International Partnership for Microbicides New Science New Hope

International Partnership for Microbicides New Science, New Hope: Giving Women Power Over AIDS The John Kevany Memorial Lecture Zeda Rosenberg, Sc. D, Chief Executive Officer NUI Maynooth, 29 November 2010

The Challenge of HIV/AIDS
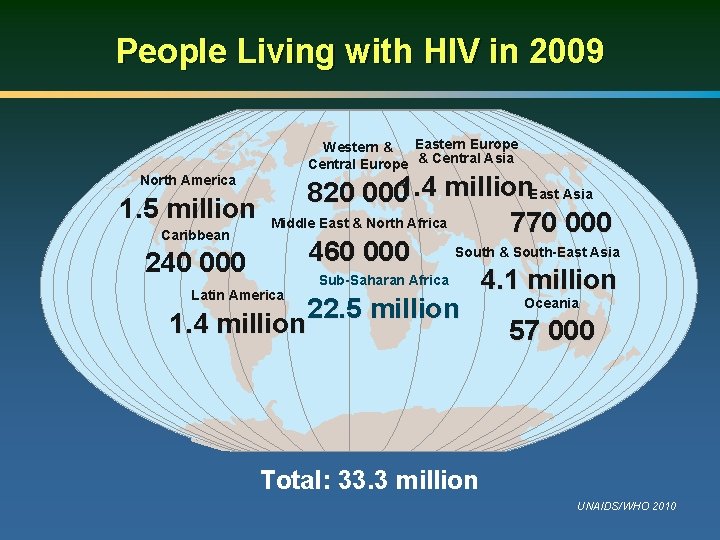
People Living with HIV in 2009 Eastern Europe Western & Central Europe & Central Asia 820 0001. 4 million. East Asia 1. 5 million Middle East & North Africa 770 000 Caribbean South & South-East Asia 460 000 240 000 Sub-Saharan Africa 4. 1 million Latin America Oceania 22. 5 million 1. 4 million 57 000 North America Total: 33. 3 million UNAIDS/WHO 2010

Women’s Vulnerability to HIV Biological, economic and socio-cultural factors: • Male-to-female transmission higher • Young women at even greater risk • Financial dependence on male partners • Inequality of women (exploitation and violence) • Cultural practices such as early marriages, intergenerational sex and concurrent partnerships

The Face of HIV/AIDS in Africa n Female Ø 60% in sub-Saharan Africa n Young Ø 3 -5 times more likely to become infected n Married and monogamous Ø Stable relationships not a haven n A mother Ø Pregnancy complicated by HIV World Bank Photo

HIV Prevention – The Global Response

From AIDS Treatment to HIV Prevention 1981 1987 1983 First AIDS case reported in the US ARV “one drug” mono -therapy approved for use HIV virus identified 2003 1997 2006 2002 Three-drug therapy: HAART. 2010 Drug combinations/ reducing pill burden 30+ approved drugs for treatment: research continues Global Fund established PEPFAR launched ARVs in gel and pill effective for HIV prevention

Current and Potential HIV/AIDS Interventions Prevention n Non-ARV: § Behavior change § Male and female condoms § STI treatment § Male circumcision § HIV vaccines Treatment and Care n ARV-based: § Prevention of mother to child transmission § Post-exposure prophylaxis (PEP) § Pre-exposure prophylaxis (Pr. EP) § Microbicides § ARV treatment § Treatment for opportunistic infections § Basic care

Promise of ARV-Based Pr. EP n Proof-of-concept for Truvada® n Appropriate for men and women n Uses approved ARV drugs n Convenient dosing: oral n Potential for different dosing strategies n Intermittent dosing n Monthly injection n Can combine more than one ARV in a single product

i. Pr. Ex: First Proof-of-Concept for Pr. EP n First efficacy trial of ARV-based pre-exposure prophylaxis (Pr. EP) for HIV prevention • Phase III: daily oral Truvada® • Two ARV drugs in one tablet: TDF and FTC • 2, 499 MSM at 11 sites in Brazil, Ecuador, Peru, South Africa, Thailand the U. S. n First Pr. EP “proof-of-concept” • 44% protection against HIV • 73% protection in volunteers who took study medication 90% or more of days • Safe, well-tolerated as tested n Pr. EP trials in other populations underway

Promise of ARV-based Microbicides n Antiretroviral (ARV)-based microbicides: Ø Target HIV specifically Ø Based on the same types of drugs successfully used to treat HIV and prevent mother to child transmission Ø Can be delivered in a variety of user-friendly forms: vaginal gel, ring, film, others Ø Potential to be developed as combination products n Proof of concept: tenofovir gel

CAPRISA 004 Trial n First efficacy trial of an ARV-based microbicide • Tenofovir gel (results July 2010) n First microbicide trial to show “proof of concept” • 889 women in South Africa • Gel used within 12 hours before and after sex • 39% protection against HIV • 51% protection against HSV-2 • Gel safe as tested & no drug resistance detected n VOICE trial ongoing; additional study(ies) planned to start in 2011

Microbicide Development

What are Microbicides? n Topical products used to prevent HIV transmission to women n Could be delivered in many forms: Vaginal gel applicator Long-acting vaginal ring Vaginal tablet, soft gel capsule, film n Ideally safe, effective, low cost, user friendly
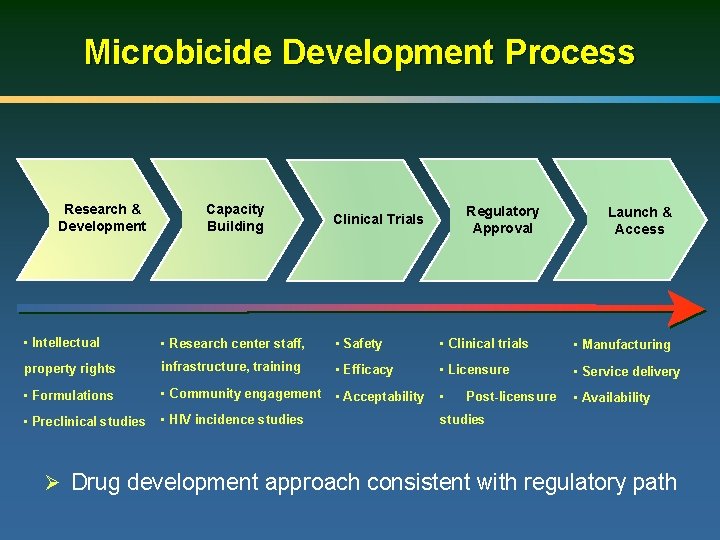
Microbicide Development Process Research & Development Capacity Building Regulatory Approval Clinical n Trials Launch & Access • Intellectual • Research center staff, • Safety • Clinical trials • Manufacturing property rights infrastructure, training • Efficacy • Licensure • Service delivery • Formulations • Community engagement • Acceptability • • Preclinical studies • HIV incidence studies Post-licensure • Availability studies Ø Drug development approach consistent with regulatory path

IPM’s Mission n Nonprofit product development partnership (PDP) n Offices in the United States, South Africa, Europe n IPM’s mission is to prevent HIV transmission by supporting the development and availability of safe and effective microbicides and other HIV prevention tools for use by women in developing countries

Partnerships With Industry Compound Dapivirine DS 001 (L-860, 167) DS 004 (L-860, 872) DS 005 (L-860, 882) DS 003 (BMS-599793) Mechanism of Action License Year Tibotec 2004 Reverse transcription: Stops virus from copying its genetic material inside human cells Merck BMS Tenofovir 2005 Cell Attachment: Prevents virus from attaching to human cells 2005 Cell Attachment 2006 Reverse transcription (IPM & CONRAD) Gilead Maraviroc Pfizer 2008 Cell Attachment DS 007 (L-000889644) Merck 2008 Cell Fusion: Prevents virus from entering human cells Non-exclusive, royalty-free licenses to develop, manufacture and distribute compounds as microbicides in developing countries
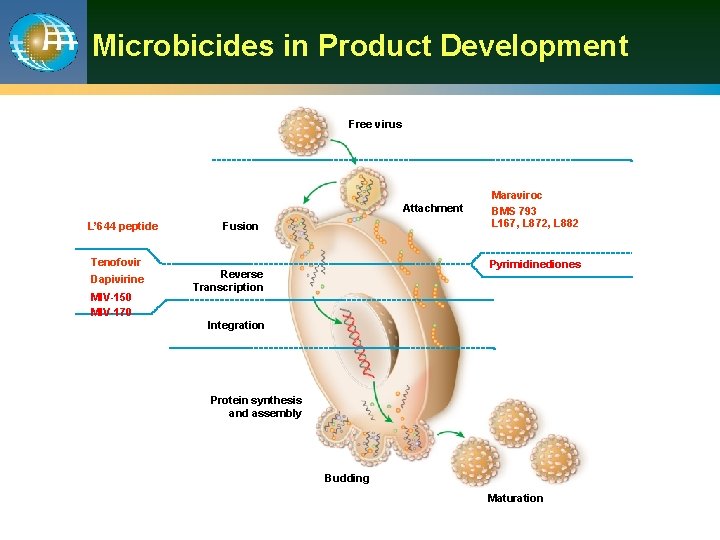
Microbicides in Product Development Free virus Attachment L’ 644 peptide Fusion Tenofovir Dapivirine MIV-150 MIV-170 Maraviroc BMS 793 L 167, L 872, L 882 Pyrimidinediones Reverse Transcription Integration Protein synthesis and assembly Budding Maturation

Need for Multiple Products & Formulations n Different women, different preferences n More product choices, more options for protection n Impact of adherence on efficacy - product won’t help if women don’t use it n Male partners’ opinions and preferences important

Vaginal Rings: An Attractive Technology n Long-acting n Easy to use • Flexible ring, can be self-inserted n Suitable for developing world • Heat-stable n Used safely for other purposes • Contraceptive and hormone therapy rings n Suitable for drug combinations n Dapivirine ring Phase III planned in 2011
IPM Partnerships n. Multilaterals n. Private sector n. Local communities n. Governments n. Civil society n IPM Pharma n. Developers, Researchers n. Research Centers n. Regulatory bodies
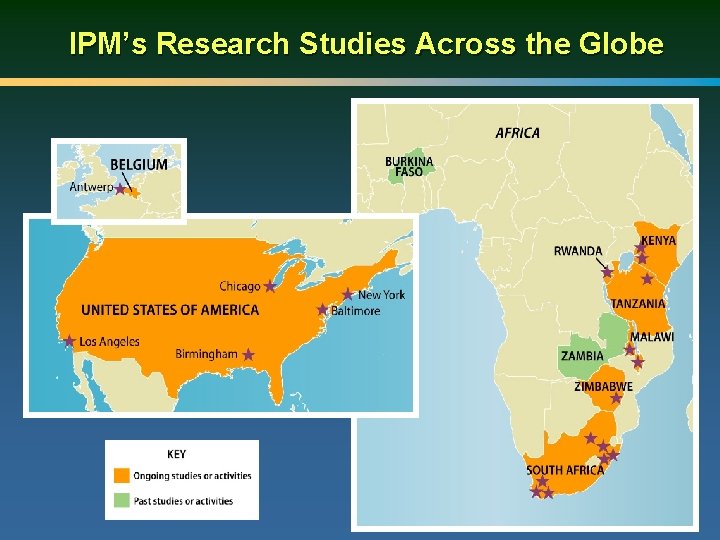
IPM’s Research Studies Across the Globe

Clinical Research Centers in Africa n. Be Part Yoluntu Centre More than 10 local partners in 6 countries conduct research studies on behalf of IPM

Capacity Building at Research Centers n Community engagement n Referral networks for medical care, treatment, support n Infrastructure and equipment n Staff training and development n Communications, messaging, tools n Financial management support n HIV incidence studies

Projet Ubuzima, Kigali, Rwanda HIV incidence studies and microbicide gel trial completed Microbicide gel trial ongoing Microbicide ring trial planned

Ladysmith, Kwa. Zulu-Natal, South Africa Site selection & renovation Staff selection & training HIV incidence study completed Microbicide ring & gel trials ongoing

Benefiting People, Communities, Countries n Promote reproductive health and HIV awareness n Empower women through education and counselling n Involve men in HIV prevention n Encourage HIV testing n Strengthen delivery of and access to health services n Engage communities, build community advisory (CAB) n Provide employment and professional development n Building medical research capabilities in geographical areas of need

Access

Access Principles: Planning for Success n Availability n Accessibility n Acceptability n Affordability

Acceptability Studies in Africa Vaginal ring, film, tablet, soft gel capsule n All formulations acceptable n Different formulations preferred in different countries n Women very likely to use all products to prevent HIV n Interest in discreet use

Access: Preparing for Success n IPM Access Strategy n Intellectual property agreements evolution • Pfizer, Merck 2008, Tibotec n Acceptability/market research studies • IPM 011, PAS 2 n Global manufacturing survey completed n LSHTM modeling of microbicide introduction n Pharma lessons learned ARV treatment intro n Annual Microbicide Access Forum

Key Regulatory Aspects of Access n Demonstration of efficacy in Phase III trial does not automatically translate into product licensure or immediate access to product n Regulatory licensure requires review for: • Product quality (CMC) • Safety (preclinical and clinical) • Efficacy n However, very limited access to product can be achieved through clinical programs

Empowering Women

Women’s Right to Health n Human right to health in many UN rights treaties n Women’s right to health in • ICPD Programme of Action (1994) • The Beijing Platform for Action (1995) n Women’s productivity and humanity undermined without high standard of health n HIV protection responds to woman’s risks of infection Microbicides could provide a vital HIV prevention tool for women and enable them to protect their health and that of their families

Microbicides and the Millennium Development Goals Microbicides have the potential to impact 7 of 8 MDGs Source: United Nations

Concluding Remarks

Ireland – Pioneering Support IPM thanks the people of Ireland the Irish Government and for your unwavering commitment to microbicide development and the needs of women in the developing world.

Women Urgently Need Microbicides “A microbicide could mean the difference between life and death for millions of women. Let us do everything in our power to accelerate its development. ” Mrs. Graça Machel

THANK YOU!
- Slides: 39