Intermolecular Forces v polar v This symbol means

Intermolecular Forces

v polar v Ø This symbol: means “partial. ” Ø This type of arrow: → | is a “dipole. ” + H Cl
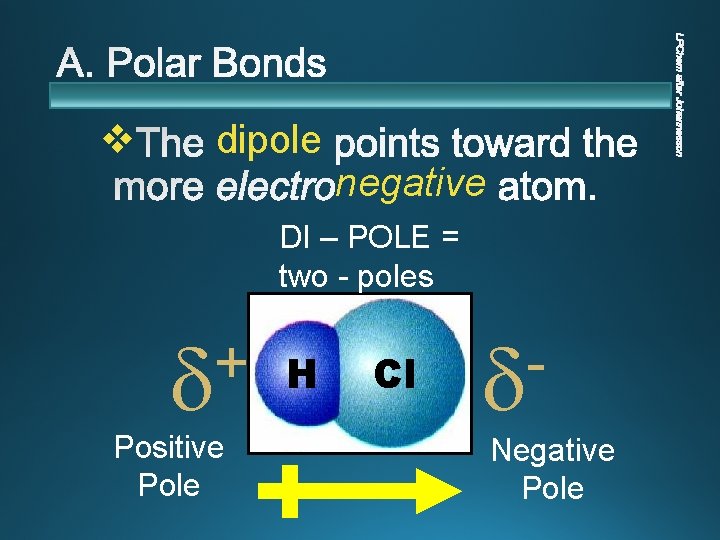
v dipole negative DI – POLE = two - poles + Positive Pole H Cl Negative Pole

v Ø B AB 3 NO net dipole moment A B B
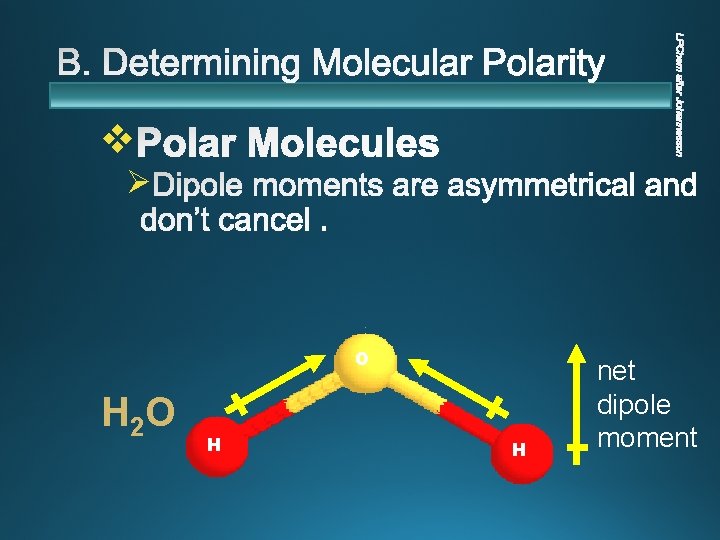
v Ø O H 2 O H H net dipole moment
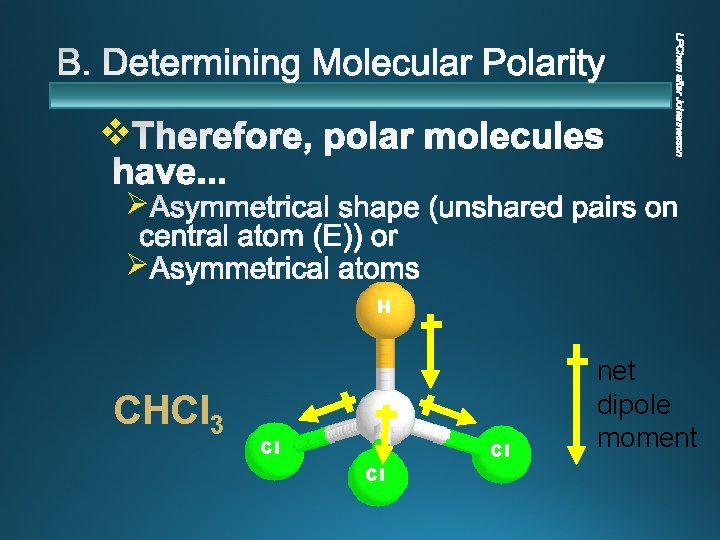
v Ø Ø H CHCl 3 Cl Cl Cl net dipole moment
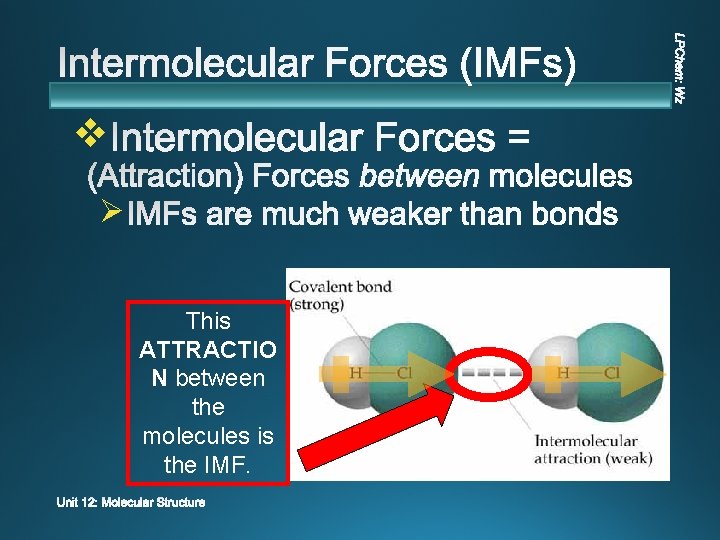
v Ø This ATTRACTIO N between the molecules is the IMF.


v v Ø attracts
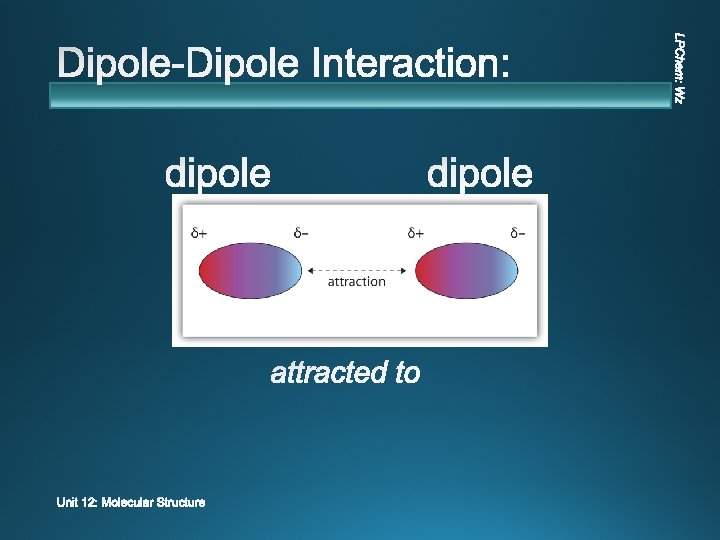

v v Charge attraction between permanent dipoles All polar molecules Medium strength It is stronger when the molecules are closer.

v temporary Ø Ø unevenly
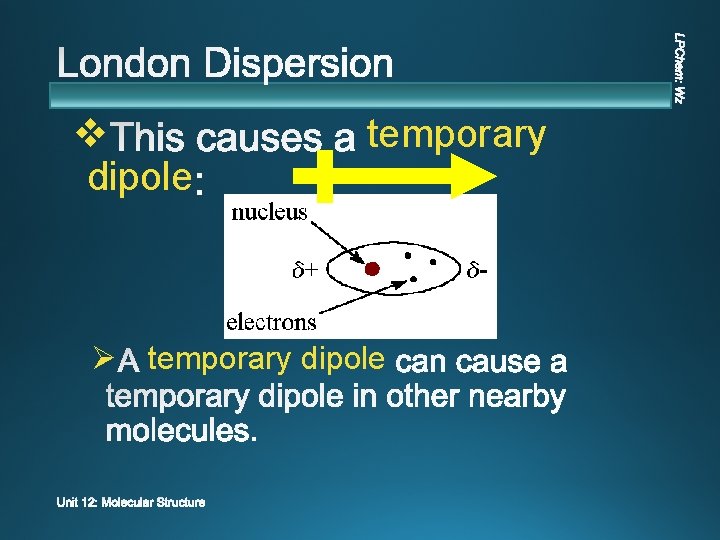
v dipole temporary Ø temporary dipole

v Ø temporary or instantaneous (only lasting for an instant) attractions.

v v Instantaneous attraction between temporary dipoles due to uneven e- dispersion All atoms & molecules v Weakest strength v Stronger for molecules with greater masses.

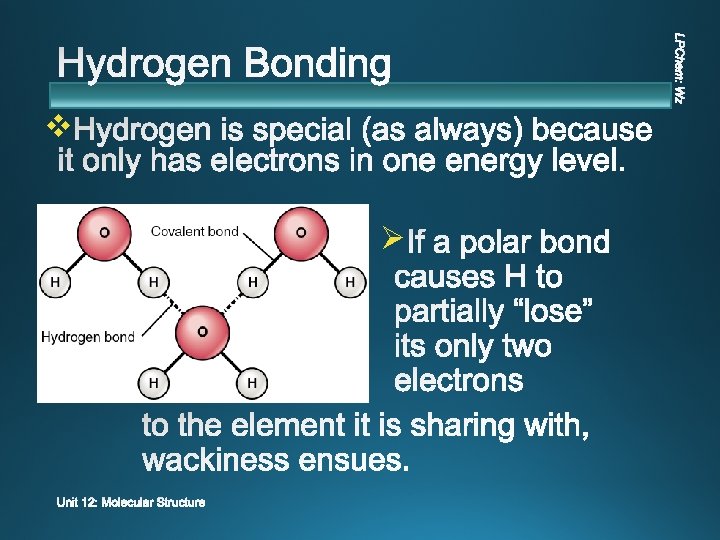
v Ø extremely polar bonds Ø force dipole-dipole
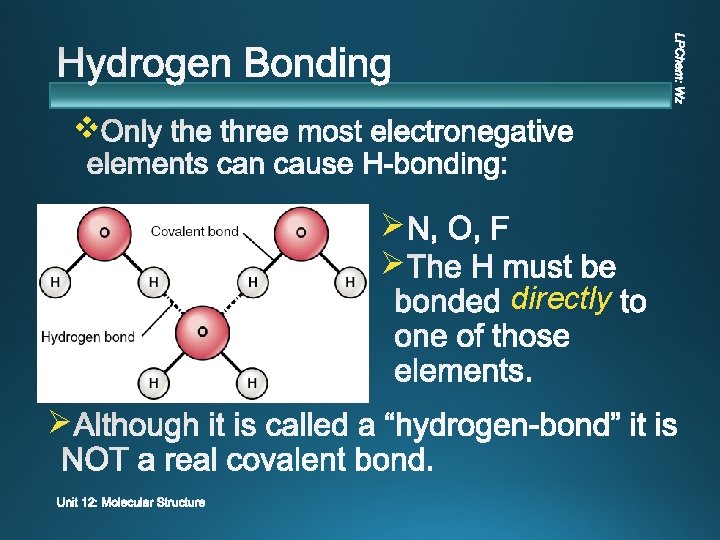
v Ø Ø Ø directly

v v v Extreme attraction between molecules with N-H, O-H, or F-H bonds. Molecules with H bound directly to N, O, F Greatest strength v Sometimes called a “pseudo bond”, but it is NOT chemical bonding.
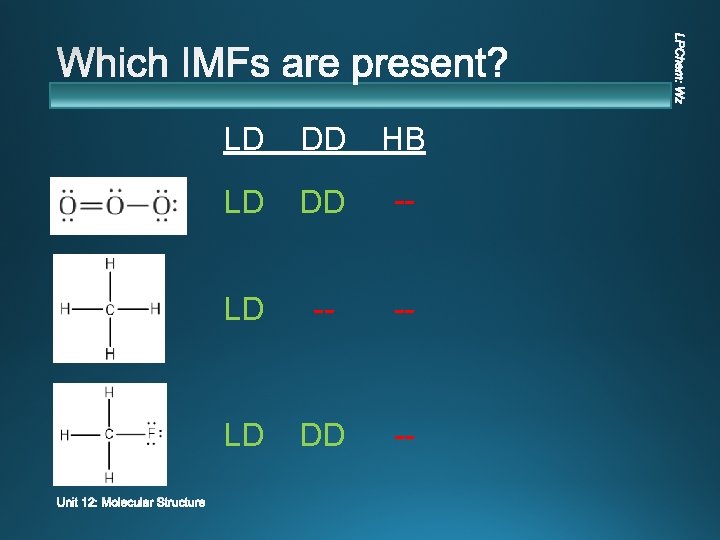
LD DD HB LD DD -- LD -- -- LD DD --
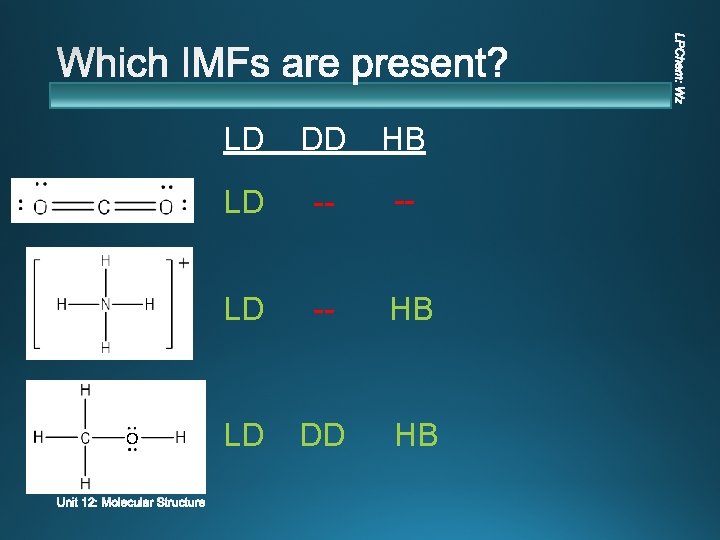
LD DD HB LD -- -- LD -- HB LD DD HB



- Slides: 24