Interventions to control chronic noncommunicable diseases the role


Interventions to control chronic non-communicable diseases: the role of randomised controlled trials K K CHENG 郑 家强 The University of Birmingham, United Kingdom

Conference Definition: The confusion of one man multiplied by the number present.

Evidence Based Medicine Importance of evidence in assessing effectiveness of health care interventions Randomised controlled trials as gold standards

Levels of evidence Ia Evidence obtained from meta-analysis of randomised controlled trials Ib Evidence obtained from at least one randomised controlled trial IIa Evidence obtained from at least one well-designed controlled study without randomisation IIb Evidence obtained from at least one other type of well-designed quasi-experimental study III Evidence obtained from well-designed non-experimental descriptive studies, such as comparative studies, correlation studies and case control studies IV Evidence obtained from expert committee reports or opinions and/or clinical experience of respected authorities. From Scottish Inter Collegiate Guidelines network

Two examples n Hepatocellular carcinoma prevention using selenium n Obesity prevention

Hepatocellular carcinoma (HCC) One of the most important types of cancer in Chinese populations 250, 000 male and 100, 000 female deaths annually 10 -12% of Chinese adults are lifelong carriers of the hepatitis B virus (HBV) 15 -25% of them develop HCC Possible role of selenium in reducing risk

Selenium and HCC Laboratory evidence Both initiation and post-initiation effects Essential components of antioxidant Immune system effects Potent inducer of apoptosis

Selenium and HCC Observational epidemiology Taiwan cohort (7, 342 men, 4, 841 being Hbs. Ag carriers) Mean follow up of 5. 3 years Quintile of baseline plasma selenium ----------Q 1 (lowest) Q 2 Q 3 Q 4 Q 5 (highest) Yu et al. (1999) Adjusted odds ratio for HCC (95% CI) -----------1. 00 0. 52 (0. 21 - 1. 33) 0. 32 ( 0. 09 – 1. 09) 0. 19 (0. 05 – 0. 75) 0. 62 (0. 21 – 1. 86)

Selenium and HCC Clinical trials evidence Qidong, Jiangsu Province Very high incidence area 4 clinical trials in 1980 s and 1990 s Reported in: Li WG, Zhu Y, Yan X, et al. Zhonghua Yu Fang Yi Xue Za Zhi 2000 Yu SY, Zhu YJ, Li WG, et al. Biol Trace Elem. Res 1991 Yu SY, Zhu YJ, Li WG. Biol Trace Elem. Res 1997

Township study Residents living in 5 townships at Qidoing From 1985 to 1992, a supplement of selenium fortified table salt in one township Plain table salt to other 4 control townships High risk families study 2474 members of families with high risk of HCC 1444 participants received 200 g of selenium daily and 1030 received placebo for 2 years

Hbs. Ag carriers study 1 226 HBs. Ag carriers Randomly allocated to receive 200 g of selenium or placebo daily for 4 years Hbs. Ag carriers study 2 2065 carriers 1112 received 228 g selenium daily and 953 received placebo for 3 years
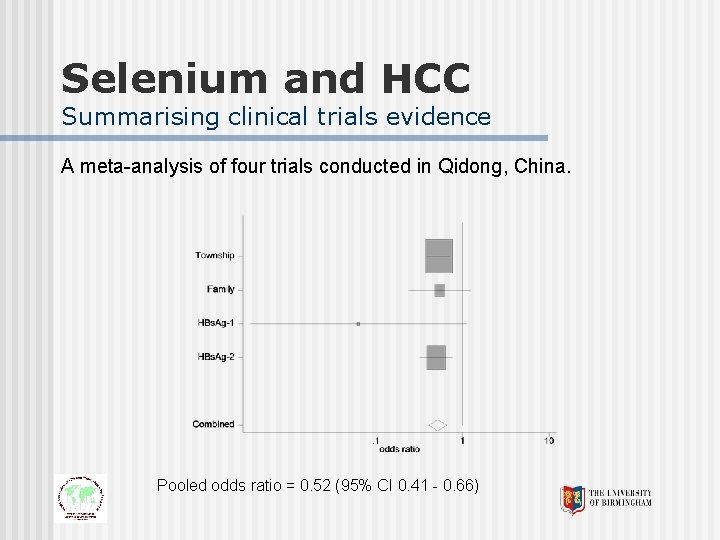
Selenium and HCC Summarising clinical trials evidence A meta-analysis of four trials conducted in Qidong, China. Pooled odds ratio = 0. 52 (95% CI 0. 41 - 0. 66)

Selenium and HCC Limitations of Qidong trials Inadequate (and/or inadequate description of) • Patient selection • Randomisation • Blinding • Baseline characteristics of treated vs placebo groups • Methods of follow up and diagnosis • Data analyses Difficulties in interpreting the results Potential loss of opportunities for prevention

Selenium and HCC If there were • well conducted and well reported clinical trials • optimal dissemination of findings HBV carriers should either be taking selenium supplements for prevention (if good trials establish a true protective effect of selenium) or they can be sure that no opportunities have been lost (if good trials show no benefit)

Obesity Likely to be one of the most important causes of chronic disease morbidity and mortality in the 21 st Century Causes cardiovascular diseases, diabetes and several types of cancers Emerging problem in Chinese populations

Obesity prevention in children Multi-agency approach needed Individual Family Education Health service Transport Agriculture Treasury

Obesity prevention in children Cochrane review (Campbell et al. ) Ten clinical trials – nine were done in western populations. One was in Thailand. Conclusions There is limited high quality data on effectiveness No generalisable conclusions can be drawn

Obesity prevention in children Interventions have to be culturally appropriate, locally acceptable and feasible Trials therefore have to be done in the local environment Outside health care settings, involving families, schools and local communities Cluster trials likely to be more appropriate • Groups of individuals (eg families, schools) as units of study • Very rarely done in Chinese populations • Require special methodological considerations

Conclusions Well conducted clinical trials are essential to determine the effectiveness of health care interventions. They may be therapeutic or preventive. They can take place in hospitals, primary care and community settings. The use of clinical trials needs to be promoted.

Ways to promote clinical trials Ensure the acceptance by health policy makers, clinicians, patients and the public of the importance of clinical trials Adequate coverage in medical education (undergraduate and postgraduate) Research funding should be earmarked for the support of strategic RCTs in key areas Clinical trials units should be set up – clinicians, trialists, statisticians, behavioural scientists, quality of life experts. . . Synthesising evidence – Cochrane Collaboration and Centres Dissemination through high quality journals Input into policy making mechanisms – new investments reserved for interventions proved to be effective?
- Slides: 21