INTERSTITIAL RESTRICTIVE LUNG DISEASES 1 GAIL FEINBERG DO

INTERSTITIAL (RESTRICTIVE) LUNG DISEASES 1 GAIL FEINBERG, DO and LIISA A. RUSSELL, MD 1

Objectives Ø Define and classify interstitial lung diseases by l l histopathology (inflammation and fibrosis vs. granulomatous) and by etiology (known vs. unknown). Distinguish epidemiologic aspects of ILDs Recognize the clinical presentation of ILDs. List the important components of evaluation of ILDs, including H&P, blood tests, imaging, special tests such as PFTs. Ø Describe therapeutic approaches and their efficacy for ILDs. Ø Discuss critical aspects of specific ILDs including Ø Ø Ø l l l l Pneumoconioses Idiopathic pulmonary fibrosis Acute cryptogenic interstitial pneumonitis ILD associated with connective tissue disorders ILD associated with medications/radiation ILD resulting from acute lung injury Hypersensitivity pneumonitis Sarcoidosis 2

General Interstitial lung diseases are chronic, nonmalignant, noninfectious diseases that cause fibrosis and remodeling of the alveolar walls. Ø They cause dyspnea and cough, show a reticular pattern of opacification on imaging and a restrictive defect on PFTs. Ø About 60 70% of the time we can identify a cause: occupational exposures (silica, asbestos), connective tissue disease, medication reaction. Ø 3

General, continued About 30 40% of the time we see ILD in people who have no known reason to develop them. These are the Idiopathic Interstitial Pneumonias and the nomenclature is confusing! Ø The treatment is steroids then cytotoxics, but response isn’t always great. In fact, some ILDs have a very poor prognosis of either inexorable progression (asbestosis) or rapid compromise (IPF/UIP, AIP). Ø 4

Terminology and Definitions ILD = interstitial lung disease ILDs = Diffuse Parenchymal Lung Diseases ILDs = a heterogeneous group of disorders that cause diffuse parenchymal lung damage, affecting the interstitium, alveolar epithelium, pulmonary capillary and lymphatic endothelium, basement membrane and perivascular and perilymphatic tissues. ILDs are grouped together because of – – – Clinical (= functional) similarity. Radiographic similarity Histologic similarity. 5

Classifying ILDs We can subdivide them according to whether the underlying cause is known or unknown Known (60 70%) Occupational and environmental exposures, e. g. pulmonary fibrosis in asbestosis, pulmonary alveolar proteinosis in acute massive silica exposure – Associated connective tissue disorder or other primary disease (chronic pulmonary edema, neurofibromatosis, lymphangioleiomyomatosis in tuberous sclerosis, vasculitides, chronic gastric aspiration Ø – 6

Classifying ILDs We can subdivide them according to whether the underlying cause is known or unknown Ø – – Unknown (30 40%) Idiopathic interstitial pneumonias. Idiopathic conditions that have distinct histology, clinical features and presentation and so are considered unique diseases – sarcoidosis, eosinophilic pulmonary diseases, pulmonary Langerhans cell histiocytosis, pulmonary alveolar proteinosis, lymphangioleiomyomatosis, . 7

WHY ALPHABET SOUP? American and British investigators have different names for the same condition. The same goes for clinicians and pathologists. 1. Usual interstitial pneumonia (UIP) aka the clinical condition of Idiopathic pulmonary fibrosis (IPF) 2. Nonspecific interstitial pneumonia (NIP or NSIP) 3. Bronchiolitis obliterans organizing pneumonia (BOOP)= cryptogenic organizing pneumonia (COP) 4. Respiratory bronchiolitis (RB) or respiratory bronchiolitis associated interstitial lung disease (RBIL) 5. Desquamative interstitial pneumonitis (DIP) 6. Acute respiratory distress syndrome (ARDS) = Diffuse Alveolar Damage (DAD) 7. Acute Interstitial Pneumonia (AIP), some call this idiopathic acute lung injury (ALI- DAD), some call this Hamman Rich syndrome. 8

CLASSIFICATION OF CHRONIC INTERSTITIAL LUNG DISEASES BY PREDOMINANT MORPHOLOGIC FEATURES There are >200 different conditions and evolving nomenclature for them! We can subdivide them according to their underlying histopathology Ø Predominantly inflammation and fibrosis (the majority) Ø Predominantly granulomatous reactions (sarcoid, hypersensitivity pneumonitis) 9

MAJOR CATEGORIES OF INTERSTITIAL LUNG DISEASE BY MORPHOLOGIC FEATURES Alveolar wall damage resulting in interstitial fibrosis: Ø Pneumoconioses Ø Collagen vascular diseases associated lung diseases Ø Drug reactions Ø Radiation pneumonitis Ø Idiopathic pulmonary fibrosis (IFP) aka cryptogenic fibrosing alveolitis: histologic pattern of usual interstitial pneumonia (UIP) and exclusion of all other causes are required for the diagnosis of IPF Ø Cryptogenic organizing pneumonia (COP), aka bronchiolitis obliterans organizing pneumonia (BOOP) Ø Non specific interstitial pneumonia (NSIP) Ø Acute lung injury ALI DAD 10

MAJOR CATEGORIES OF INTERSTITIAL LUNG DISEASE BY MORPHOLOGIC FEATURES Granulomatous inflammation resulting in interstitial fibrosis: Ø Hypersensitivity pneumonitis Ø Sarcoidosis Eosinophilic: Ø Loeffler syndrome Ø Drug allergy related Ø Idiopathic chronic eosinophilic pneumonia 11

CHRONIC NTERSTITIAL LUNG DISEASES BY ETIOLOGY Post ALI-DAD Occupational and environmental exposure Asbestosis, silicosis, coal worker’s pneumoconiosis, hypersensitivity pneumonitis, etc. Ø Drug- or treatment related Chemotherapeutic agents, radiation, oxygen Ø Smoking-related - Desquamative interstitial pneumonitis (DIP), respiratory bronchiolitsi (RB) Ø Immunologic lung disease Sarcoidosis Collagen vascular diseases Necrotizing granulomatous polyangiitis Goodpasture syndrome Allograft rejection Ø Idiopathic pulmonary fibroses Ø Other Alveolar proteinosis Ø Ø 12

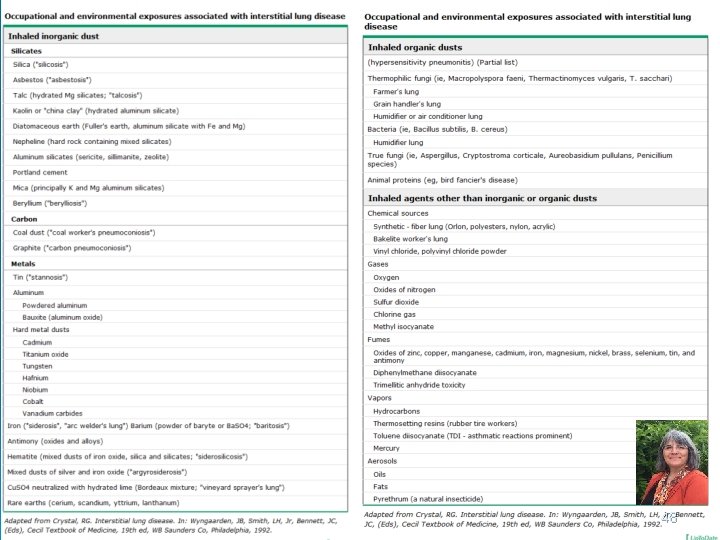
LUNG DISEASE ASSOCIATED WITH AIR POLLUTANTS Mineral dusts: Ø Coal dust, silica, asbestos, beryllium, iron oxide, zinc oxide, barium sulfate Organic dusts that induce hypersensitivity pneumonitis: Ø Moldy hay, bagasse, bird droppings Organic dusts that induce asthma: Ø Cotton, flax, hemp, red cedar dust Chemical fumes and vapors: Ø Nitrous oxide, sulfur dioxide, ammonia, benzene, insecticides Urban air 13

ØAs a PCP, I am usually just distinguishing along the top tier! ØThe further classifications will be handled by pulmonology, radiology and pathology. Why? To guide tx 14

Presentation of ILD 1. Respiratory symptoms l Progressive exertional dyspnea l Persistent, usually nonproductive cough l Hemoptysis, wheezing, chest pain less common 2. Incidental finding on CXR l Interstitial opacities in a reticular, nodular, or reticulonodular pattern l Hilar lymphadenopathy for early stage sarcoidosis 15
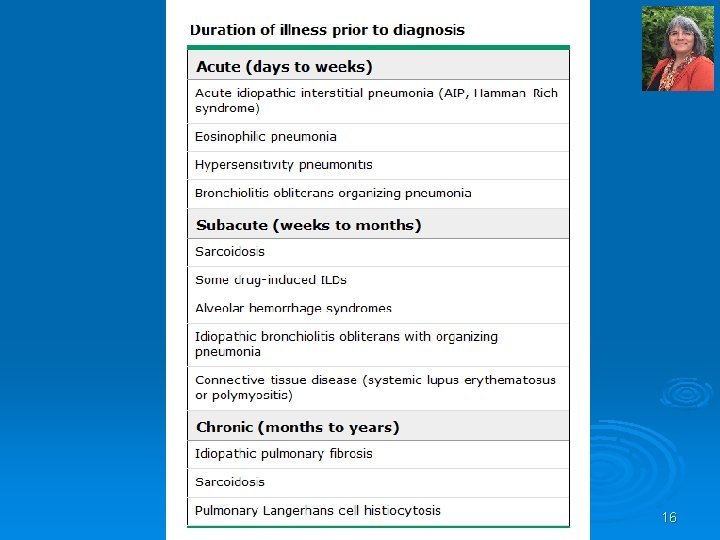
16

Relevant History Ø Age Ø Gender Ø Smoking history Ø Duration of illness Ø Prior medication use Ø Family history Ø Occupational history and environmental exposures 17

Physical Exam Findings Crackles (velcro rales) – may hear even if normal CXR Ø Inspiratory squeaks Ø Cor pulmonale – left parasternal systolic lift, loud S 2 exaggerated in the pulmonic region Ø Cyanosis – late stage Ø Clubbing – late stage Ø Extrapulmonary findings Ø 18
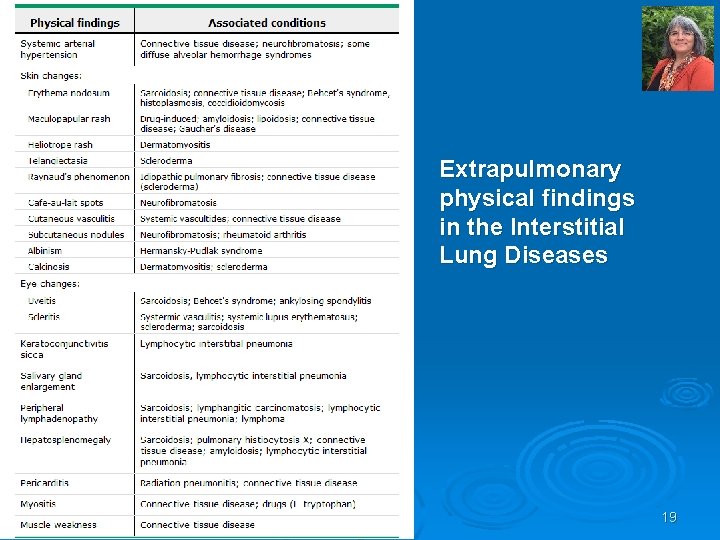
Extrapulmonary physical findings in the Interstitial Lung Diseases 19

INTERSTITIAL LUNG DISEASE WORK-UP 20

Work up of ILDs Ø History and physical exam l Ø Evaluate for known causes and coexisting dz Functional Evaluation PFTs l ABGs l Cardiopulmonary Exercise Testing Ø Imaging l CXR l HRCT Ø Tissue Diagnosis l Bronchoscopy with bronchoalveolar lavage or transbronchial biopsy l Lung biopsy l 21
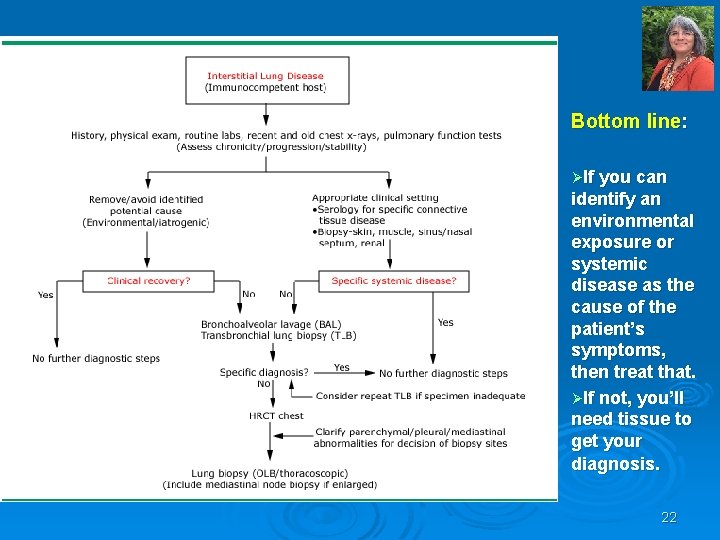
Bottom line: ØIf you can identify an environmental exposure or systemic disease as the cause of the patient’s symptoms, then treat that. ØIf not, you’ll need tissue to get your diagnosis. 22
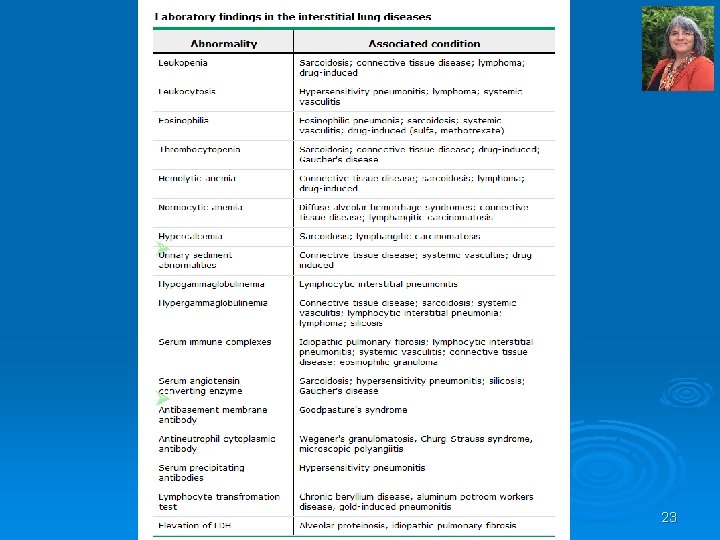

Complete PFTs 1. 2. 3. Ø Spirometry Lung volumes Diffusing capacity Most forms of ILD produce a restrictive defect with reduced total lung capacity, functional residual capacity and residual volume. Flow rates are decreased , but changes are in proportion to the decreased lung volumes; thus, the FEV 1/FVC ratio is usually normal or increased. Lung volumes decrease as lung stiffness worsens with disease progression. 24

ABGs Ø Resting arterial blood gas results may show l l Normal Hypoxemia Alkalosis CO 2 retention at end stages of disease 26

Cardiopulmonary Exercise Testing Sometimes patients with normal O 2 sat at rest quickly desaturate with exertion (or with sleep). To detect this important condition, exercise testing with pulse ox and ABGs can be performed. Ø This happens because the patient fails to decrease dead space appropriately with exercise (high VD/VT ratio). Ø 27

Imaging ILDs Ø CXR Ø High resolution CT l l Distinguishes airspace from interstitial dz Better assesses extent and distribution of disease More likely to detect underlying or coexisting dz (occult mediastinal adenopathy, carcinoma, emphysema) May help avoid biopsy if there is a diagnostic pattern 28
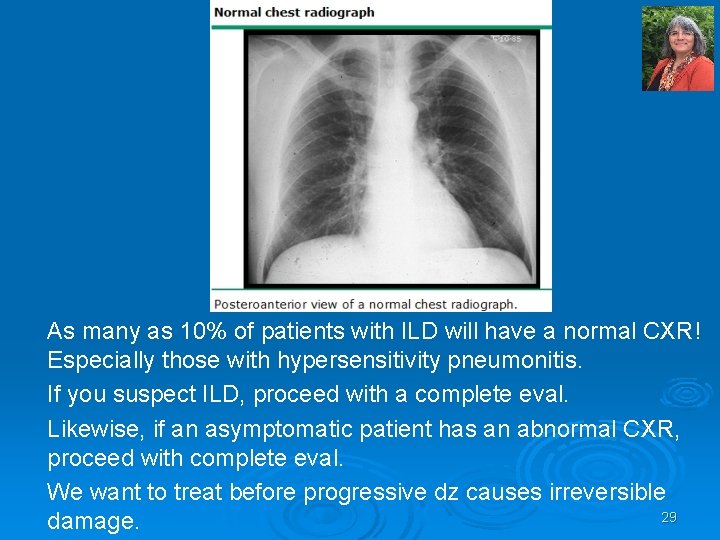
As many as 10% of patients with ILD will have a normal CXR! Especially those with hypersensitivity pneumonitis. If you suspect ILD, proceed with a complete eval. Likewise, if an asymptomatic patient has an abnormal CXR, proceed with complete eval. We want to treat before progressive dz causes irreversible 29 damage.
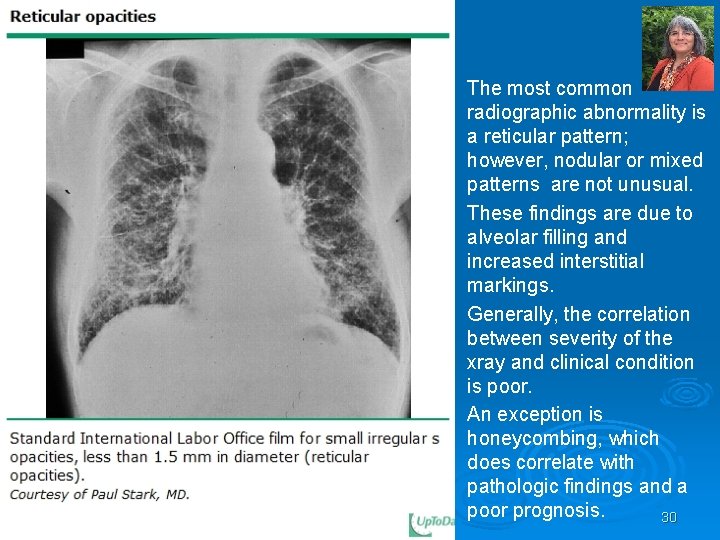
The most common radiographic abnormality is a reticular pattern; however, nodular or mixed patterns are not unusual. These findings are due to alveolar filling and increased interstitial markings. Generally, the correlation between severity of the xray and clinical condition is poor. An exception is honeycombing, which does correlate with pathologic findings and a poor prognosis. 30
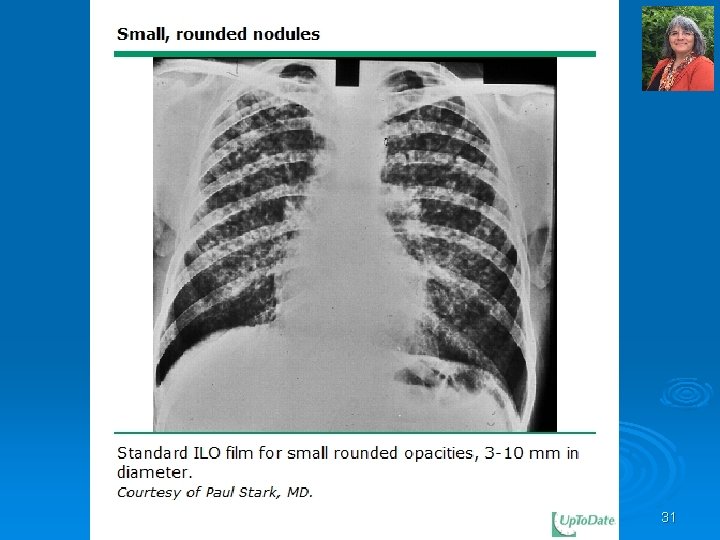
31

Indications for Tissue Diagnosis Differential DX includes: infections, malignancy, multiple types of ILDs with multiple possible associated primary diagnoses Ø Definitive diagnosis helps assess disease activity, guide appropriate therapy, and avoid the confusion and anxiety of an inaccurate prognosis. Ø Transbronchial biopsy via bronchoscopy may suffice (only, if the disease is evident in the peribronchial parenchyma as in sarcoidosis), but usually a surgical biopsy is needed (most often video asisted thoracoscopic biopsy, less often open lung biopsy) 32 Ø

Specific ILDs we are going to look at: 1. 2. 3. 4. 5. 6. 7. Pneumoconiosis Sarcoidosis Acute lung injury AIL DAD (separate lecture on ARDS DAD) Acute Interstitial Pneumonitis aka Hamman Rich Idiopathic Pulmonary Fibrosis/UIP ILD associated with Connective Tissue Disorders Radiation induced interstitial lung disease 34
GETTING BACK TO: ØSource: Interstitial Lung Diseases , Harrison's Principles of Internal Medicine, 19 e Source: Interstitial Lung Diseases, ØCitation: Kasper D, Fauci A, Hauser S, Longo D, Jameson J, Loscalzo J. Harrison's Principles of Internal Medicine, 19 e; 2015 Citation: Kasper D, Fauci A, Hauser S, Longo D, Jameson J, Loscalzo J. Harrison's 19 e; 2015 35
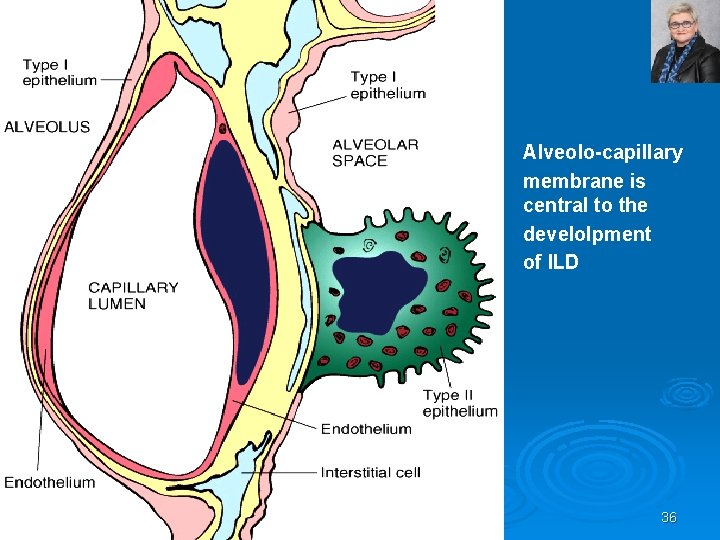
Alveolo-capillary membrane is central to the develolpment of ILD 36
WHATIZZIT? ? ? ? ? 37

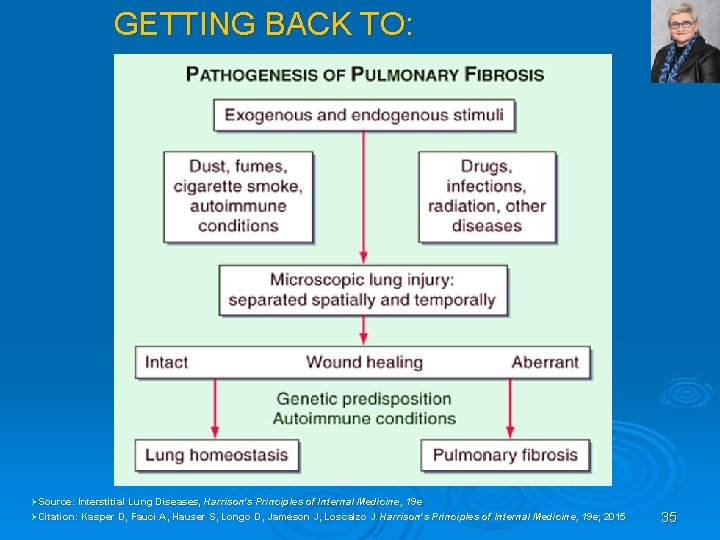
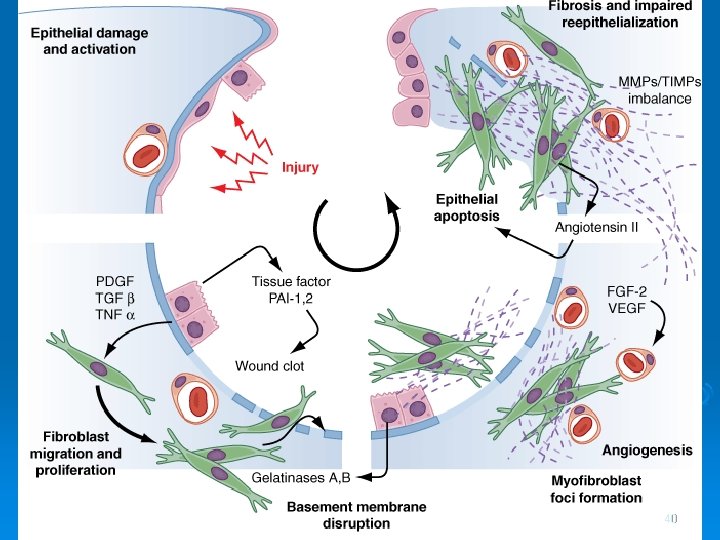
INTERSTITIAL LUNG DISEASES Pathogenesis: Ø Alveolar injury is the primary problem regardless of the type or specific cause of interstitial disease Ø In mild or self limiting injury healing is complete Ø Severe diffuse acute alveolar damage (DAD) or ongoing low grade alveolar damage heals by fibrosis and leads to loss of functioning lung parenchyma and results in respiratory insufficiency Ø Activation of pulmonary macrophages is a key event in the development of pulmonary fibrosis 38

INTERSTITIAL LUNG DISEASES Pathogenesis, continued: Ø Chemoattractants from activated macrophages activate and recruit PMNs (e. g. IL 8 and leukotriene B 4 ) Ø Oxidants and proteases from macrophages and PMNs injure type I pneumocytes and degrade lung scleroproteins Ø Type II pneumocytes derived from alveolar epithelial stem cells proliferate and repair the epithelium l Secrete chemotactic and growth factors that attract more macrophages to alveoli l Stimulate fibroblast growth (platelet derived growth factor, TGF β, and others) l Produce gelatinases (members of the matrix metallo proteinase family), which degrade basement membrane components 39
40

CASE STUDY… 41

History Ø 60 y/o male presents to the office complaining of chronic shoulder pain bilaterally with decreased work endurance. He has worked for Granite City for the past 40 years, cutting out the quartz and granite for countertops and helping to install when needed, and is having difficulty with production at work. 42

Past Medical History Ø Positive for very well controlled hypertension, otherwise negative. Ø Non smoker, occasional beer on weekends. Ø No surgeries Ø No daily meds (occasionally aspirin for shoulder pain which is really not helpful) 43

Physical Ø Other than decreased range of motion of the upper extremities bilaterally with some crepitace with ROM testing, the physical is benign. Ø …and you order xrays of both shoulders in your office (because you can), which show advanced degenerative arthritis…. however…. 44
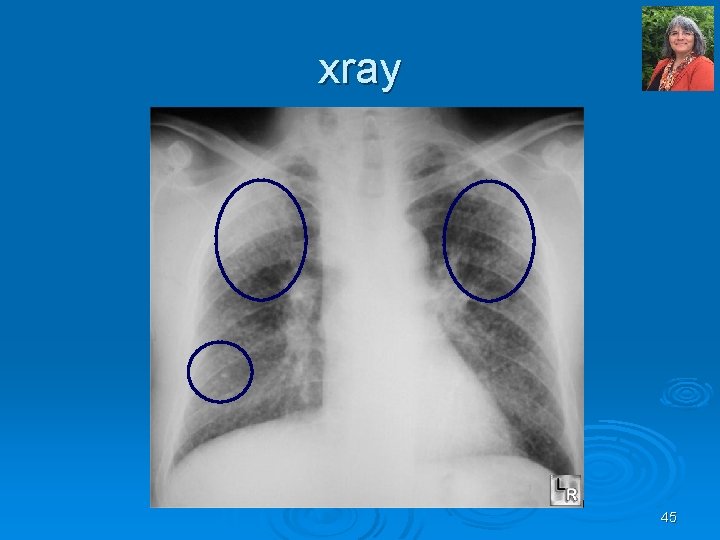
xray 45

Differential (for lung)? Ø Pneumonia Ø COPD Ø Hypersensitivity pneumonitis Ø Miliary Tuberculosis Ø Silicosis Ø Coal workers pneumoconiosis Ø Asbestosis 46

Pneumoconiosis Occupational lung disease caused by dust inhalation Anthracosis – carbon Ø Coalworkers – black lung Ø Silicosis – grinder’s dz Ø Bauxite fibrosis Ø Berylliosis Ø Siderosis – iron dust Ø Byssinosis – cotton Ø Silicosiderosis – mix of silica and iron Ø Labrador lung – miners exposed to iron, silica and anthophyllite dust (a type of asbestos) Ø Lunar lung – astronauts exposed to lunar dust Ø 47
48

PNEUMOCONIOSES GENERAL Ø Occupational lung diseases induced by inorganic and organic particulates and chemical fumes and vapors Ø The development of pneumoconiosis depends on: The amount of dust retained in the airways and lung parenchyma The size and shape of particles: The most dangerous particles measure 1 5μm, because they can reach the terminal airways and alveoli and get deposited there. Smaller particles stay in the air and get exhaled. Larger particles get stuck in the mucociliary blanket and get expectorated out. Particle solubility and physiochemical reactivity Additional effects of other irritants, such as tobacco smoke 49
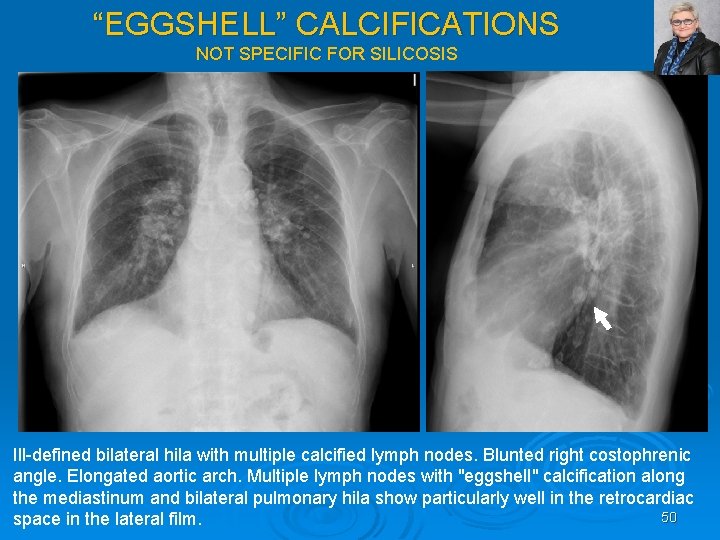
“EGGSHELL” CALCIFICATIONS NOT SPECIFIC FOR SILICOSIS Ill defined bilateral hila with multiple calcified lymph nodes. Blunted right costophrenic angle. Elongated aortic arch. Multiple lymph nodes with "eggshell" calcification along the mediastinum and bilateral pulmonary hila show particularly well in the retrocardiac 50 space in the lateral film.
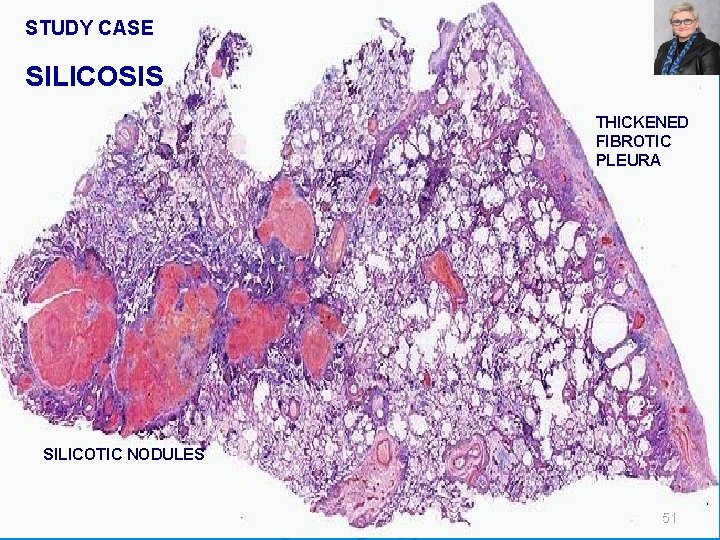
STUDY CASE SILICOSIS THICKENED FIBROTIC PLEURA SILICOTIC NODULES 51
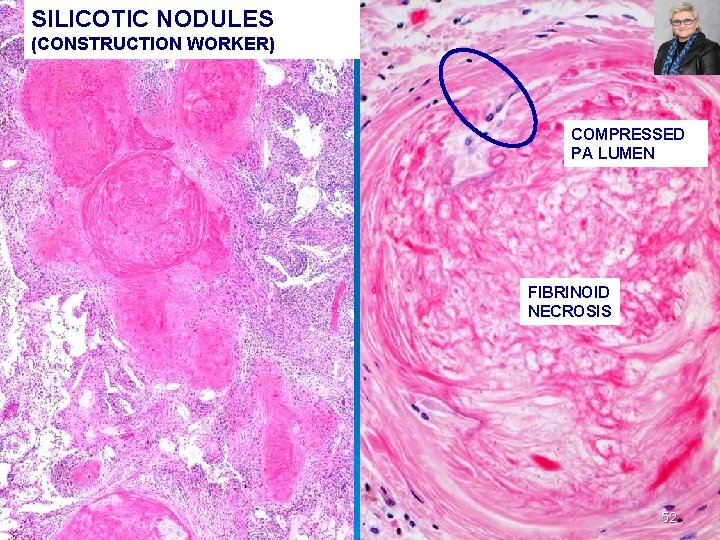
SILICOTIC NODULES (CONSTRUCTION WORKER) COMPRESSED PA LUMEN FIBRINOID NECROSIS 52
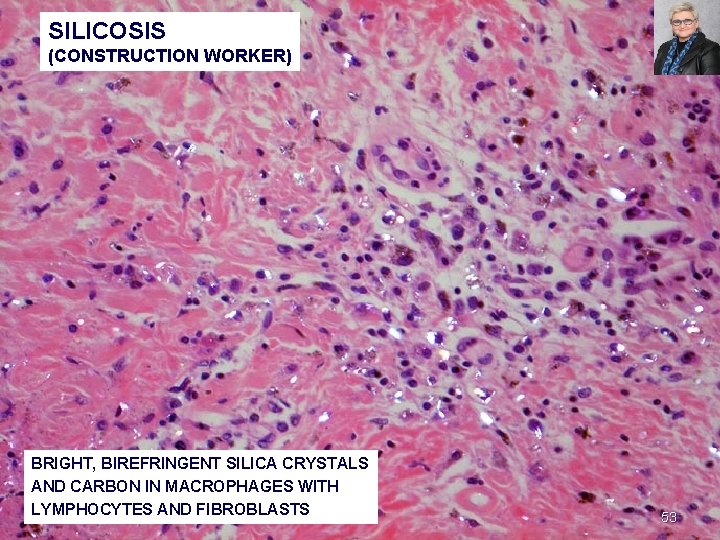
SILICOSIS (CONSTRUCTION WORKER) BRIGHT, BIREFRINGENT SILICA CRYSTALS AND CARBON IN MACROPHAGES WITH LYMPHOCYTES AND FIBROBLASTS 53
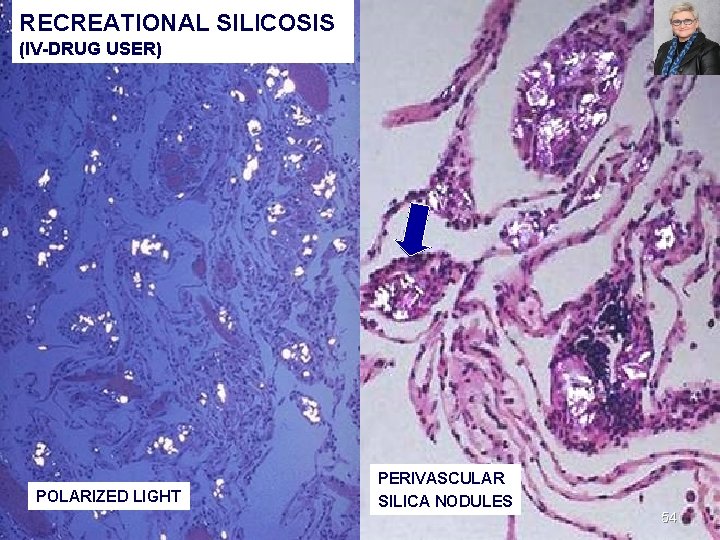
RECREATIONAL SILICOSIS (IV-DRUG USER) POLARIZED LIGHT PERIVASCULAR SILICA NODULES 54
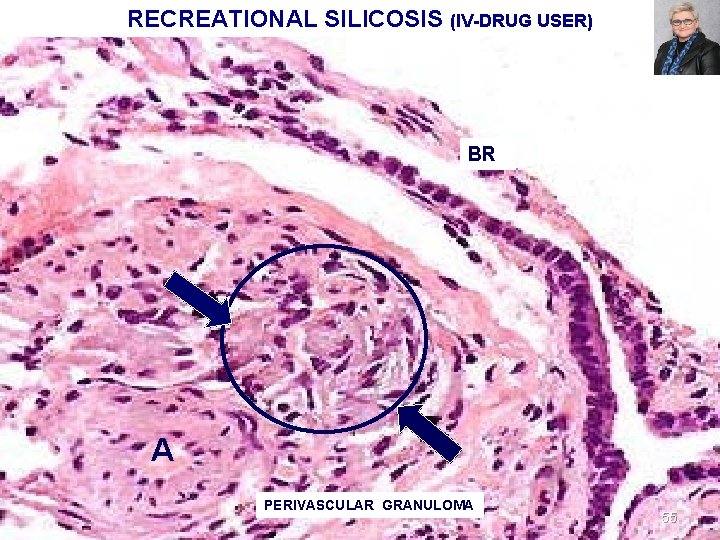
RECREATIONAL SILICOSIS (IV-DRUG USER) BR A PERIVASCULAR GRANULOMA 55
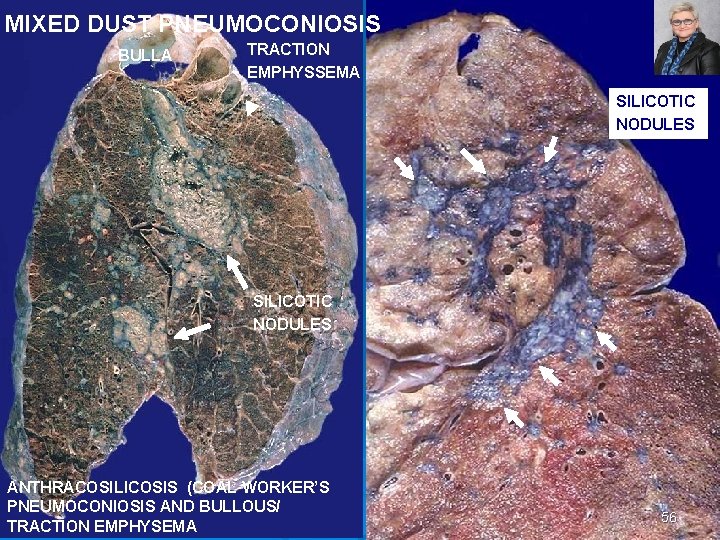
MIXED DUST PNEUMOCONIOSIS BULLA TRACTION EMPHYSSEMA SILICOTIC NODULES ANTHRACOSILICOSIS (COAL WORKER’S PNEUMOCONIOSIS AND BULLOUS/ TRACTION EMPHYSEMA 56
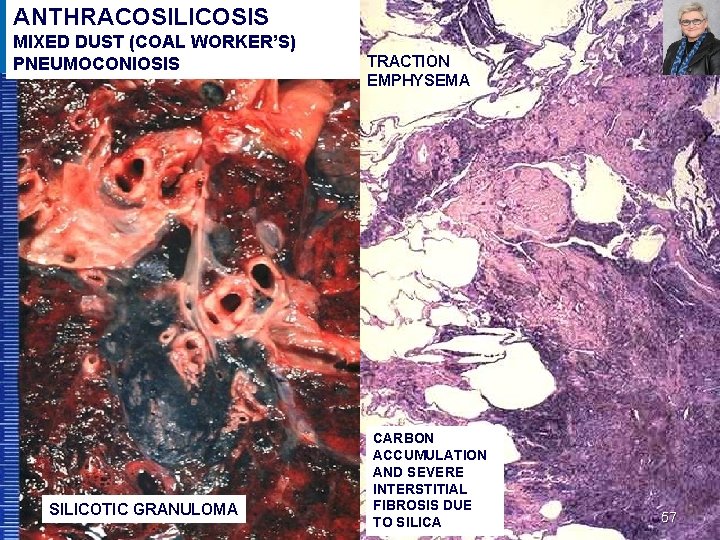
ANTHRACOSILICOSIS MIXED DUST (COAL WORKER’S) PNEUMOCONIOSIS SILICOTIC GRANULOMA TRACTION EMPHYSEMA CARBON ACCUMULATION AND SEVERE INTERSTITIAL FIBROSIS DUE TO SILICA 57
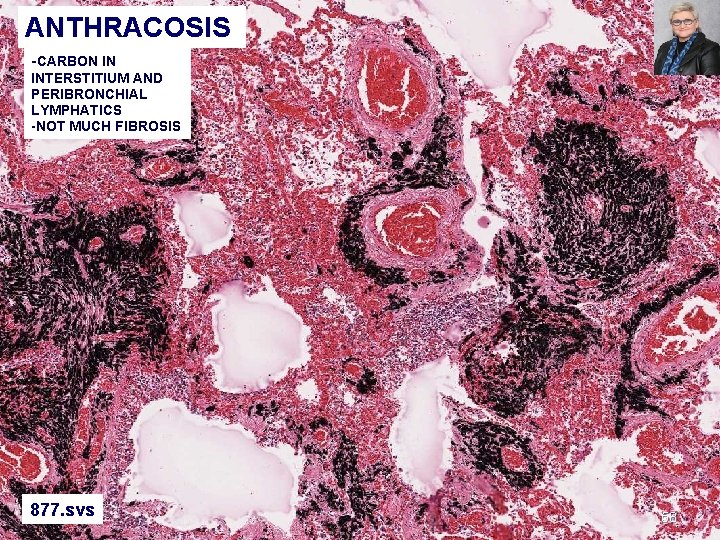
ANTHRACOSIS -CARBON IN INTERSTITIUM AND PERIBRONCHIAL LYMPHATICS -NOT MUCH FIBROSIS 877. svs 58

Asbestosis Asbestos is a mineral fiber that was used for insulation of steam pipes, houses, acoustic tiles, brake pads, and sprayed on girders from the early 20 th c. through late 1970 s when it was outlawed and abatement began. Ø Patients exposed to asbestos develop Ø l l l Pulmonary fibrosis Pleural disease focal and diffuse benign pleural plaques Pulmonary malignancies – non small cell, small cell carcinomas, malignant mesothelioma 59

Asbestosis Latency roughly 20 years from first exposure, shorter latency with heavier exposure. Victims continue to present even now (abatement workers, firefighters, etc). Ø Radiographic findings Ø l l interstitial disease pleural plaques and or calcifications 60

ASBESTOSIS The disease-causing capabilities of the different forms of asbestos depend on concentration, size, shape, and solubility. Ø Two distinct geometric forms. Ø Both are fibrogenic and increasing doses are associated with higher incidence of asbestos-related disease. Ø Ø SERPENTINE CHRYSOTILE ØAccounts for 90% of the asbestos used in industry. ØFlexible, curled structure, likely to become impacted in the upper respiratory passages and removed by the mucociliary elevator. ØChrysotiles are soluble and are gradually leached from the tissues. Ø AMPHIBOLE Ø Less prevalent, but more pathogenic than chrysotiles. Ø Straight, stiff amphiboles may align themselves in the airstream and thus can be delivered deeper into the lungs, where they can penetrate epithelial cells and reach the interstitium. Ø Penetration through the pleural surface irritates mesothelial cells and produces fibrosis. 61 Ø Carcinogenic; Induction of pleural mesothelioma.
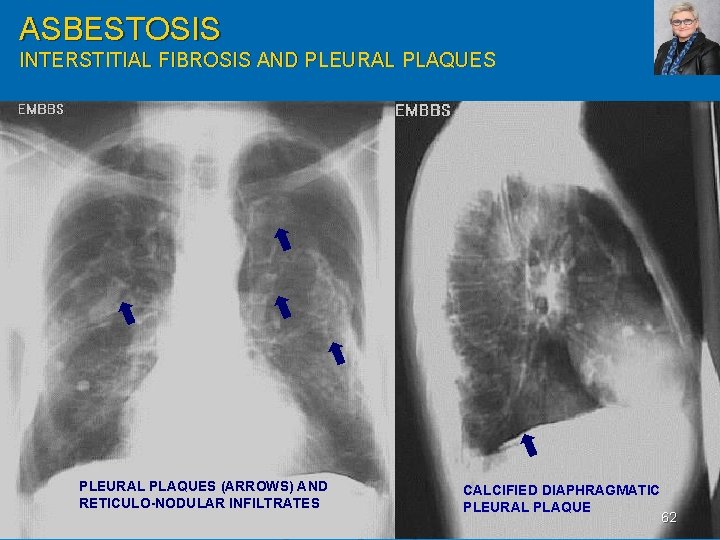
ASBESTOSIS INTERSTITIAL FIBROSIS AND PLEURAL PLAQUES (ARROWS) AND RETICULO-NODULAR INFILTRATES CALCIFIED DIAPHRAGMATIC PLEURAL PLAQUE 62
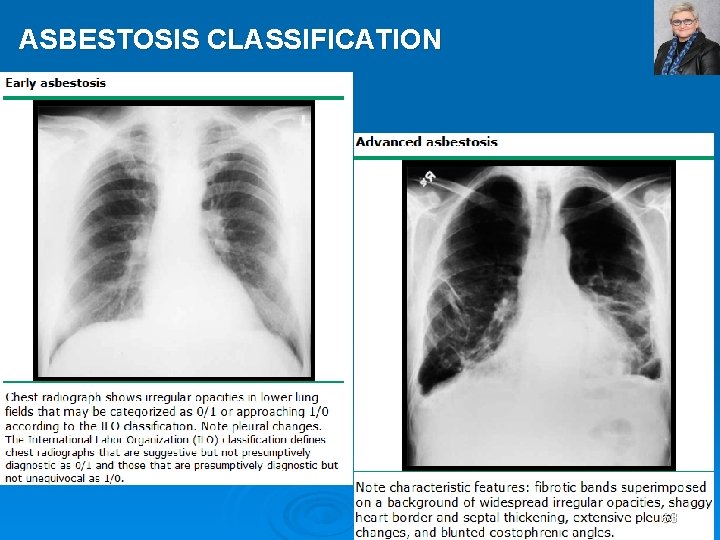
ASBESTOSIS CLASSIFICATION 63
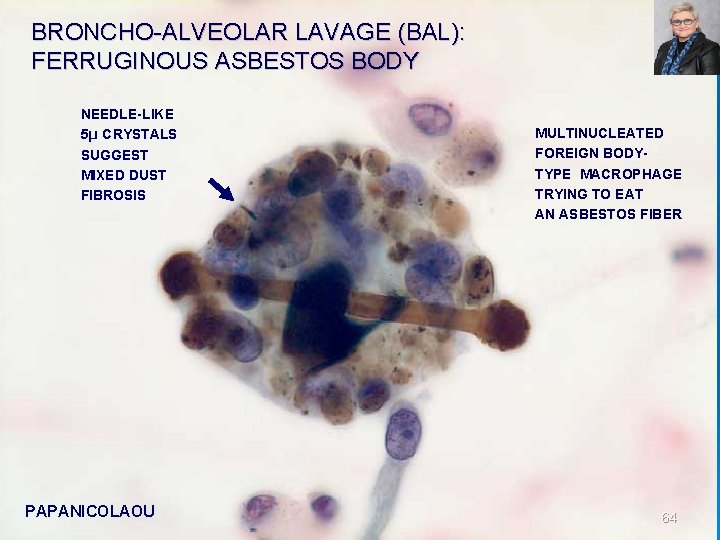
BRONCHO ALVEOLAR LAVAGE (BAL): FERRUGINOUS ASBESTOS BODY NEEDLE-LIKE 5µ CRYSTALS SUGGEST MIXED DUST FIBROSIS PAPANICOLAOU MULTINUCLEATED FOREIGN BODYTYPE MACROPHAGE TRYING TO EAT AN ASBESTOS FIBER 64
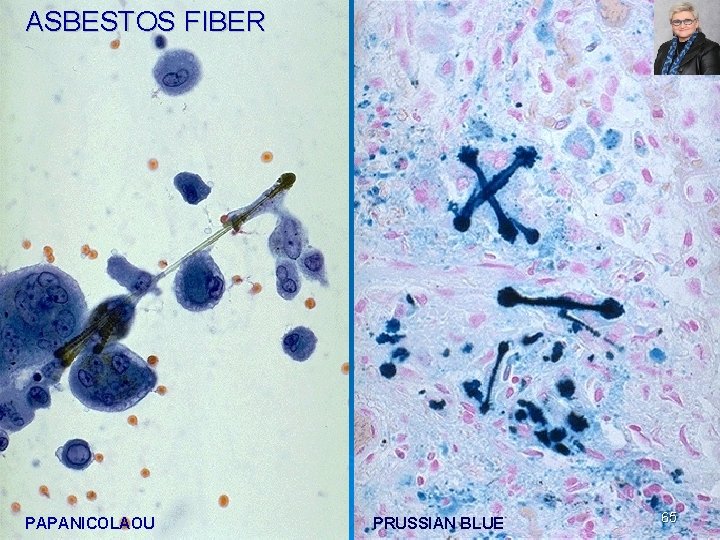
ASBESTOS FIBER PAPANICOLAOU PRUSSIAN BLUE 65
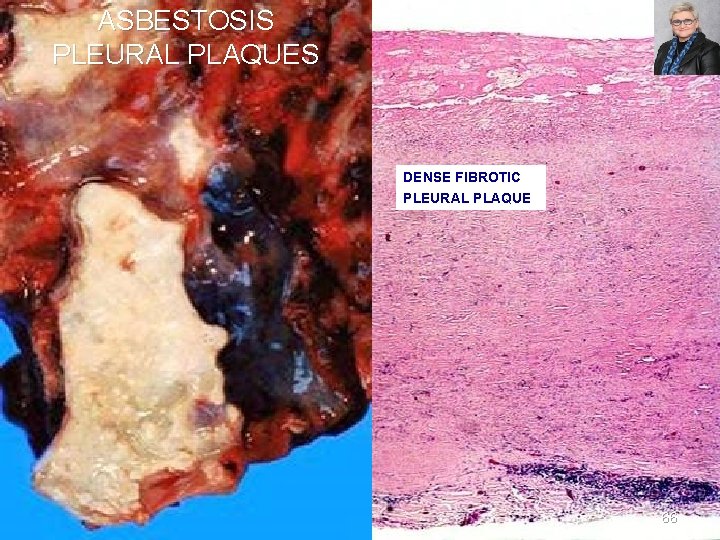
ASBESTOSIS PLEURAL PLAQUES DENSE FIBROTIC PLEURAL PLAQUE 66
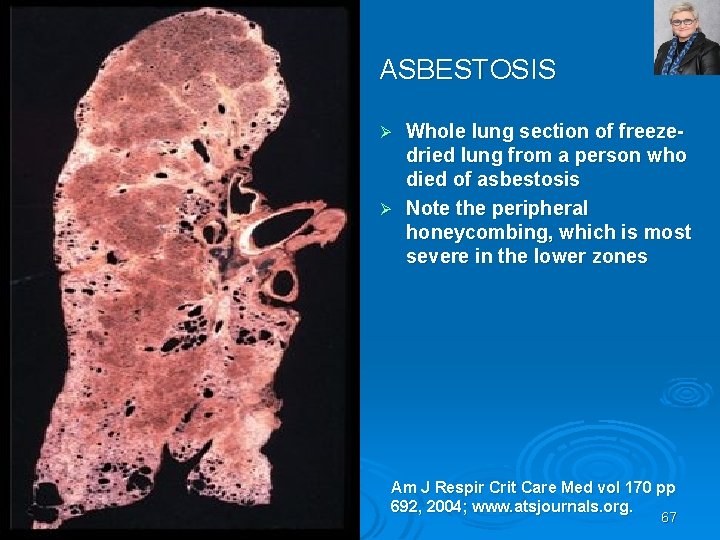
ASBESTOSIS Whole lung section of freezedried lung from a person who died of asbestosis Ø Note the peripheral honeycombing, which is most severe in the lower zones Ø Am J Respir Crit Care Med vol 170 pp 692, 2004; www. atsjournals. org. 67
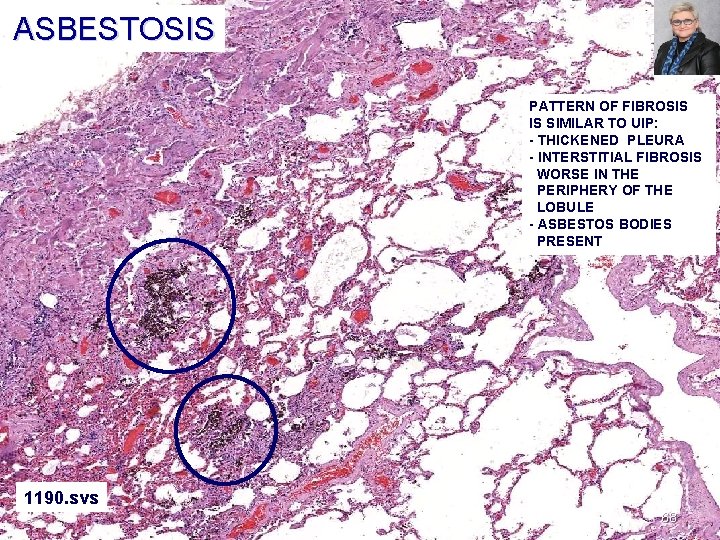
ASBESTOSIS CASE 5 PATTERN OF FIBROSIS IS SIMILAR TO UIP: - THICKENED PLEURA - INTERSTITIAL FIBROSIS WORSE IN THE PERIPHERY OF THE LOBULE - ASBESTOS BODIES PRESENT 1190. svs 68
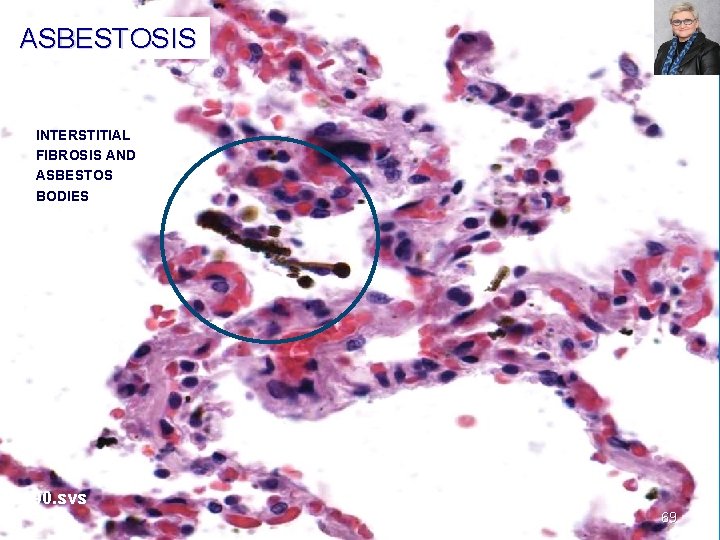
ASBESTOSIS CASE 5 INTERSTITIAL FIBROSIS AND ASBESTOS BODIES 1190. svs 69

70
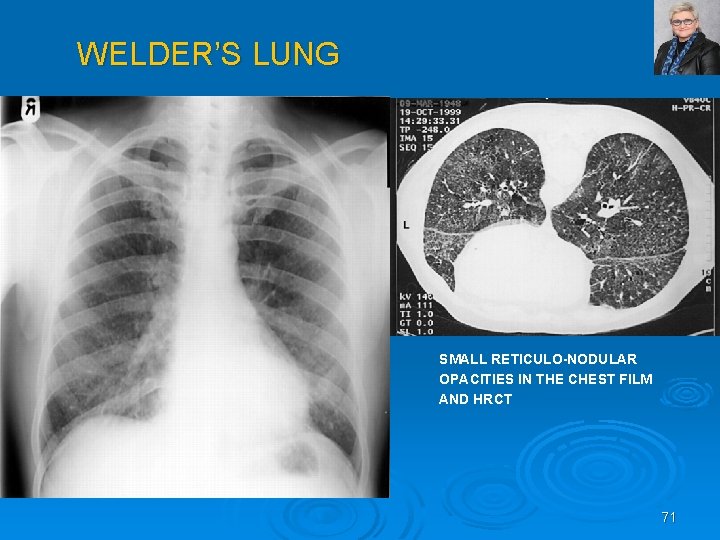
WELDER’S LUNG SMALL RETICULO-NODULAR OPACITIES IN THE CHEST FILM AND HRCT 71
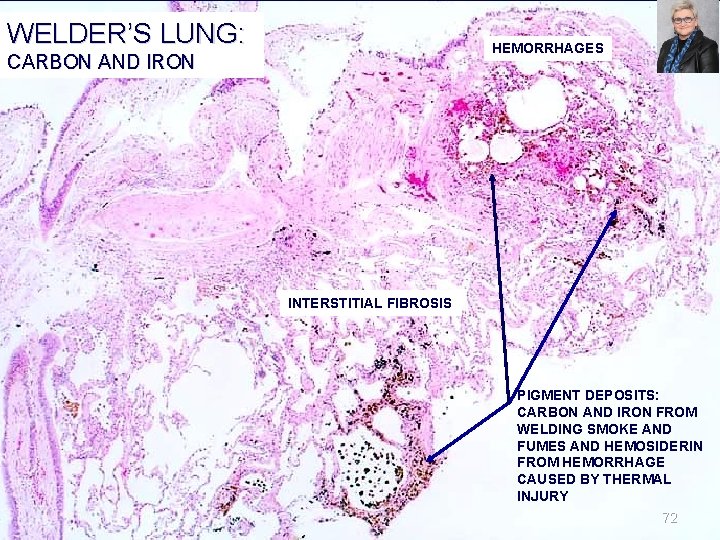
WELDER’S LUNG: HEMORRHAGES CARBON AND IRON INTERSTITIAL FIBROSIS PIGMENT DEPOSITS: CARBON AND IRON FROM WELDING SMOKE AND FUMES AND HEMOSIDERIN FROM HEMORRHAGE CAUSED BY THERMAL INJURY 72
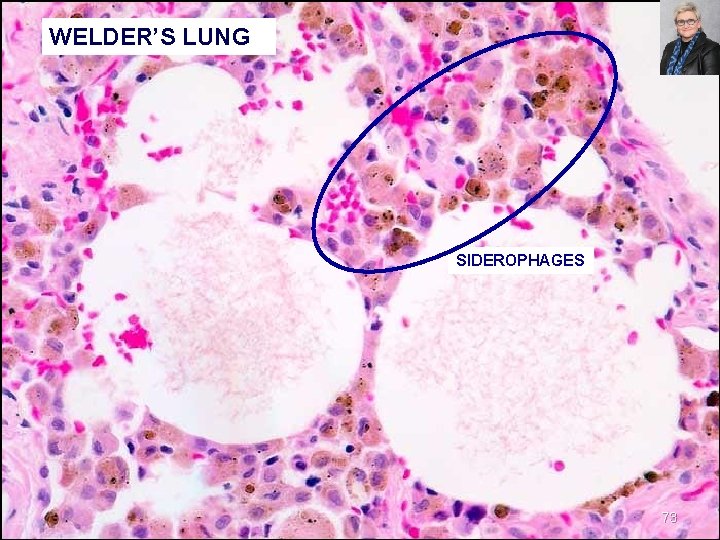
WELDER’S LUNG SIDEROPHAGES 73
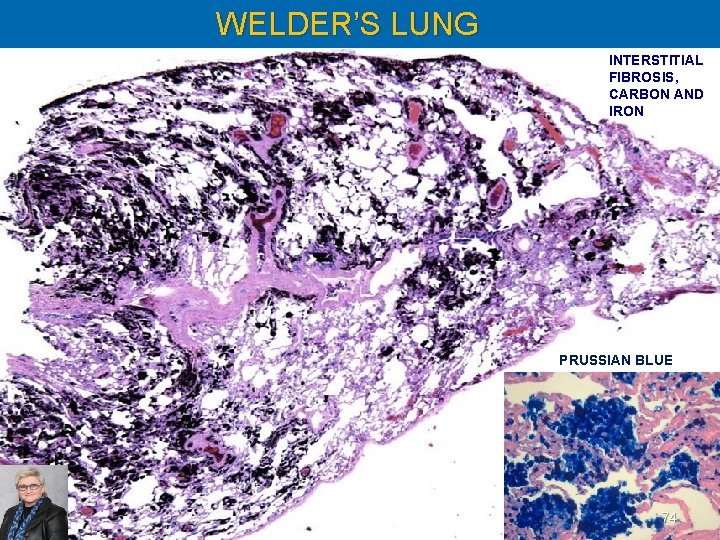
WELDER’S LUNG INTERSTITIAL FIBROSIS, CARBON AND IRON PRUSSIAN BLUE 74

IDIOPATHIC FIBROSING INTERSTITIAL LUNG DISEASES 75
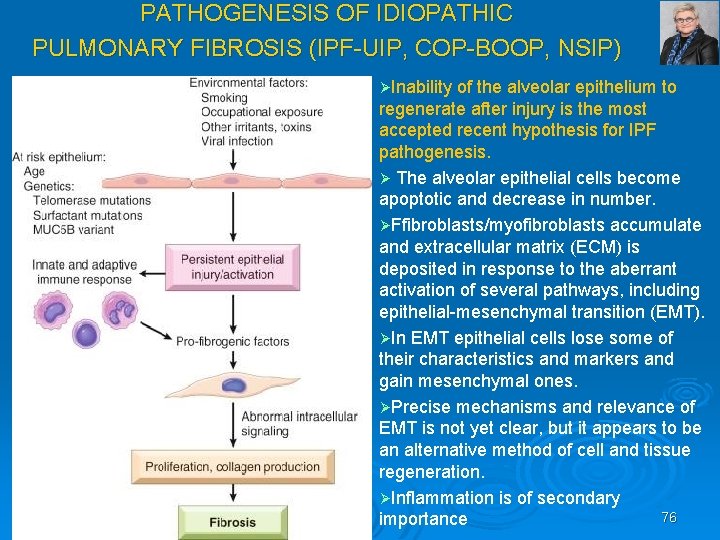
PATHOGENESIS OF IDIOPATHIC PULMONARY FIBROSIS (IPF UIP, COP BOOP, NSIP) ØInability of the alveolar epithelium to regenerate after injury is the most accepted recent hypothesis for IPF pathogenesis. Ø The alveolar epithelial cells become apoptotic and decrease in number. ØFfibroblasts/myofibroblasts accumulate and extracellular matrix (ECM) is deposited in response to the aberrant activation of several pathways, including epithelial mesenchymal transition (EMT). ØIn EMT epithelial cells lose some of their characteristics and markers and gain mesenchymal ones. ØPrecise mechanisms and relevance of EMT is not yet clear, but it appears to be an alternative method of cell and tissue regeneration. ØInflammation is of secondary 76 importance

IDIOPATHIC PULMONARY FIBROSIS (IPF) USUAL INTERSTITIAL PNEUMONIA (UIP) CRYPTOGENIC FIBROSING ALVEOLITIS Ø Ø Ø A clinicopathologic syndrome marked by progressive interstitial pulmonary fibrosis and respiratory failure. IPF has characteristic clinical, radiologic, and pathologic features. IPF has characteristic appearance in CT scans. The histologic pattern of fibrosis is referred to as usual interstitial pneumonia (UIP) The UIP pattern can also be seen in other diseases, e. g. connective tissue diseases, chronic hypersensitivity pneumonia, and asbestosis, which must be differentiated from IPF based on other clinical, laboratory, and histological features. 77

Idiopathic Pulmonary Fibrosis (IPF) Other names: Cryptogenic fibrosing alveolitis Usual interstitial pneumonia (UIP) Ø Unknown etiology Ø The most common of the idiopathic interstitial pneumonias Ø Insidious onset: patients present with chronic (>12 wk) exertional dyspnea, non productive cough and inspiratory crackles; digital clubbing may be present Ø Males are affected more often than females Ø 60% of patients are older than 60 at the time of presentation Ø Immune mechanism is suspected Ø PFTs usually show a restrictive pattern with reduced Dlco Ø Patients have arterial hypoxemia, which is elicited or exaggerated by exercise. Ø 78
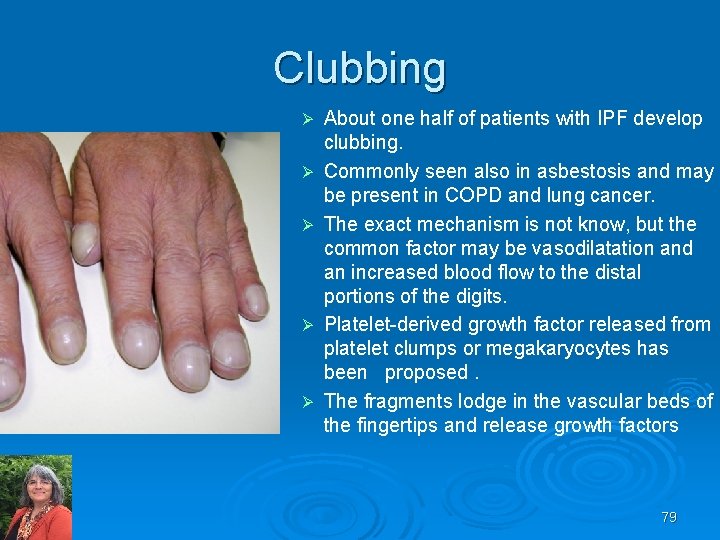
IPF Clubbing Ø Ø Ø About one half of patients with IPF develop clubbing. Commonly seen also in asbestosis and may be present in COPD and lung cancer. The exact mechanism is not know, but the common factor may be vasodilatation and an increased blood flow to the distal portions of the digits. Platelet derived growth factor released from platelet clumps or megakaryocytes has been proposed. The fragments lodge in the vascular beds of the fingertips and release growth factors 79
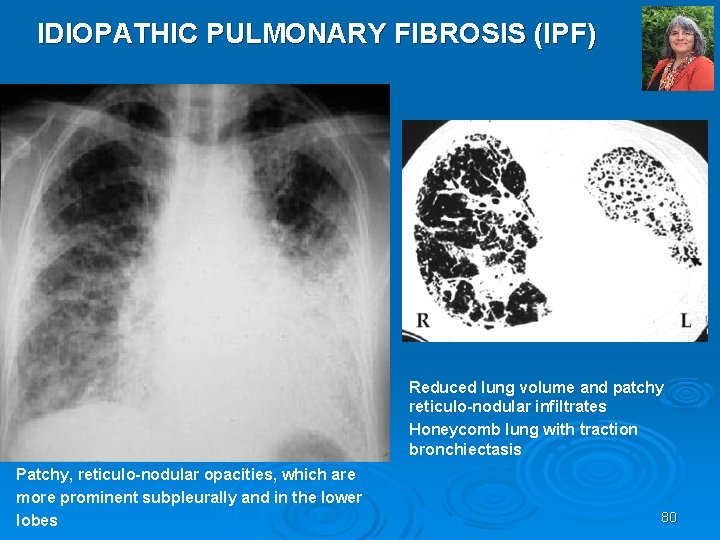
IDIOPATHIC PULMONARY FIBROSIS (IPF) Reduced lung volume and patchy reticulo-nodular infiltrates Honeycomb lung with traction bronchiectasis Patchy, reticulo-nodular opacities, which are more prominent subpleurally and in the lower lobes 80
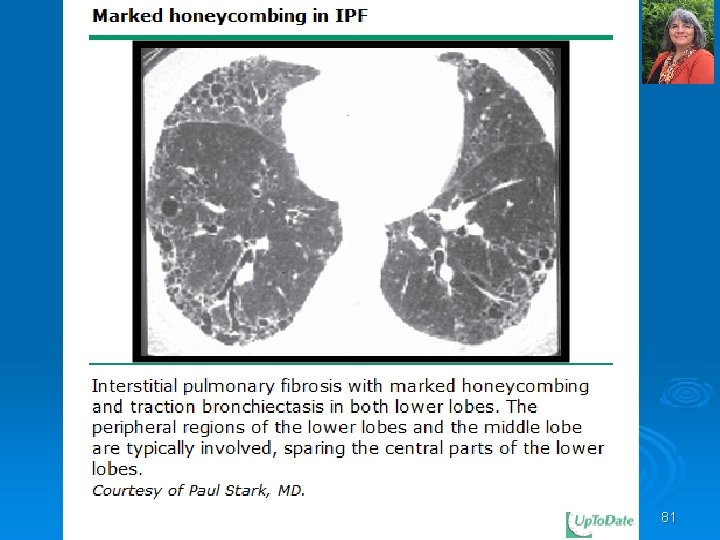
81
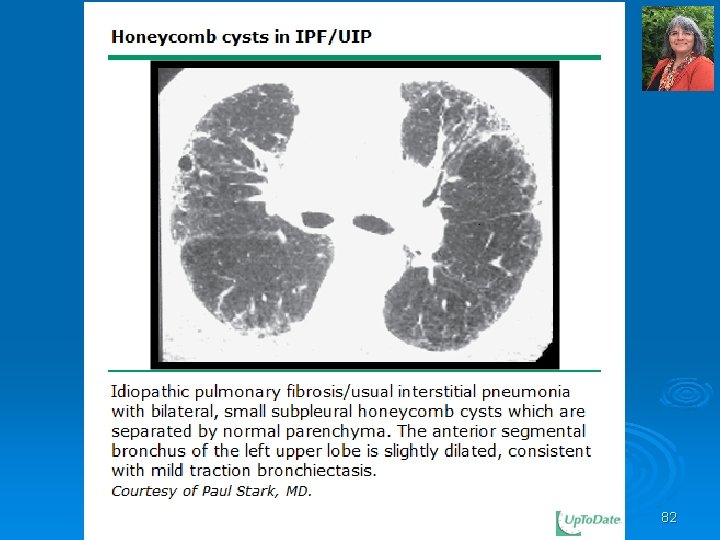
82

IPF Prognosis Ø Ø Distinctly poor response to therapy 50 70% mortality within 5 years. Surgical biopsy is usually required for a correct diagnosis. Acute deterioration often occurs either from acute exacerbation of the IPF itself, super infections, pulmonary embolism, pneumothorax or heart failure. 83
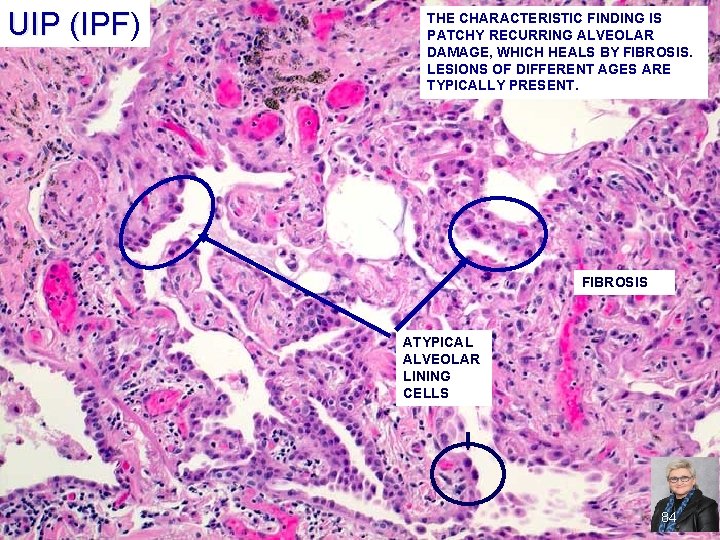
UIP (IPF) UIP (IPF THE CHARACTERISTIC FINDING IS PATCHY RECURRING ALVEOLAR DAMAGE, WHICH HEALS BY FIBROSIS. LESIONS OF DIFFERENT AGES ARE TYPICALLY PRESENT. FIBROSIS ATYPICAL ALVEOLAR LINING CELLS 84
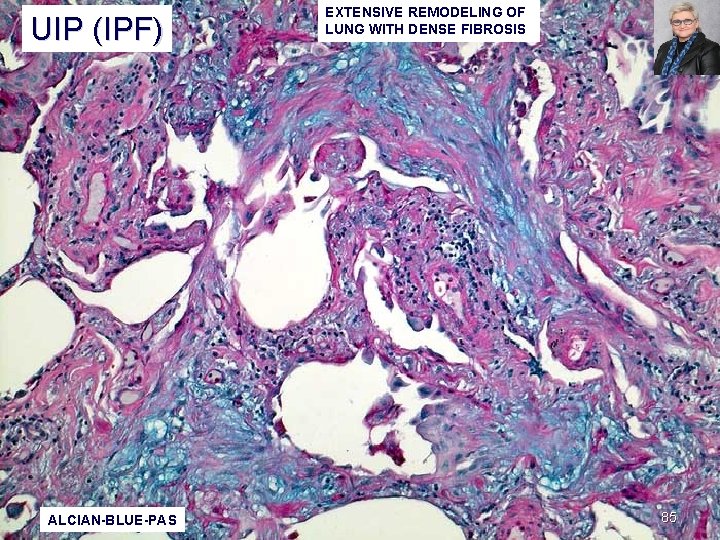
UIP (IPF) ALCIAN-BLUE-PAS EXTENSIVE REMODELING OF LUNG WITH DENSE FIBROSIS 85
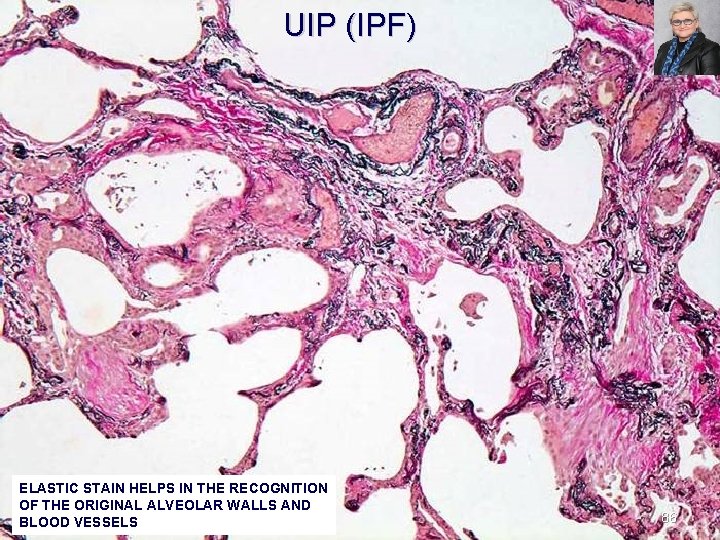
UIP (IPF) ELASTIC STAIN HELPS IN THE RECOGNITION OF THE ORIGINAL ALVEOLAR WALLS AND BLOOD VESSELS 86
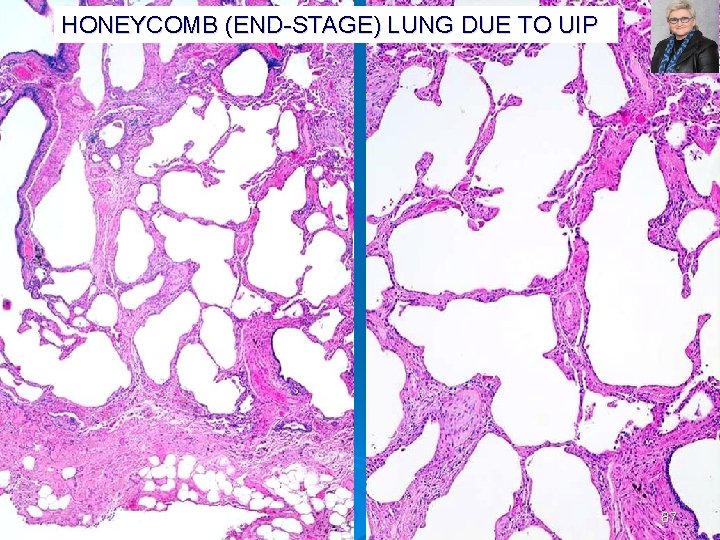
HONEYCOMB (END STAGE) LUNG DUE TO UIP PLEURA 87
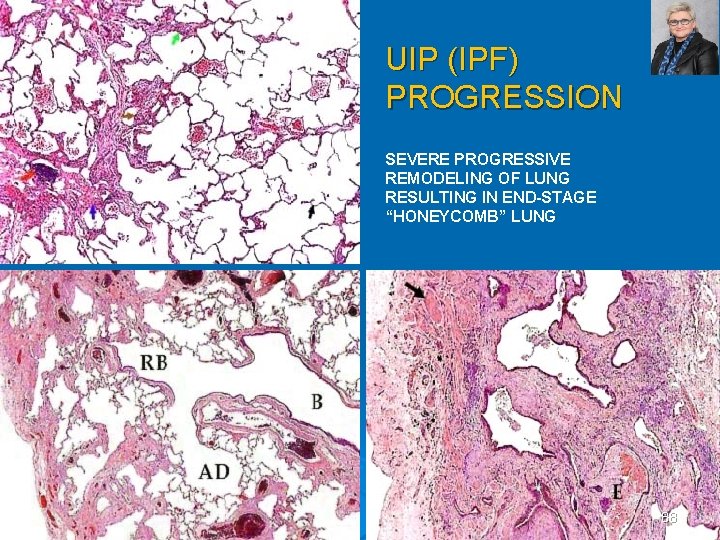
UIP (IPF) PROGRESSION SEVERE PROGRESSIVE REMODELING OF LUNG RESULTING IN END-STAGE “HONEYCOMB” LUNG 88
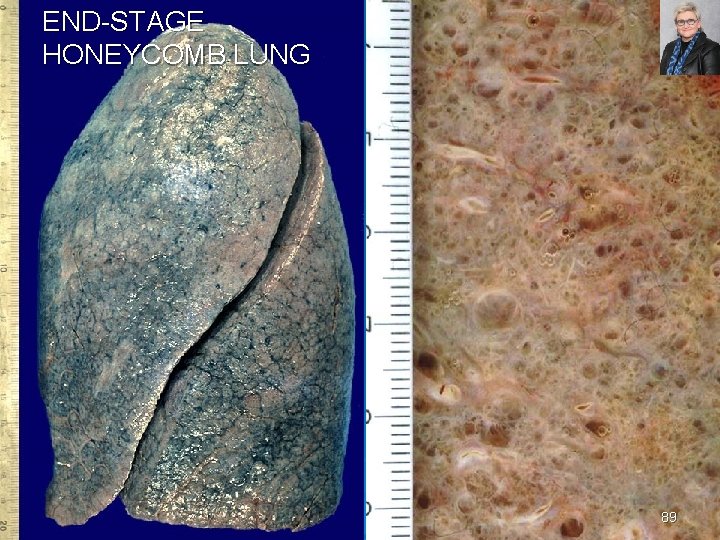
END STAGE HONEYCOMB LUNG 89

CRYPTOGENIC ORGANIZING PNEUMONIA (COP) AKA BRONCHIOLITIS OBLITERANS ORGANIZING PNEUMONIA (BOOP ) Clinical features: Cough and dyspnea of unknown etiology Some patients recover spontaneously, but most need treatment with oral steroids for 6 months or longer for complete recovery. Ø Radiologic features: Patchy subpleural or peribronchial areas of airspace consolidation Ø Histologic features: - Healing lung injury pattern; resembles DAD, but has a patchy - distribution - Polypoid plugs of loose, organizing connective tissue (Masson bodies) in alveolar ducts, alveoli and bronchioli Connective tissue is all of the same age in contrast to UIP No honeycomb changes usually Ø 90
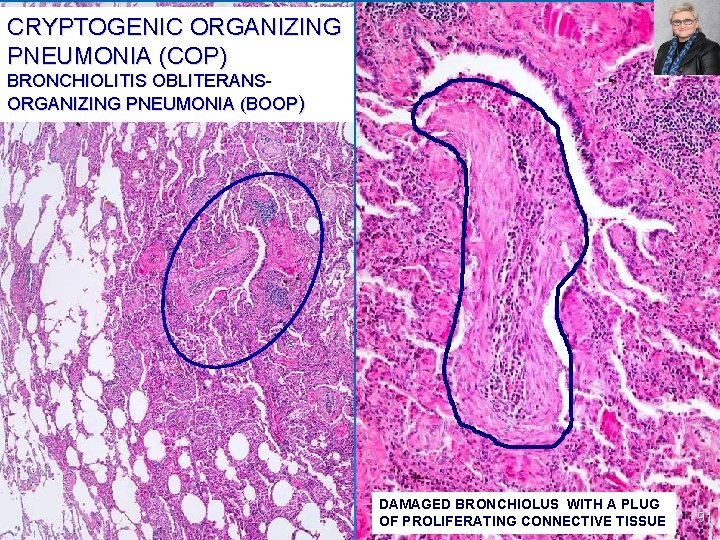
CRYPTOGENIC ORGANIZING PNEUMONIA (COP) BRONCHIOLITIS OBLITERANS ORGANIZING PNEUMONIA (BOOP) DAMAGED BRONCHIOLUS WITH A PLUG OF PROLIFERATING CONNECTIVE TISSUE 91
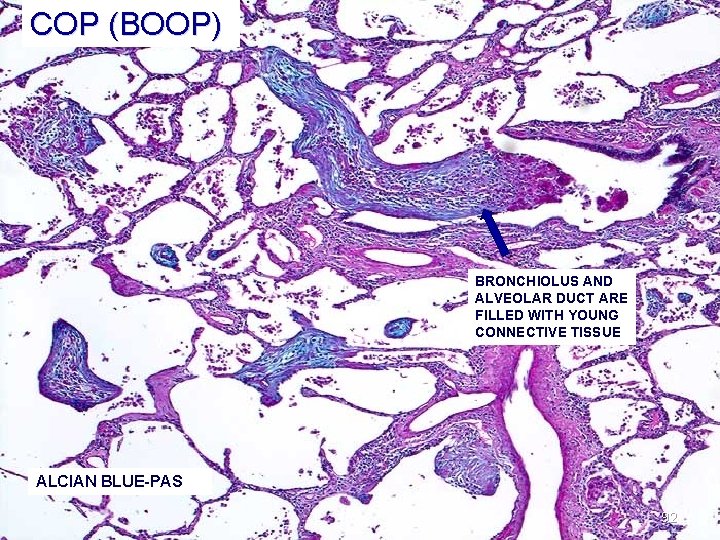
COP (BOOP) BRONCHIOLUS AND ALVEOLAR DUCT ARE FILLED WITH YOUNG CONNECTIVE TISSUE ALCIAN BLUE-PAS 92
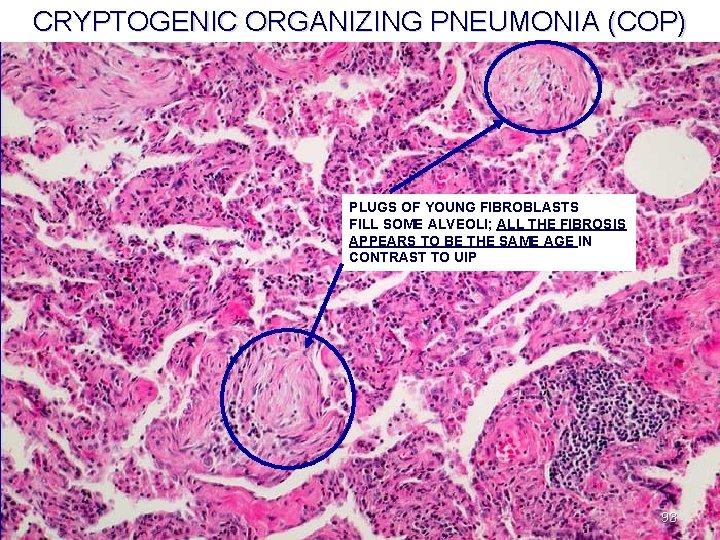
CRYPTOGENIC ORGANIZING PNEUMONIA (COP) PLUGS OF YOUNG FIBROBLASTS FILL SOME ALVEOLI; ALL THE FIBROSIS APPEARS TO BE THE SAME AGE IN CONTRAST TO UIP 93
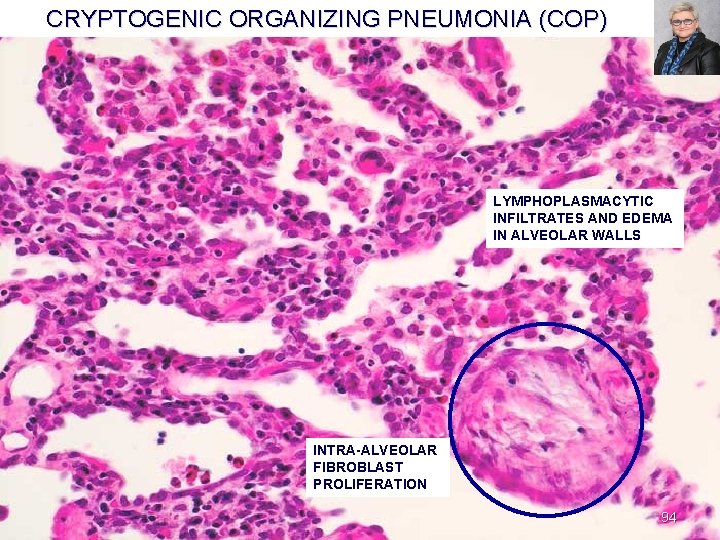
CRYPTOGENIC ORGANIZING PNEUMONIA (COP) LYMPHOPLASMACYTIC INFILTRATES AND EDEMA IN ALVEOLAR WALLS INTRA-ALVEOLAR FIBROBLAST PROLIFERATION 94

NONSPECIFIC INTERSTITIAL PNEUMONIA (NSIP) ØPatients present with dyspnea and cough of several months' duration. ØMore likely to be 50 60 year old female nonsmokers ØBilateral, symmetric, predominantly lower lobe reticular opacities in high resolution CT ØPatients have a diffuse interstitial lung disease, but lung biopsies lack the diagnostic features of any of the other well characterized interstitial diseases. ØThis entity is important to recognize, because these patients typically have a much better prognosis than those with usual interstitial pneumonia. ØNonspecific interstitial pneumonia may be idiopathic or associated with connective tissue disease, e. g. rheumatoid arthritis. 95

NONSPECIFIC INTERSTITIAL PNEUMONIA (NSIP) Morphology: Ø Cellular pattern: Mild to moderate chronic interstitial inflammation in a uniform or patchy distribution with lymphocytes and a few plasma cells Ø Fibrosing pattern: Diffuse or patchy interstitial fibrotic lesions of roughly the same stage of development (an important distinction from UIP) No hyaline membranes, granulomas, fibroblastic foci, or honeycombing usually Ø Prognosis: Patients with the cellular pattern are younger than those with the fibrosing pattern and have a better prognosis Ø 96

PULMONARY INVOLVEMENT IN AUTOIMMUNE (CONNECTIVE TISSUE) DISEASES Ø Many autoimmune diseases, particularly systemic lupus erythematosus, rheumatoid arthritis, progressive systemic sclerosis (scleroderma), dermatomyositis polymyositis, and ankylosing spondylitis can involve the dermatomyositis polymyositis, and ankylosing spondylitis can lung at some point in their course and can manifest in several histologic patterns. Ø The most common patterns: Nonspecific interstitial pneumonia Usual interstitial pneumonia Vascular sclerosis Organizing pneumonia Bbronchiolitis Pulmonary involvement in these diseases has a variable prognosis that is determined by the extent and histologic pattern of involvement. Ø 97

PULMONARY INVOLVEMENT IN AUTOIMMUNE (CONNECTIVE TISSUE) DISEASES CONTINUED Rheumatoid arthritis : Pulmonary involvement in 30% to 40% of patients Chronic pleuritis, with or without effusion Diffuse interstitial pneu monitis and fibrosis Intrapulmonary rheumatoid nodules Follicular bronchiolitis Pulmonary hypertension Ø Systemic sclerosis (scleroderma): Diffuse interstitial fibrosis (NSIP more common than UIP) and pleural involvement Ø Lupus erythematosus : Patchy, transient parenchymal infiltrates Occasionally severe lupus pneumonitis Pleurisy and pleural effusions Ø 98
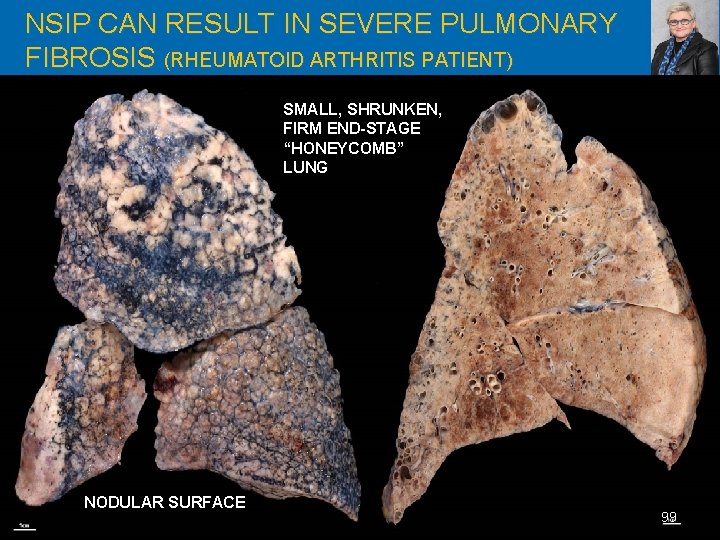
NSIP CAN RESULT IN SEVERE PULMONARY FIBROSIS (RHEUMATOID ARTHRITIS PATIENT) SMALL, SHRUNKEN, FIRM END-STAGE “HONEYCOMB” LUNG NODULAR SURFACE 99

Acute Interstitial Pneumonia aka Hamman Rich Syndrome Ø Rare. Ø Acute presentation with fever, cough, dyspnea x 1 2 wk. Ø Fulminant course with rapid development of Acute Respiratory Distress Syndrome. Ø CXR with diffuse, bilateral, opacification; HRCT ground glass attentuation, air space consolidation. Ø >60% mortality within 6 months. 100

ACUTE INTERSTITIAL PNEUMONIA (AIP) Ø Pathologists do not have a problem with AIP, but do not like the diagnosis called Hamman Rich syndrome, because as it was originally reported, it was not a distinct clinical or histologic entity. Ø After the original paper was published and the cases examined more closely, it turned out that they were a combination of diffuse alveolar damage in various stages, viral infections and COP. Ø The closest histologic equivalent of Acute Interstitial Pneumonia is acute lung injury ALI DAD. 101

ROBBINS: ROBBINS ACUTE INTERSTITIAL PNEUMONIA (AIP) Ø Ø Ø Acute interstitial pneumonia is a term that is used to describe widespread ALI of unknown etiology associated with a rapidly progressive clinical course. It is sometimes referred to as idiopathic ALI DAD. It is an uncommon disorder that occurs at a mean age of 59 years and has no sex predilection. Patients present with acute respiratory failure often following an illness of less than 3 weeks' duration that resembles an upper respiratory tract infection. The radiographic and pathologic features are identical to those of the organizing stage of ALI. The mortality rate varies from 33% to 74%, with most deaths occurring within 1 to 2 months. Recurrences and chronic interstitial disease may occur in the survivors. 102

BOTTOM LINE ABOUT IPF/UIP AND AIP/HAMMAN RICH Both are idiopathic interstitial pneumonias. UIP is more chronic, patients are usually older than 50. Histologically the lesions are of varying ages Poor prognosis, only 3 5 years. AIP is more acute, patients are younger, and all lesions are histologically the same age. Fulminant course of only months. Bottom Line: If you see Hamman Rich as a boards question, know it’s an idiopathic interstitial pneumonia with a fulminant course and a poor prognosis. 103

OBSTRUCTIVE VS. RESTRICTIVE LUNG DISEASE COMPARISON OBSTRUCTIVE RESTRICTIVE Inflammation in airway walls Inflammation/injury in alveolar walls Thickening of airway walls Thickening of alveolar walls Hyperplasia and metaplasia of airway Hyperplasia and metaplasia of epithelium alveolar epithelium Restructuring of lung Decrease of tissue mass Increase of tissue mass Irregular thin-walled spaces Irregular thick-walled spaces Loss of capillary bed Thickening of arterial walls Loss of lung parenchyma/vascular bed 104
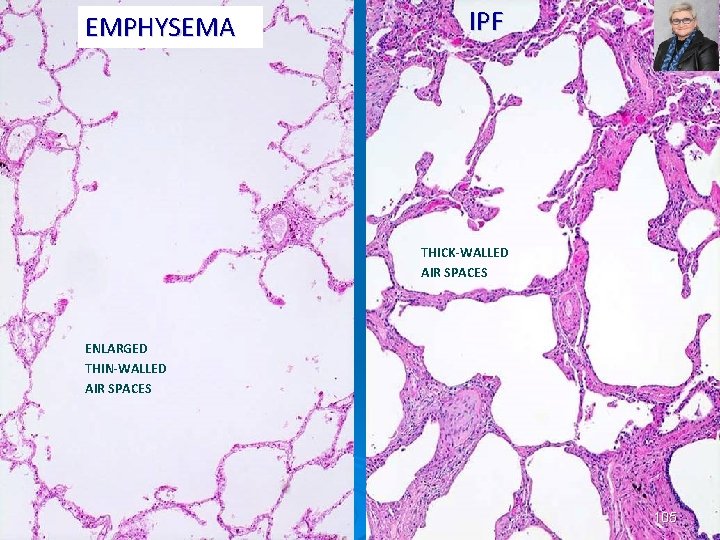
EMPHYSEMA IPF THICK-WALLED AIR SPACES ENLARGED THIN-WALLED AIR SPACES 105

QUESTIONS? 106
- Slides: 104