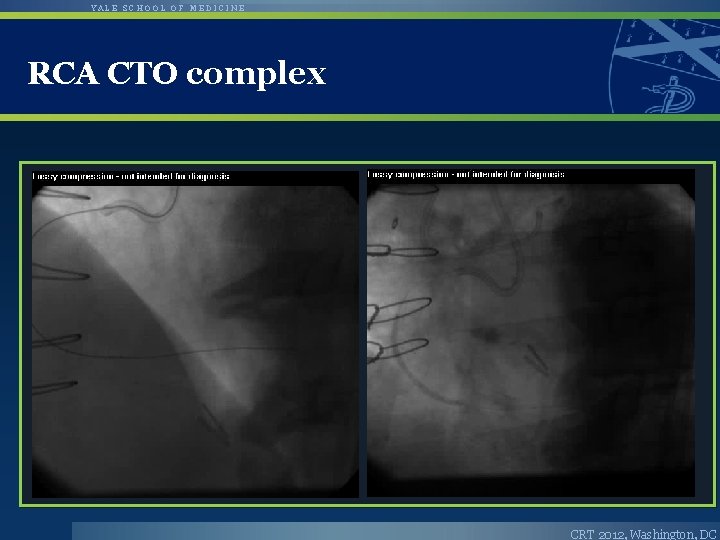
YALE SCHOOL OF MEDICINE RCA CTO complex CRT 2012, Washington, DC
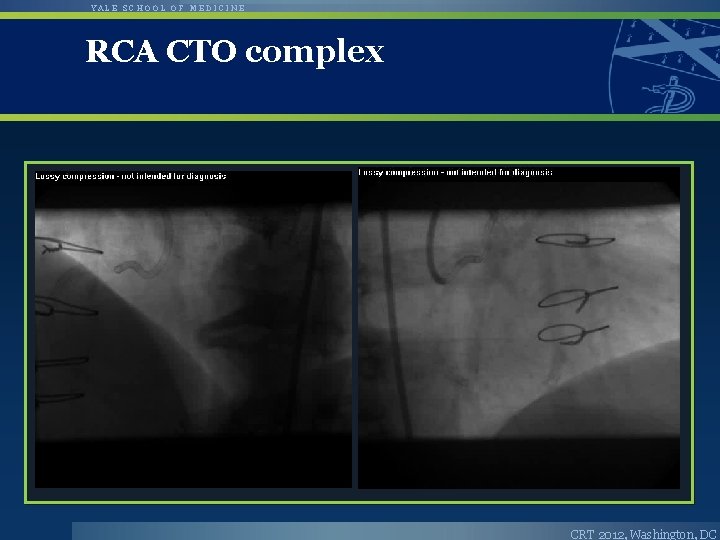
YALE SCHOOL OF MEDICINE RCA CTO complex CRT 2012, Washington, DC
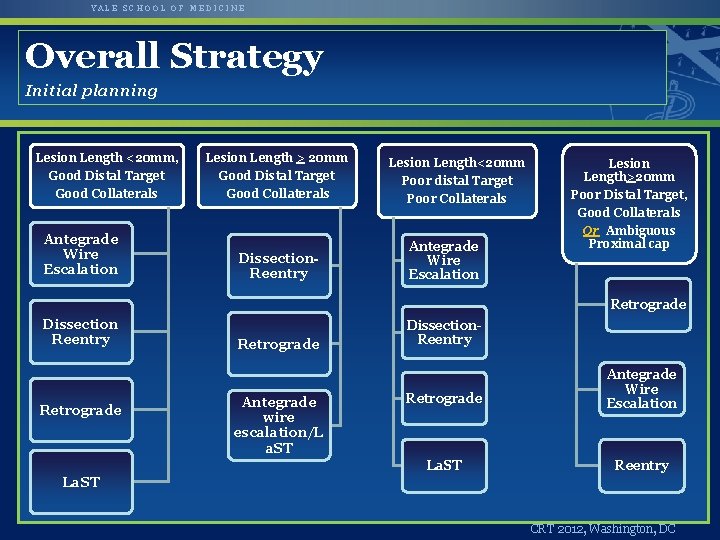
YALE SCHOOL OF MEDICINE Overall Strategy Initial planning Lesion Length <20 mm, Lesion Length > 20 mm Good Distal Target Good Collaterals Antegrade Wire Escalation Dissection. Reentry Lesion Length<20 mm Poor distal Target Poor Collaterals Antegrade Wire Escalation Lesion Length>20 mm Poor Distal Target, Good Collaterals Or Ambiguous Proximal cap Retrograde Dissection Reentry Retrograde Antegrade wire escalation/L a. ST Dissection. Reentry Retrograde Antegrade Wire Escalation La. ST Reentry La. ST CRT 2012, Washington, DC
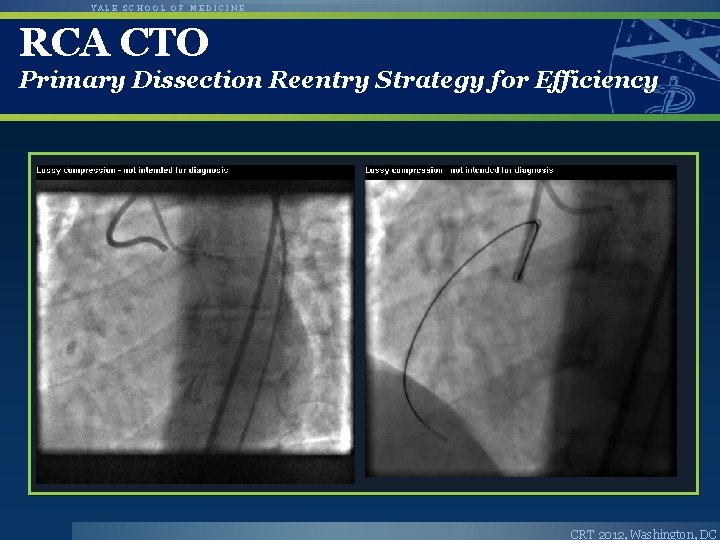
YALE SCHOOL OF MEDICINE RCA CTO Primary Dissection Reentry Strategy for Efficiency CRT 2012, Washington, DC
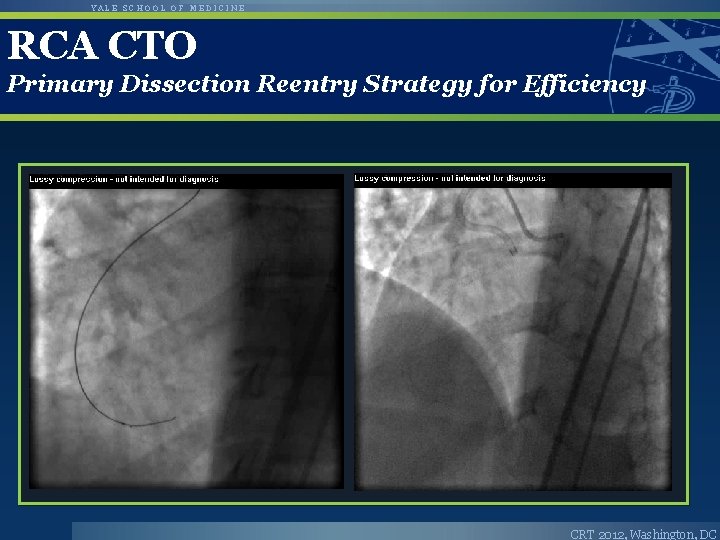
YALE SCHOOL OF MEDICINE RCA CTO Primary Dissection Reentry Strategy for Efficiency CRT 2012, Washington, DC
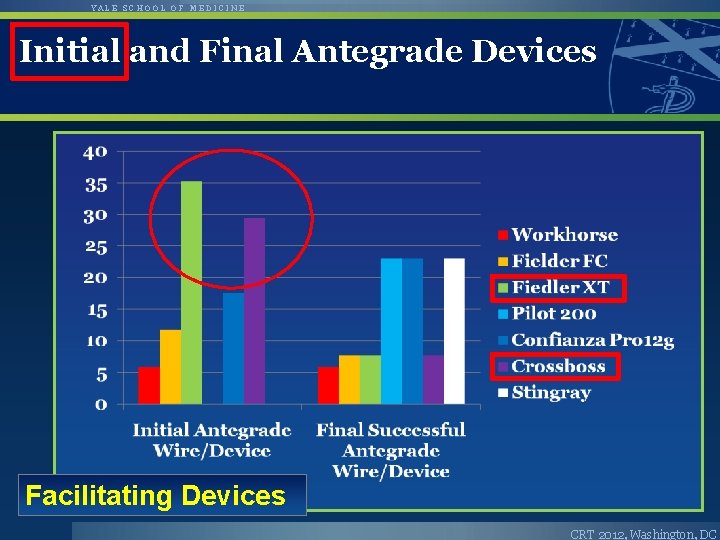
YALE SCHOOL OF MEDICINE Initial and Final Antegrade Devices Facilitating Devices CRT 2012, Washington, DC
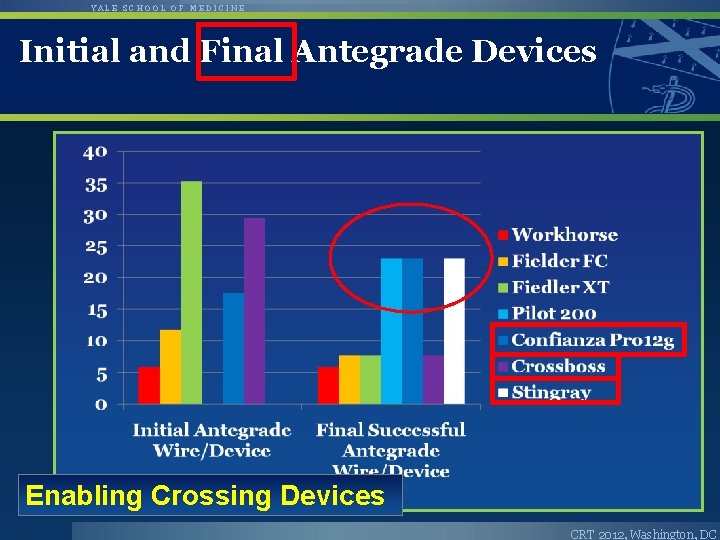
YALE SCHOOL OF MEDICINE Initial and Final Antegrade Devices Enabling Crossing Devices CRT 2012, Washington, DC

YALE SCHOOL OF MEDICINE Crossboss CTO Catheter, Sapien, Watchman Among Picks for Best Cardiac Devices of 2011 January 25, 2012 — Which Medical Device announced the winner of its Device of the Year Award 2011 in the cardiology category. The expert panel of judges reviewed the nominations for the most significant, innovative and invaluable devices, and the winner and runners-up in the cardiology category are: Winner The winner of the cardiology category is the Cross. Boss CTO Catheter manufactured by Bridge. Point Medical. Which Medical Device Senior Editor for Cardiology Azfar Zaman commends this novel piece of technology with a good success rate in opening chronic occlusions, saying, “It is a quantum leap in the world of CTOs. ” Runner-up The judges chose both the SAPIEN Transcatheter Heart Valve from Edwards Life Sciences and Medtronic’s Corevalve. The panel commented that, “Both these devices have stood the test of time and the medium term (five years) performance of both valves is sufficiently robust to hope that TAVI may well become the first preferred option in patients with aortic stenosis. ” Special commendations Special mention is given to the Watchman device from Boston Scientific, which has the potential to improve lives by stopping the need for warfarin in patients with atrial fibrillation. The judging panel also praised the Absorb Bioresorbable Vascular Scaffold from Abbott Vascular, saying “We believe that this is the next generation for stents; it promises much but needs to deliver. ” For more information: www. whichmedicaldevice. com, www. sagepublications. com CRT 2012, Washington, DC

YALE SCHOOL OF MEDICINE FAST-CTOs Conclusions 1. The Bridge. Point Medical CTO System is safe and effective in the treatment of coronary epicardial chronic total occlusions 2. Procedure time with the System is shorter with lower fluoroscopy times compared to historical controls 3. The Bridgepoint Medical CTO system’s effectiveness, safety, and efficiency compares favorably with other investigated devices and techniques for therapy of CTO CRT 2012, Washington, DC
- Slides: 33