INTRACTABLE EPILEPSY DRUG RESISTANCE EPILEPSY ERNESTO ALONSO MD

“INTRACTABLE EPILEPSY” DRUG RESISTANCE EPILEPSY ERNESTO ALONSO, MD NEUROLOGY RESIDENT, PGY-4 KUMC 4/21/17

-DEFINITION -EPIDEMIOLOGY -RISK FACTORS -PATHOGENESIS -COMPLICATIONS -DIFFERENTIAL DIAGNOSIS -EVALUATION -TREATMENT OPTIONS -SUMARY

DEFINITION Drug-resistant epilepsy by the ILAE (Kwan et al. , 2010). Failure of adequate trials of two tolerated and appropriately chosen and used AEDs to achieve sustained seizure freedom for a sufficiently long period of time. Recommended replacing the term “intractable” with “drug resistant epilepsy” DRE.

EPIDEMIOLOGY Incidence and prevalence are uncertain. Estimates of the proportion of DRE are between 20 -40 %

RISK FACTORS No single factor has been found A combination of two or more of risk factors help to define a possible DRE

RISK FACTORS 1 -The response to the first AED (the most important). 2 - High number of seizures prior to diagnosis and treatment. 3 -The underlying etiology and seizure classification.

RISK FACTORS 4 -Certain pediatric epilepsy syndromes are almost invariably medically intractable. - Early neonatal myoclonic encephalopathy - Early infantile epileptic encephalopathy - Lennox Gastaut syndrome - Rasmussen encephalitis among others.

RISK FACTORS 5 -Other findings -Presentation with status epilepticus -Longer duration of epilepsy -Family history of epilepsy -History of febrile convulsions -Abnormal EEG findings -An abnormal neurologic examination and/or developmental delay

RISK FACTORS 6 -Age at presentation. -Later childhood or adolescence more likely associated than seizures onset between 5 and 10 years old. >65 years, less likely than younger adults.

PATHOGENESIS The mechanisms are not completely understood More likely multifactorial. 1 -The transporter hypothesis Overexpression of multidrug efflux transporters causing a decrease of intracerebral drug concentration. 2 -The target hypothesis Suggest alterations of the cellular targets of AED

PATHOGENESIS 3 -The morphologic Hypothesis MTS is a progressive condition. Pathology, demonstrates Neuroimaging, support a progressive process. Alterations in neural circuit may lead to an epileptic network that becomes drug resistant over time.

PATHOGENESIS 4 -The genetic hypothesis Polymorphism in the drug transporter gene (ABCB 1 or MDR 1) and DRE, but was not corroborated in follow-up studies.

COMPLICATIONS 1 -Mortality: Estimated at 1. 37 per 100 person-years Patients with recurrent seizures is 4. 69 Related to the underlying cause of epilepsy (neoplasm, neurodegenerative disease) Seizure-related (SE and in seizure-related accidents) (SUDEP), 40 times more likely among patients who continue to have seizures than in seizure free patients

COMPLICATIONS 2 -Nonfatal injuries (head injury, burns, and fractures, among others; most are seizure-related) 3 -Disability and diminished quality of life. ( include poor academic achievement, unemployment, and social isolation, most patients cannot drive) 4 -Psychological problems (depression, psychosis , postictal psychotic disorders, alternative psychosis)

DIFFERENTIAL DIAGNOSIS 1. Apparent intractability 2. Psychogenic nonepileptic seizures (PNES) 3. Other nonepileptic paroxysmal disorders

DIFFERENTIAL DIAGNOSIS 1 -Apparent intractability -Misdiagnosis -Incorrect drug choice -Inappropriate dose -AED drug toxicity -Noncompliance -Lifestyle factors (recreational drug or alcohol abuse, sleep deprivation)

DIFFERENTIAL DIAGNOSIS 2 -Psychogenic nonepileptic seizures (PNES) -Typically do not respond to AED. -video-EEG monitoring is the gold standard test for the diagnosis of PNES.

DIFFERENTIAL DIAGNOSIS 3 -Other nonepileptic paroxysmal disorders • Syncope • Cardiac arrythmias • Psychological disorders • Sleep disorders • Paroxysmal movement disorders • Migraine • Miscellaneous neurologic events
TREATMENT OPTIONS 1. 2. 3. 4. 5. 6. 7. AED Epilepsy surgery Vagus nerve stimulation Cortical stimulation Other stimulation approaches Ketogenic diet Cannabinoids

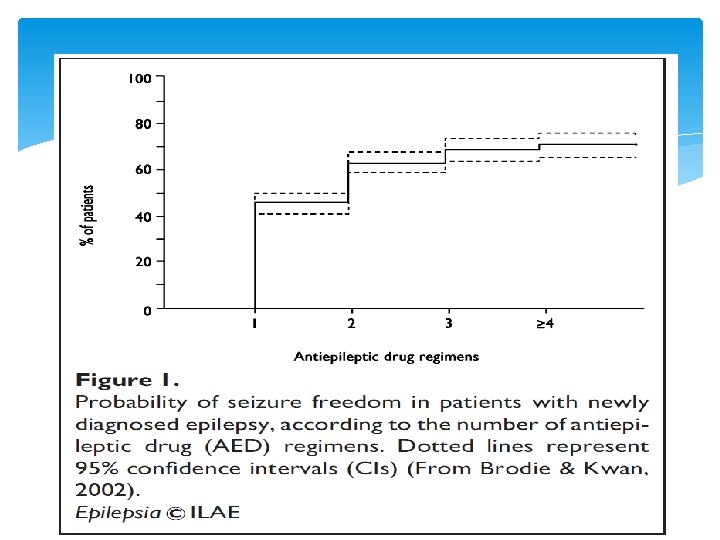
AED Seizure freedom is the goal of epilepsy treatment. It induces morbidity and mortality, prevents sudden death from epilepsy and improves quality of life. In the series of 480 patients newly diagnosed with epilepsy - 47 % became seizure free on the first AED - 13 % responded to a second AED. - 4 % responded to a third AED (Kwan, Brodie, 2002), Similar (Mohanraj, Brodie, 2006)

Choosing an AED Drug effectiveness for the seizure type Potential side effects Interactions with other medications Comorbid medical conditions, especially but not limited to hepatic and renal disease Age and gender, including childbearing plans Lifestyle and patient preferences Cost

AED Broad spectrum Focal and Generalized onset Brivaracetam Clobazam Felbamate Lamotrigine Levetiracetam Perampanel Rufinamide Topiramate Valproate Zonisamide

AED Narrow Spectrum Focal seizures Carbamazepine Eslicarbazepine Ezogabine Gabapentin Lacosamide Oxcarbazepine Phenobarbital Phenytoin Pregabalin Tiagabine Vigabatrin

AED Narrow Spectrum Absence seizures (type of generalized seizure) Ethosuximide

EPILPESY SURGERY All the patients with focal DRE need an early evaluation for resective epilepsy surgery. Surgery is the Gold standard therapeutic option. Earlier surgery is better from the clinical, psychosocial, and economic viewpoints (Engel, 2008).
EPILPESY SURGERY Good candidates: 1 - Epilepsy well localized based on testing 2 -Disabling Seizures 3 - The location away from eloquent regions of brain 4 - Risks to cognition and memory post op, preformed Not surgical candidates include: 1 -Evidence of bilateral onset of seizures 2 -Severe psychiatric or medical comorbidities 3 -Primary generalized epilepsy.

EPILEPSY SURGERY Types of surgical treatments 1 -Standardized resections § Anterior temporal resections (most common) § Amygdalohippocampectomy § Hemispherectomy 2 -Tailored resections § Localized cortical resections § Lesionectomies, including hypothalamic hamartomas § Multilobar resections
EPILEPSY SURGERY Types of surgical treatments 3 -Disconnections § Corpus callosotomy § Multiple subpial transections 4 -Others § Gamma Knife § DBS § Response stimulations § Laser Ablation
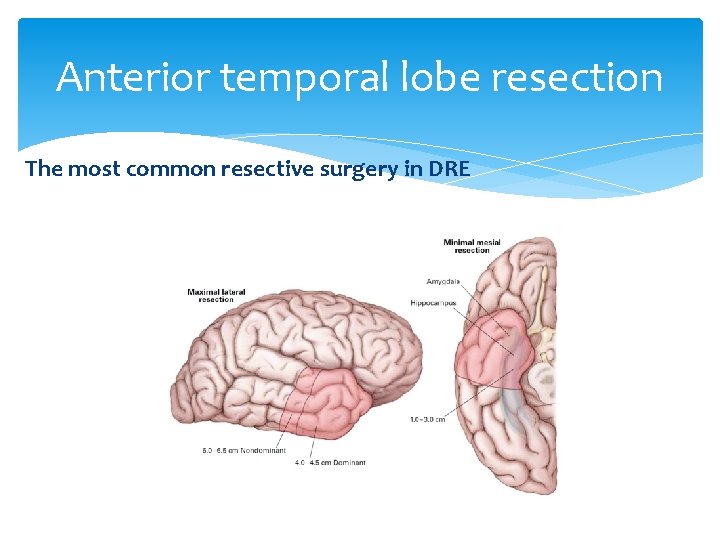
Anterior temporal lobe resection The most common resective surgery in DRE

EVALUATION § High resolution MRI § Video EEG monitoring

VEEG Inpatient video-EEG monitoring > 25 % In some series, of individuals referred for monitoring are found to have PNES.
MRI reveals an etiology for epilepsy Absence of a causative lesion, an epileptogenic focus can be defined using advanced neuroimaging techniques including (PET), (SPECT)

Pre-surgical evaluation Goals: 1 -Localize the epileptogenic zone. 2 -Identify any evidence that indicate the need for additional tests including invasive EEG 3 -Determine surgical risks to cerebral functions.

Pre-surgical evaluation Phase I Evaluation EMU SPECT Study MRI of Brain Inter-ictal PET Scan Neuropsychological Testing Psychiatric Evaluation

Pre-surgical evaluation Phase I Evaluation WADA Test Functional MRI MEG Surgery Conference

Pre-surgical evaluation Phase II Evaluation Continuous video-EEG monitoring with intracranial electrodes -Helps to localize the area where the seizures are starting within only a few millimeter radiuses. -Helps to learn if the seizures are coming from more than one area of the brain. -Brain mapping is performed with electrocorticography and stimulation.

VNS FDA approved in 1997 -Partial onset seizures (adults and children >12 years) ~ 30 to 40 % of patients achieve a > 50 % reduction seizure frequency, benefit sustained over time -Offer the opportunity to abort a seizure at its onset. -Valid option for DRE in patients: a) Opposed to intracranial surgery b) Not candidates for intracranial surgery c) Seizures not improved by intracranial epilepsy surgery.

VNS Contraindications Cardiac Arrhythmias, tracheotomy (relative) Debilitated patients with little subcutaneous fat (significant risk of skin breakdown), (relative) Others : vagotomies (left-sided or bilateral), significant upper airway, pharyngeal, cardiac, or GI problems. Caution in patients with history of vasovagal syncope or other brain stimulator implantation.
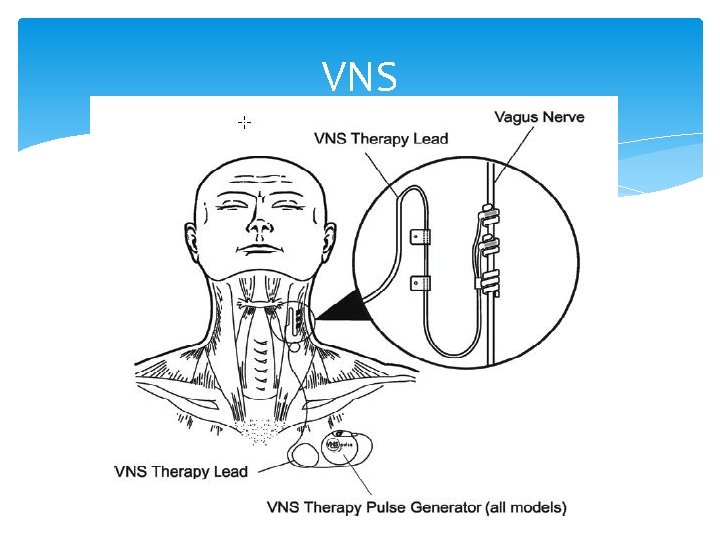
VNS
CORTICAL STIMULATION FDA approved in 2013 for patients with well delineated seizure focus Useful when resective surgery is not possible Patients with >1 seizure focus or epilepsy originating from eloquent cortex May be associated with a substantial reduction in seizure number, intensity, and duration.

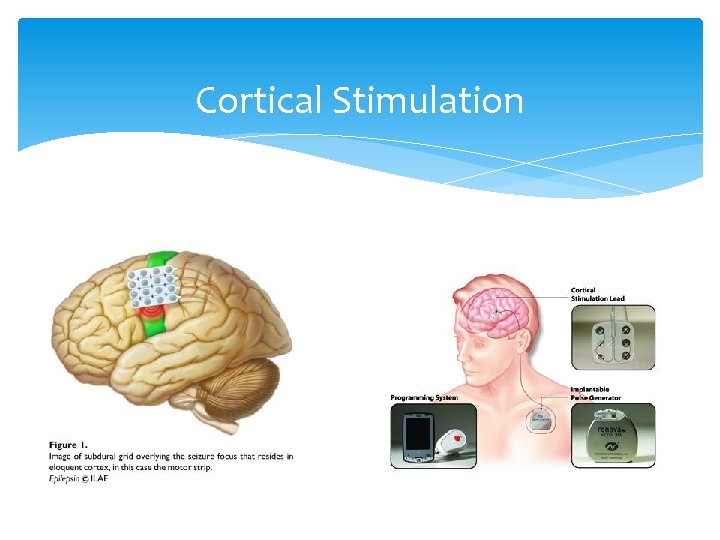
Cortical Stimulation

RNS FDA approved in November 2013 Patient s >18 years, DRE with no more than two epileptic foci. A multicenter double blind, randomized, controlled study showed, a 37. 9 % decline in mean seizure frequency, long term showed a 44% mean reduction seizure frequency in 1 year and 53 % at 2 years.

RNS Electrodes are implanted near the epileptic focus using depth or subdural strips, device are programmed to trigger direct cortical stimulation, when seizure is detected. RNS: Called closed loop stimulation, just activated with seizures. VNS: Called open loop stimulation, provided near continuous stimulation.
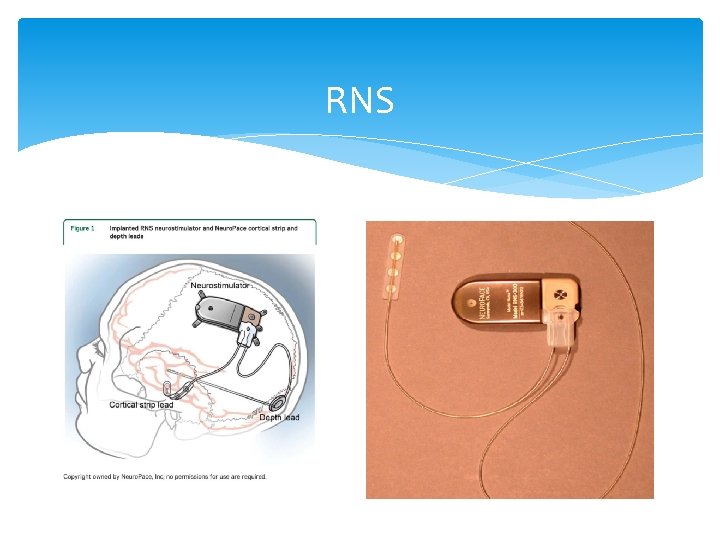
RNS

OTHER STIMULATION APPROACHES DBS : Reduce seizure frequency by 50 % or more, long term efficacy and safety Transcranial magnetic stimulation – Low frequency transcranial magnetic stimulation reduces cortical excitability Trigeminal nerve stimulation – Low frequency applied externally reduce seizures, associated with improvements in mood. Approved for use in the European Union, still being investigated in the USA.

KETOGENIC DIET Fasting has been proposed in the treatment of epilepsy since the time of Hippocrates. KD was proposed as alternative in 1921 prior to introduction of the modern AED. Classic KD (high-fat, low protein diet), induce urinary ketosis, mimic starvation, preserving necessary caloric intake. The efficacy of the KD, not less than 38 % of patients, achieve a 50% reduction of seizures after 1 year of treatment Effective for all seizures types, long-term outcomes, only 10 % of patients remained on the diet at 3 to 6 years. -Modified Atkins and glycemic index diet , have began to be prescribed in the treatment of epilepsy.

KETOGENIC DIET -Inclusion criteria DRE after failed to two or three AED Age, between 1 to 8 years old (infant, adolescents, and adult can be included) Patient with structural brain disorders -Exclusion criteria Patients with liver, kidney, heart, GI, psychiatric disorders. Patients with progressive encephalopathy with or without a known metabolic marker. Inability to maintain adequate nutrition Parent or caregiver noncompliance

CANNABINOIDS The American Academy of neurology’s position statement on marijuana-based products supports continued rigorous research to evaluate long term safety and efficacy because there is not sufficient evidence to support any conclusive statement on its effectiveness for many neurological conditions.

SUMARY - 20 to 40 % of patients with epilepsy will not have complete seizure control with AED. -The mechanisms of DRE are not completely understood and are likely to be multifactorial -Failure to the first AED trial is the most important risk factor -DRE patients have an increased risk of mortality and other disabilities. -DRE patients, need a video EEG to confirm the diagnosis of epilepsy. -More than 25 % of patients with DRE will have PNES. -v. EEG will help with seizure classification; semiology correlation needed. -Localization-related DRE should have (MRI) to identify a potential surgical lesion. ( SPECT, PET) can be done in patients that the MRI does not show any lesion, or MRI lesion does not correspond to the EEG localization, or who have dual pathology. -Recommended surgical evaluation for all the patients with localizationrelated or partial epilepsy -Surgery is a gold standard treatment option for focal DRE -Patients that are not candidate for surgery or if seizures persist after surgery, recommended others options (VNS, RNS, CS, DBS, TMS)

REFERENCES 1 -Kwan P, Schachter SC, Brodie MJ. Drug-resistant epilepsy. N Engl J Med 2011; 365: 919. 2 -Berg AT, Kelly MM. Defining intractability: comparisons among published definitions. Epilepsia 2006; 47: 431. 3 -Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med 2000; 342: 314. 4 -Berg AT, Shinnar S, Levy SR, et al. Early development of intractable epilepsy in children: a prospective study. Neurology 2001; 56: 1445. 5 -Kwan P, Brodie M. Issues of medical intractability for surgical candidacy. In: The treatment of epilepsy and practice, 4 th, Wyllie E, Gupta A, Lachhwani D. (Eds), Lippincott, Williams & Wilkins, Philadelphia 2006. p. 983. 6 -Brodie MJ, Kwan P. Staged approach to epilepsy management. Neurology 2002; 58: S 2. 7 -Kwan P, Arzimanoglou A, Berg AT, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia 2010; 51: 1069. 8 -Brodie MJ, Barry SJ, Bamagous GA, et al. Patterns of treatment response in newly diagnosed epilepsy. Neurology 2012; 78: 1548. 9 -Benbadis SR, Tatum WO, Vale FL. When drugs don't work: an algorithmic approach to medically intractable epilepsy. Neurology 2000; 55: 1780. 10 -Mohanraj, R, Brodie, MJ. Prediction of refractory epilepsy [abstract]. Epilepsia 2003; 44(Suppl 8): 156. 11 -Perucca P, Hesdorffer DC, Gilliam FG. Response to first antiepileptic drug trial predicts health outcome in epilepsy. Epilepsia 2011; 52: 2209. 12 -Hitiris N, Mohanraj R, Norrie J, et al. Predictors of pharmacoresistant epilepsy. Epilepsy Res 2007; 75: 192. 13 -Mac. Donald BK, Johnson AL, Goodridge DM, et al. Factors predicting prognosis of epilepsy after presentation with seizures. Ann Neurol 2000; 48: 833. 14 -Semah F, Picot MC, Adam C, et al. Is the underlying cause of epilepsy a major prognostic factor for recurrence? Neurology 1998; 51: 1256.

REFERENCES 15 -Berg AT, Levy SR, Novotny EJ, Shinnar S. Predictors of intractable epilepsy in childhood: a case-control study. Epilepsia 1996; 37: 24. 16 -Mohanraj R, Brodie MJ. Outcomes in newly diagnosed localization-related epilepsies. Seizure 2005; 14: 318. 17 -Berg AT, Levy SR, Testa FM, D'Souza R. Remission of epilepsy after two drug failures in children: a prospective study. Ann Neurol 2009; 65: 510. 18 -Siddiqui A, Kerb R, Weale ME, et al. Association of multidrug resistance in epilepsy with a polymorphism in the drug-transporter gene ABCB 1. N Engl J Med 2003; 348: 1442. 19 -Sills GJ, Mohanraj R, Butler E, et al. Lack of association between the C 3435 T polymorphism in the human multidrug resistance (MDR 1) gene and response to antiepileptic drug treatment. Epilepsia 2005; 46: 643. 20 -Tan NC, Heron SE, Scheffer IE, et al. Failure to confirm association of a polymorphism in ABCB 1 with multidrug-resistant epilepsy. Neurology 2004; 63: 1090. 21 -Szoeke C, Sills GJ, Kwan P, et al. Multidrug-resistant genotype (ABCB 1) and seizure recurrence in newly treated epilepsy: data from international pharmacogenetic cohorts. Epilepsia 2009; 50: 1689. 22 -Tomson T. Mortality in epilepsy. J Neurol 2000; 247: 15. 23 -Mohanraj R, Norrie J, Stephen LJ, et al. Mortality in adults with newly diagnosed and chronic epilepsy: a retrospective comparative study. Lancet Neurol 2006; 5: 481. 24 -Smith D, Defalla BA, Chadwick DW. The misdiagnosis of epilepsy and the management of refractory epilepsy in a specialist clinic. QJM 1999; 92: 15. 25 -Cramer JA, Glassman M, Rienzi V. The relationship between poor medication compliance and seizures. Epilepsy Behav 2002; 3: 338. 26 -Schuele SU, Lüders HO. Intractable epilepsy: management and therapeutic alternatives. Lancet Neurol 2008; 7: 514. 27 -Szaflarski JP, Ficker DM, Cahill WT, Privitera MD. Four-year incidence of psychogenic nonepileptic seizures in adults in hamilton county, OH. Neurology 2000; 55: 1561. 28 -Engel J Jr, Wiebe S, French J, et al. Practice parameter: temporal lobe and localized neocortical resections for epilepsy: report of the Quality Standards Subcommittee of the American Academy of Neurology, in association with the American Epilepsy Society and the American Association of Neurological Surgeons. Neurology 2003; 60: 538. 29 -www. uptodate. com, intractable epilepsy 30 -Dileep R, et al. management of drug resistance epilepsy, continuum, aan 2016; 157 -172

THANKS!!!! QUESTIONS ? ? ?
- Slides: 54