Intracavity Laser Absorption Spectroscopy of Nickel Fluoride in

Intracavity Laser Absorption Spectroscopy of Nickel Fluoride in the Near-Infrared James J. O'Brien Department of Chemistry & Biochemistry University of Missouri, St Louis, MO 63121 Rachel A. Harris and Leah C. O'Brien Department of Chemistry Southern Illinois University, Edwardsville, IL 62026 1

Previous Work o High-resolution spectroscopy of Ni. F started over 30 years ago o o in the UV region by Bernard Pinchemel More recently, Pinchemel and Bernath groups have studied the visible and near-IR region by laser induced fluorescence spectroscopy (LIF) and FT emission spectroscopy Energy level diagram (presented later) based on their work Additionally, Chen et al. have examined transitions in the 435 -570 nm region by LIF of Ni. F in a jet source Calculations by Zou and Liu (2006) on Ni halides, and by Koukounas and Mavridis (2008) on diatomic fluorides 2
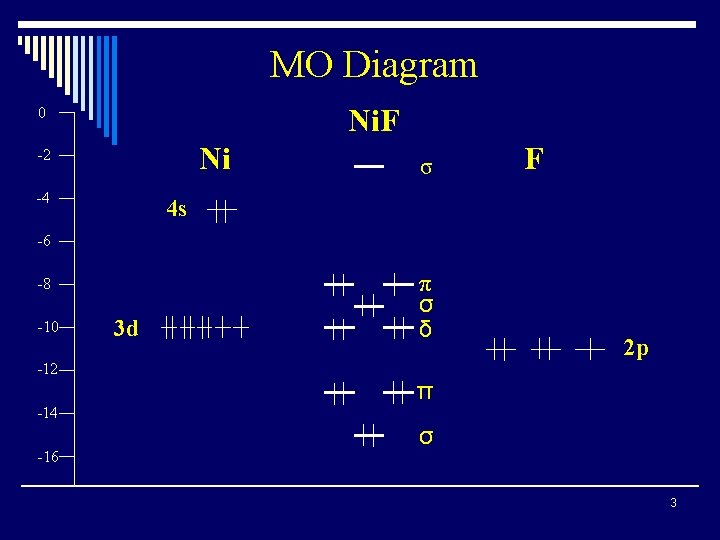
MO Diagram Ni. F 0 Ni -2 -4 σ F 4 s -6 -8 -10 3 d π σ δ -12 -14 -16 2 p π σ 3

Energy levels of Ni. F with Te < 15000 cm-1. Left: Calculated electronic states [Zou and Liu, JCP (2006)] Right: Known electronic states [Krouti, Hirao, Dufour, Boulezhar, Pinchemel, Bernath (JMS 214, 152 -174 (2002)] 4

Intracavity Laser Spectroscopy (ILS) Technique Gaseous absorber contained INSIDE resonator cavity of multimode laser that is operated in a timemodulated fashion n Absorption lines act as wavelength dependent loses, which are enhanced as the laser evolves in time n Amplified absorption lines appear superimposed on the spectrally broad output of the laser n Laser’s output is directed to a high-resolution spectrograph n 5

ILS details (Beer-Lambert relationship) ILS laser observed at well defined time after the onset of laser operation, the averaged time-resolved spectrum (for initial ~500 μs) is given by: Absorbance = ln [I 0(ν)/I(ν)] = (ν) N [c • tg • l/L], I 0(ν), I(ν) is intensity of laser without and with absorption at frequency ν, (ν) is the absorption coefficient at ν N is the number density [ pressure or concentration] c is the speed of light, 3 x 108 meter/second tg is the generation time (≈ 100 µs) l/L is the fraction of cavity occupied by the absorber i. e. , Effective absorption pathlength = [c • tg • l/L] tg determines sensitivity (Leff = 20 km for tg = 100 µs, l/L = 2/3), permits high dynamic range tg ~ 500 µs relatively easy for standing wave lasers; longer times possible with ring configured systems 1000’s miles of pathlength! • “World record” effective absorption pathlengths (Leff) is 70, 000 km [V. M. Baev and coworkers, Applied Physics B 69, 171 (1999)] 6

ILS Schematic Diagram 7

Intracavity Laser Chamber 8
![Recorded (1, 0) band of [11. 1] 2Π 3/2 – X 2Π 3/2 transition Recorded (1, 0) band of [11. 1] 2Π 3/2 – X 2Π 3/2 transition](http://slidetodoc.com/presentation_image/f75c7f417c6cff191446b221661eb3aa/image-9.jpg)
Recorded (1, 0) band of [11. 1] 2Π 3/2 – X 2Π 3/2 transition of Ni. F using ILS o Molecular source, a Nickel-lined, 2 -inch long hollow cathode o o o located inside the cavity of a Ti: sapphire laser Laser beam carries the signal to a 2 m Mc. Pherson with 1024 channel diode-array detector SF 6 as oxidant in Argon; 1. 6– 1. 7 Torr pressure Set 0. 6 Amp plasma discharge current Recorded 11680 -11725 cm-1 region; 3 cm-1 per scan For each discharge scan also record background with discharge off and divide the pair Calibrate all spectra using I 2 lines observed in an extracavity oven using ILS laser as light source 9

10

11

12
![The (0, 0) band of the [11. 1] 2Π 3/2 – X 2Π 3/2 The (0, 0) band of the [11. 1] 2Π 3/2 – X 2Π 3/2](http://slidetodoc.com/presentation_image/f75c7f417c6cff191446b221661eb3aa/image-13.jpg)
The (0, 0) band of the [11. 1] 2Π 3/2 – X 2Π 3/2 transition o The (0, 0) band of this transition is known [Pinchemel et al. , JMS 215, 262 -268 (2002)] o The ground state is known from microwave study [Tanimoto et al. , JMS 207, 66 -69 (2001)] o The [11. 1] 2Π 3/2 v=0 state required an extra parameter, a, to separate the e/f levels: n E = BJ(J+1) − DJ 2(J+1)2 ± a/2 ± p/2 (J+½) ± p. J/2 J(J+1)(J+½) o Nearby perturbing electronic state 13
![The (1, 0) band of the [11. 1] 2Π 3/2 – X 2Π 3/2 The (1, 0) band of the [11. 1] 2Π 3/2 – X 2Π 3/2](http://slidetodoc.com/presentation_image/f75c7f417c6cff191446b221661eb3aa/image-14.jpg)
The (1, 0) band of the [11. 1] 2Π 3/2 – X 2Π 3/2 transition o Bandhead at 11722. 27 cm-1 (8528. 43 Å) o Two R-branches and two P-branches o Lines assigned using microwave parameters for ground state energy levels and Δ 2 F values o A Hund’s case (c) Ω=3/2 polynomial was used to represent the energy levels for the excited and ground states: n E = BJ(J+1) − DJ 2(J+1)2 ± p/2 (J+½) ± p. J/2 J(J+1)(J+½) o Inclusion of the “a” parameter in the excited state did not improve the fit, nor was it determined by the fit o Perturber not affecting the v=1 level of the excited state 14

o 140 lines o Isotopologue structure for Ni (58 Ni, 60 Ni) was not observed o J″min = 1. 5 o J″max = 55. 5 15
![Molecular Parameters o X 2Π 3/2 and [11. 1] 2Π 3/2 v=0 values from Molecular Parameters o X 2Π 3/2 and [11. 1] 2Π 3/2 v=0 values from](http://slidetodoc.com/presentation_image/f75c7f417c6cff191446b221661eb3aa/image-16.jpg)
Molecular Parameters o X 2Π 3/2 and [11. 1] 2Π 3/2 v=0 values from Pinchemel et al. [JMS 2002] o Ground state parameters held fixed in the fit o ∆G½ = 620. 2 cm-1 for [11. 1] 2Π 3/2 o From calculations: n ωe' = 633 [Zou and Liu] n ωe' = 657 [Koukounas and Mavridis] 16
![Conclusions o The (1, 0) band of the [11. 1] 2Π 3/2 – X Conclusions o The (1, 0) band of the [11. 1] 2Π 3/2 – X](http://slidetodoc.com/presentation_image/f75c7f417c6cff191446b221661eb3aa/image-17.jpg)
Conclusions o The (1, 0) band of the [11. 1] 2Π 3/2 – X 2Π 3/2 transition of Ni. F has been recorded by intracavity laser absorption spectroscopy and analyzed to obtain the molecular parameters of the upper state o Excited state v=1 levels do not require additional “a” parameter o First metal-fluoride molecule from our lab 17

Acknowledgements o Funding from NSF (JJOB and LCOB) and PRF (LCOB) o Undergraduate student Rachel Harris at SIU Edwardsville 18
![Bond Length from [11. 1] – X data for 58 Ni 19 F 19 Bond Length from [11. 1] – X data for 58 Ni 19 F 19](http://slidetodoc.com/presentation_image/f75c7f417c6cff191446b221661eb3aa/image-19.jpg)
Bond Length from [11. 1] – X data for 58 Ni 19 F 19
- Slides: 19