Interpretation of Arterial Blood Gases James Wigfull Critical

Interpretation of Arterial Blood Gases James Wigfull Critical Care and Anaesthesia james. wigfull@sth. nhs. uk

What do you get? • • p. H p. O 2 (Fi. O 2) p. CO 2 Bicarbonate Base Excess Sodium Potassium Chloride • • Calcium Lactate Haemoglobin COHb/Met. Hb Sa. O 2 Strong Ion Diff Strong Ion Gap Anion Gap

What do you get? • • p. H p. O 2 (Fi. O 2) p. CO 2 Bicarbonate Base Excess Sodium Potassium Chloride • • Calcium Lactate Haemoglobin COHb/Met. Hb Sa. O 2 Strong Ion Diff Strong Ion Gap Anion Gap

Oxygen content

Variable performance device 30 Flow 6 0 Time

Hydrogen ion homoeostasis Extracellular H+ = 40 nmol/l Intracellular Varies in different subcellular compartments (7. 17 to 6. 69) Mean p. H varies between cell types Skeletal muscle 7. 07 Proximal renal tubule 7. 13

p. H is a logarithmic scale p. H 7. 80 7. 70 7. 60 7. 50 7. 40 7. 30 7. 20 7. 10 7. 00 6. 90 6. 80 [H+] nmol/l 16 20 26 32 40 50 63 80 100 125 160
![Importance of [H+] H+ ions have a greater affinity for negatively charged proteins than Importance of [H+] H+ ions have a greater affinity for negatively charged proteins than](http://slidetodoc.com/presentation_image_h2/524eacd17aed7cabebc28136da618d8a/image-8.jpg)
Importance of [H+] H+ ions have a greater affinity for negatively charged proteins than Na+ or K+ Any change to [H+] causes net loss or gain of H+ ions bound to proteins Because H+ ions are substantially smaller than Na+ or K+ such changes affect the charge distribution and subsequently tertiary protein structure
![Importance of [H+] Example: Glycolysis Enzyme function is inversely proportional to [H+] BUT different Importance of [H+] Example: Glycolysis Enzyme function is inversely proportional to [H+] BUT different](http://slidetodoc.com/presentation_image_h2/524eacd17aed7cabebc28136da618d8a/image-9.jpg)
Importance of [H+] Example: Glycolysis Enzyme function is inversely proportional to [H+] BUT different enzymes have different sensitivities to p. H changes Acidosis inhibits glycolysis overall but the aerobic pathway is inhibited more than the anaerobic pathway Lactate rises proportionately to [H+].

Strong vs Weak Strong ions & acids are those that completely dissociate in water Weak ions and acids are those that incompletely dissociate in water

Buffers A weak acid or base that can donate or accept hydrogen ions in relationship to the concentration of free hydrogen ions in solution, allowing for relatively large changes in total hydrogen ion content to take place with relatively little change in free (ionized) hydrogen ion concentration.
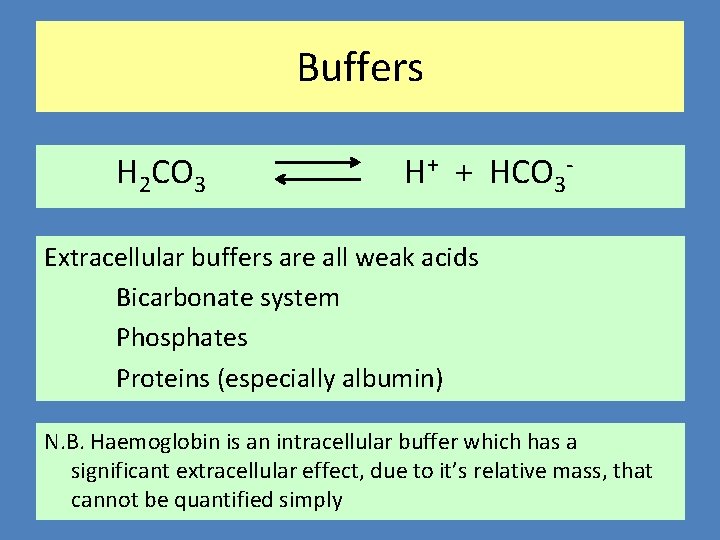
Buffers H 2 CO 3 H+ + HCO 3 - Extracellular buffers are all weak acids Bicarbonate system Phosphates Proteins (especially albumin) N. B. Haemoglobin is an intracellular buffer which has a significant extracellular effect, due to it’s relative mass, that cannot be quantified simply

Total Carbon Dioxide Carbonic Anhydrase CO 2 + H 2 O H 2 CO 3 H+ + HCO 3 -

Bicarbonate Actual bicarbonate is calculated from p. H and p. CO 2 p. H = 6. 1 + log[HCO 3 -]/(0. 23 xp. CO 2) Standard Bicarbonate (removes respiratory influence) the concentration of HCO 3 - that would be present if: Sa. O 2 = 100% temp = 37 o p. CO 2 = 5. 33 k. Pa

Base Excess BE is the amount of acid or base required to return 1 litre of blood to ‘normal’ mean standard bicarbonate of 22. 9 m. Eq/l BE = 1. 2 (s. HCO 3 - - 22. 9)

Compensation Mechanisms Respiratory • Chemoreceptors located on the ventral surface of the medulla • Respond to changes in p. H of CSF • Lower p. H stimulates respiration Metabolic (Renal) • Control of SID by removal of Cl- from extracellular fluid in response to acidosis • Initially by transcellular pumps • Subsequently by urinary loss • Concomitant increase in HCO 3 - is due to maintenance of electrical neutrality

A survival guide How to interpret ABGs if biology was simple Principles to remember: 1. Increased CO 2 reduces p. H 2. Increased HCO 3 increases p. H 3. Respiratory compensation alters CO 2 in response to primary metabolic change 4. Renal compensation alters HCO 3 in response to primary respiratory change 5. No compensatory mechanism can overshoot (iatrogenic intervention can)
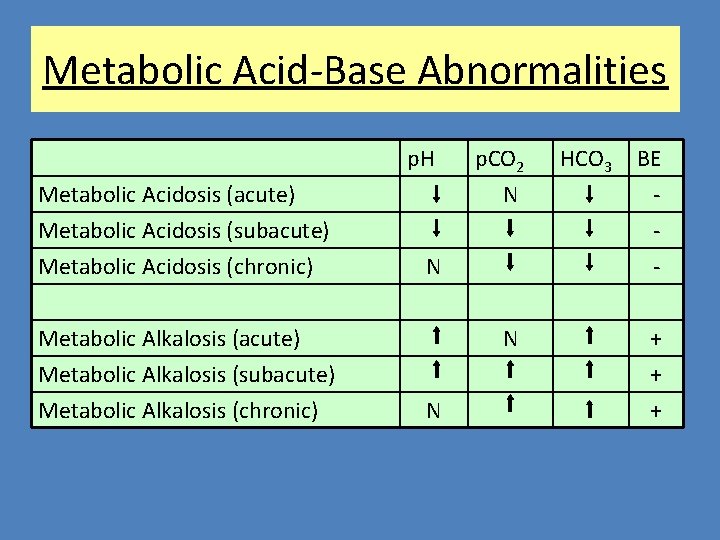
Metabolic Acid-Base Abnormalities p. H Metabolic Acidosis (acute) Metabolic Acidosis (subacute) Metabolic Acidosis (chronic) Metabolic Alkalosis (acute) Metabolic Alkalosis (subacute) Metabolic Alkalosis (chronic) p. CO 2 N N HCO 3 BE + + +
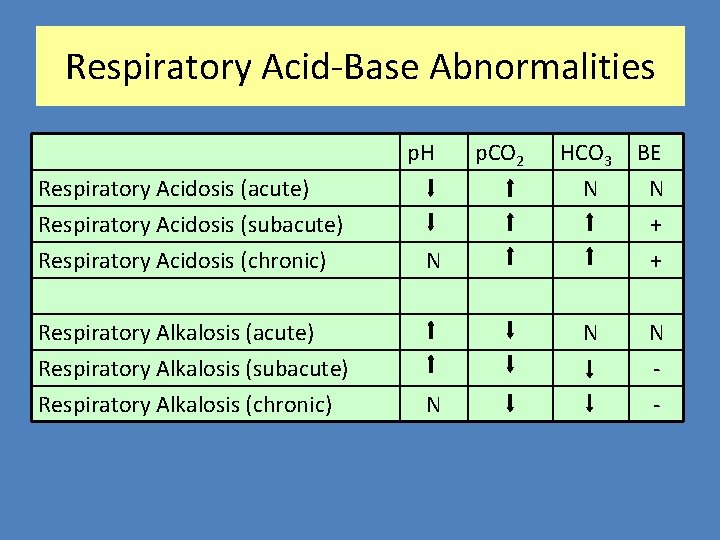
Respiratory Acid-Base Abnormalities p. H Respiratory Acidosis (acute) Respiratory Acidosis (subacute) Respiratory Acidosis (chronic) Respiratory Alkalosis (acute) Respiratory Alkalosis (subacute) Respiratory Alkalosis (chronic) p. CO 2 HCO 3 N BE N + + N N - N N
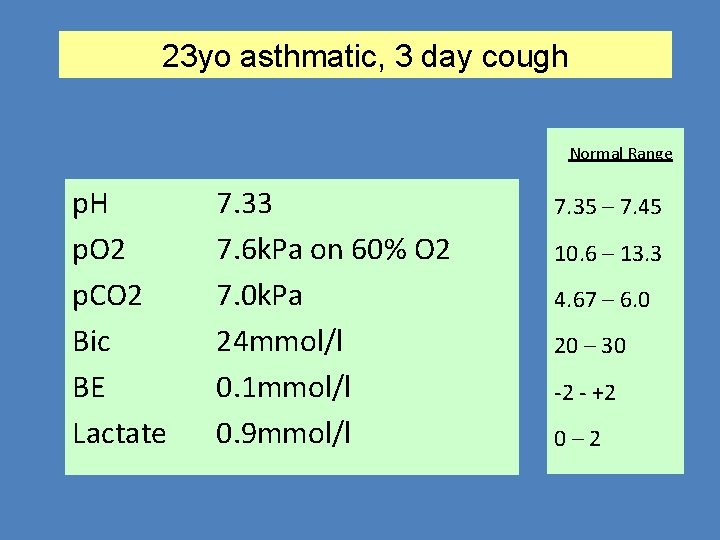
23 yo asthmatic, 3 day cough Normal Range p. H p. O 2 p. CO 2 Bic BE Lactate 7. 33 7. 6 k. Pa on 60% O 2 7. 0 k. Pa 24 mmol/l 0. 1 mmol/l 0. 9 mmol/l 7. 35 – 7. 45 10. 6 – 13. 3 4. 67 – 6. 0 20 – 30 -2 - +2 0– 2
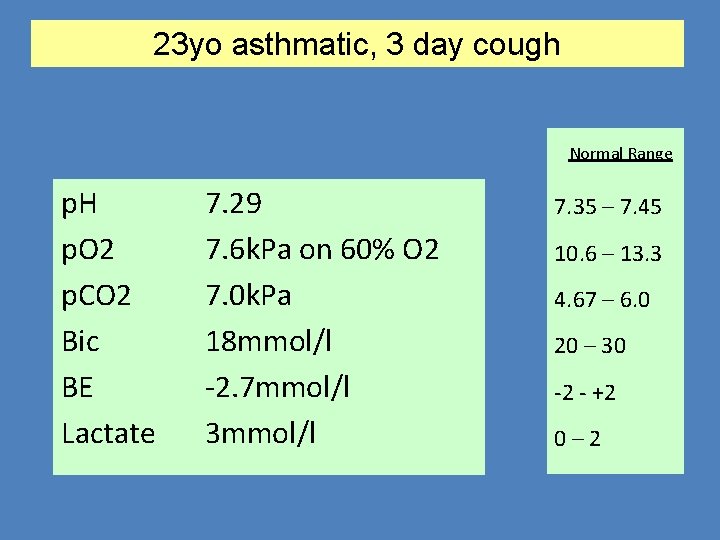
23 yo asthmatic, 3 day cough Normal Range p. H p. O 2 p. CO 2 Bic BE Lactate 7. 29 7. 6 k. Pa on 60% O 2 7. 0 k. Pa 18 mmol/l -2. 7 mmol/l 3 mmol/l 7. 35 – 7. 45 10. 6 – 13. 3 4. 67 – 6. 0 20 – 30 -2 - +2 0– 2
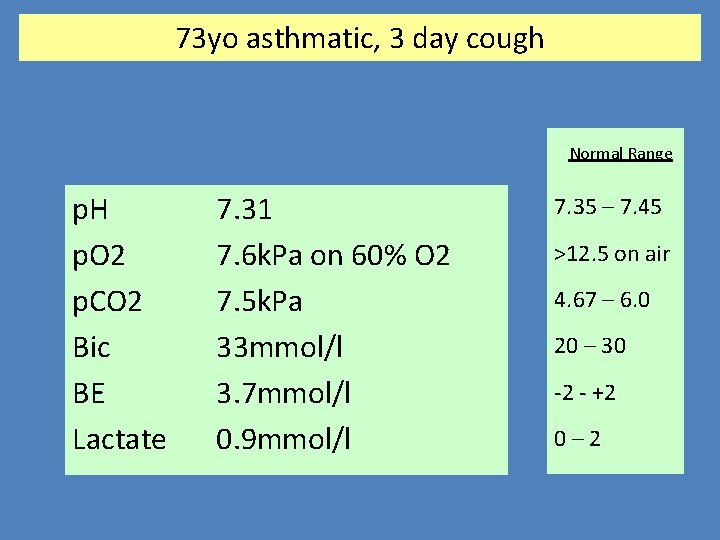
73 yo asthmatic, 3 day cough Normal Range p. H p. O 2 p. CO 2 Bic BE Lactate 7. 31 7. 6 k. Pa on 60% O 2 7. 5 k. Pa 33 mmol/l 3. 7 mmol/l 0. 9 mmol/l 7. 35 – 7. 45 >12. 5 on air 4. 67 – 6. 0 20 – 30 -2 - +2 0– 2
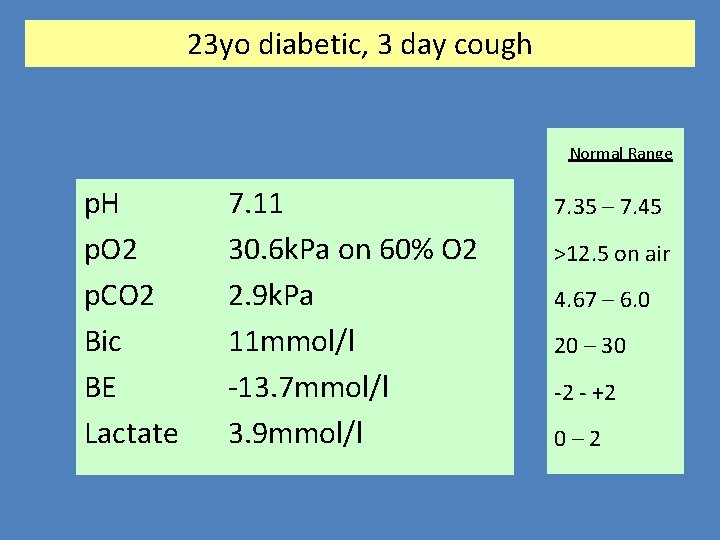
23 yo diabetic, 3 day cough Normal Range p. H p. O 2 p. CO 2 Bic BE Lactate 7. 11 30. 6 k. Pa on 60% O 2 2. 9 k. Pa 11 mmol/l -13. 7 mmol/l 3. 9 mmol/l 7. 35 – 7. 45 >12. 5 on air 4. 67 – 6. 0 20 – 30 -2 - +2 0– 2

This approach simply says: If there is more base than acid, p. H >7. 4 If there is more acid than base, p. H <7. 4 This is a simplistic method that ignores the complexities of biology and frequently fails to explain what is going on in real patients
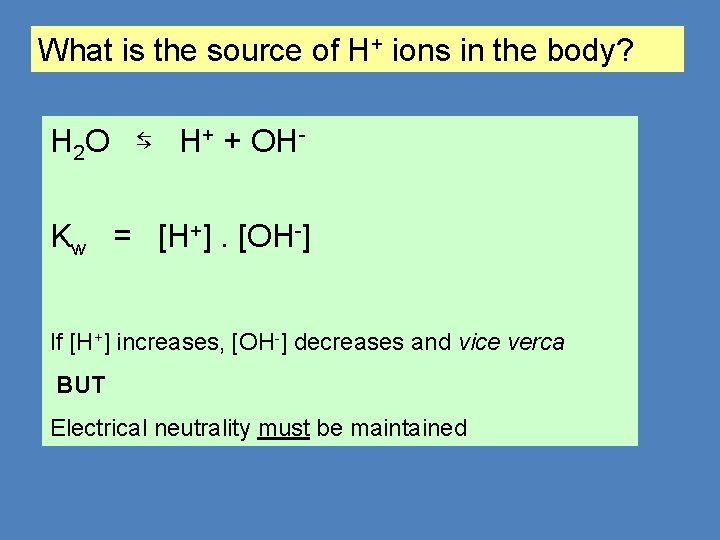
What is the source of H+ ions in the body? H 2 O ⇆ H+ + OHKw = [H+]. [OH-] If [H+] increases, [OH-] decreases and vice verca BUT Electrical neutrality must be maintained

The Law of Mass Action The velocity of a reaction is proportional to the product of the concentrations of the reactants H 2 O H+ + OHVelocity to the right: V 1 = k 1. [H 2 O] Velocity to the left: V 2 = k 2. [H+]. [OH-] At equilibrium, V 1 = V 2 k 1. [H 2 O] = k 2. [H+]. [OH-] K* = k 1/k 2 = [H+]. [OH-] / [H 2 O] BUT [H 2 O] is so large it is effectively constant K*. [H 2 O] = Kw = [H+]. [OH-] Therefore as [H+] increases [OH-] must decrease and vice verca

• ECF is a complex solution of: – Electolytes and proteins – Measured and unmeasured compounds – Buffers • ECF must obey laws of physics • Electrical neutrality • Dissociation equillibria for water, weak acids, bicarbonate and carbonate ions

• Weak acids and bicarbonate are negatively charged • To maintain electrical neutrality there must be more strong cations than strong anions • [Na]+[K]+[Ca]+[Mg] = [Cl]+[HCO 3]+[A] • [Na]+[K]+[Ca]+[Mg]-[Cl] = 44 Meq = SID
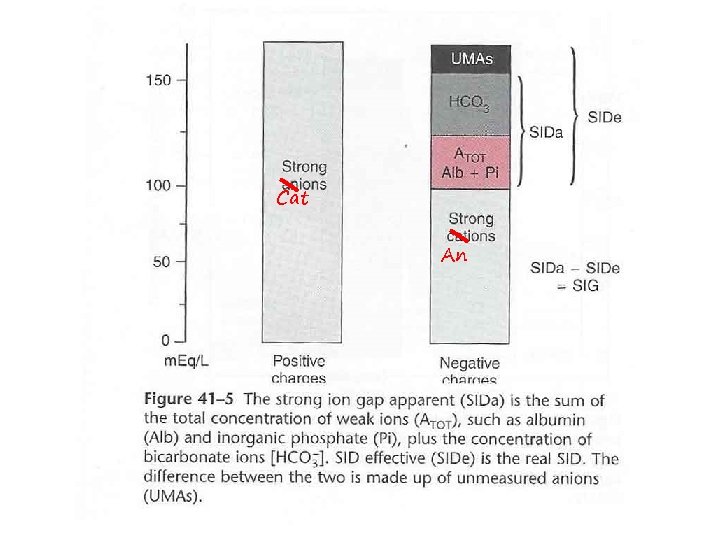
Cat An
![• If [OH-] goes up, [H+] goes down • If the difference between • If [OH-] goes up, [H+] goes down • If the difference between](http://slidetodoc.com/presentation_image_h2/524eacd17aed7cabebc28136da618d8a/image-30.jpg)
• If [OH-] goes up, [H+] goes down • If the difference between any negatively charged group and strong cations change there will be a “force” on other equillibria to restore electrical neutrality • Including water H 2 O OH- + H+
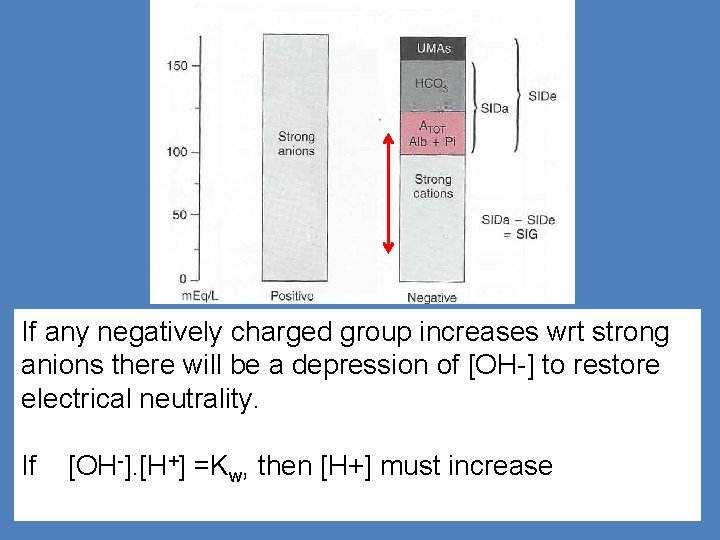
If any negatively charged group increases wrt strong anions there will be a depression of [OH-] to restore electrical neutrality. If [OH-]. [H+] =Kw, then [H+] must increase
![• [H+] is only influenced by SID, p. CO 2 and [Atot] • • [H+] is only influenced by SID, p. CO 2 and [Atot] •](http://slidetodoc.com/presentation_image_h2/524eacd17aed7cabebc28136da618d8a/image-32.jpg)
• [H+] is only influenced by SID, p. CO 2 and [Atot] • Other variables [H+], [HCO 3], [OH-] are dependant variable and cannot influence acidbase status

A patient with pneumonia has been critically ill on ITU for 10 days p. H p. CO 2 HCO 3 BE 7. 42 5. 33 24 +1. 0 Na+ Cl. Lactate Albumin 130 105 5. 2 10
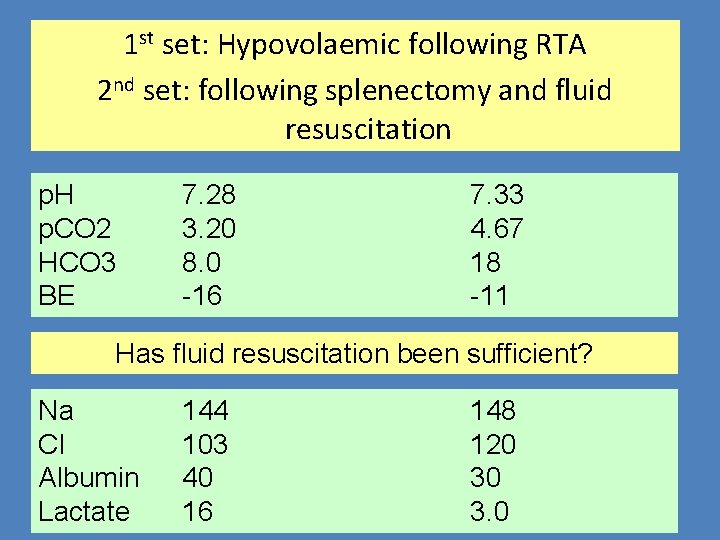
1 st set: Hypovolaemic following RTA 2 nd set: following splenectomy and fluid resuscitation p. H p. CO 2 HCO 3 BE 7. 28 3. 20 8. 0 -16 7. 33 4. 67 18 -11 Has fluid resuscitation been sufficient? Na Cl Albumin Lactate 144 103 40 16 148 120 30 3. 0

Conclusion Acid-Base regulation is horribly complicated and not fully understood by anyone Use the survival guide as a first line method. If this doesn’t work tell your consultant that you suspect an imbalance of strong ions and weak acids with reference to the Stewart-Fencl theory and you recommend referral to a Biochemist/Renal Physician/Intensivist. N. B. This will either shut them up or make them really cross. Good luck!
- Slides: 35