INTERMOLECULAR FORCES OF ATTRACTION Attraction existing between molecules

INTERMOLECULAR FORCES OF ATTRACTION Attraction existing between molecules in a given substance - Responsible for the phase of the substance at a given temperature; for example, strong IMFA – solid at room temperature; weak IMFA – gas at room temperature -
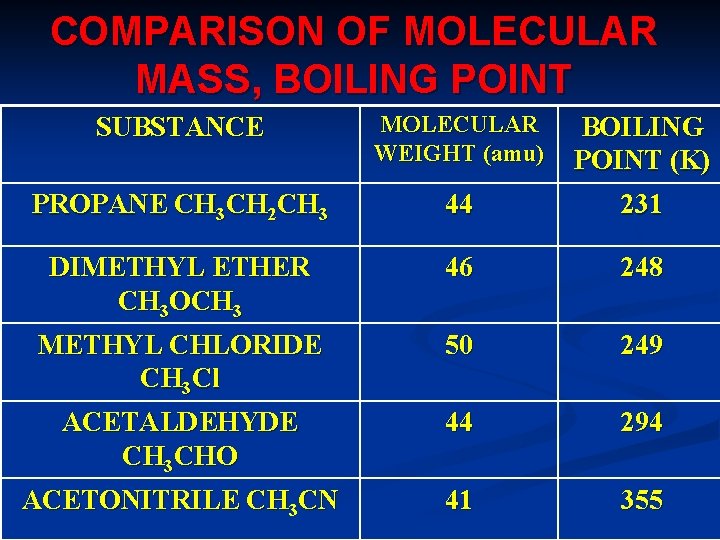
COMPARISON OF MOLECULAR MASS, BOILING POINT SUBSTANCE MOLECULAR WEIGHT (amu) BOILING POINT (K) PROPANE CH 3 CH 2 CH 3 44 231 DIMETHYL ETHER CH 3 OCH 3 46 248 METHYL CHLORIDE CH 3 Cl 50 249 ACETALDEHYDE CH 3 CHO 44 294 ACETONITRILE CH 3 CN 41 355

Relative Magnitudes of Forces The types of bonding forces vary in their strength as measured by average bond energy. Strongest Covalent bonds (400 kcal) Hydrogen bonding (12 -16 kcal ) Weakest Dipole-dipole interactions (2 -0. 5 kcal) London forces (less than 1 kcal)

TYPES OF IMFA (weakest to strongest) 1. VAN DER WAAL’S FORCES a. London dispersion force – caused by the movement of the electrons around the nucleus - effect is very temporary - present in all substances and only force present in non-polar
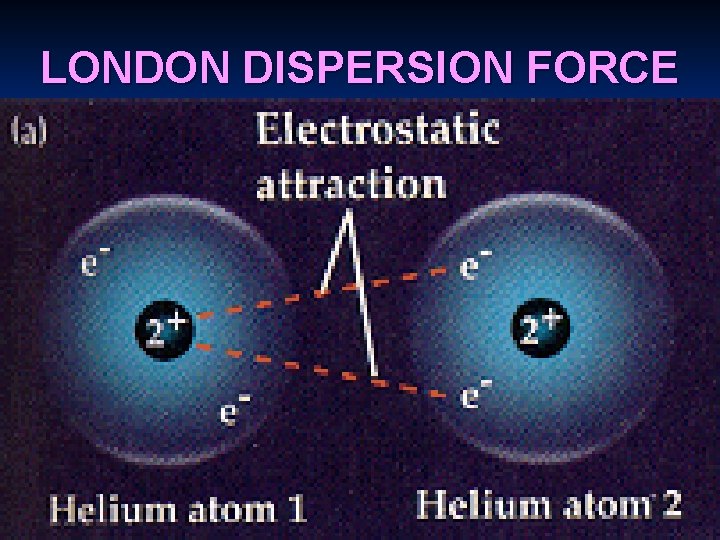
LONDON DISPERSION FORCE

Induced dipole – temporary charges due to presence of electrons from neighboring atoms
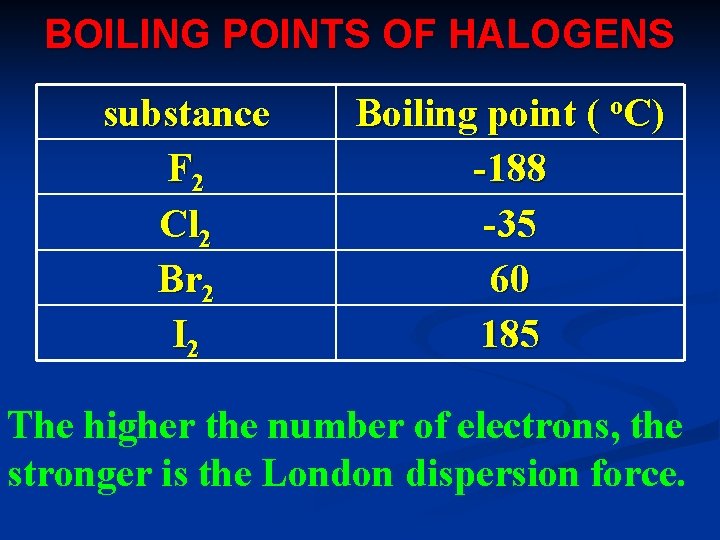
BOILING POINTS OF HALOGENS substance F 2 Cl 2 Br 2 I 2 Boiling point ( o. C) -188 -35 60 185 The higher the number of electrons, the stronger is the London dispersion force.
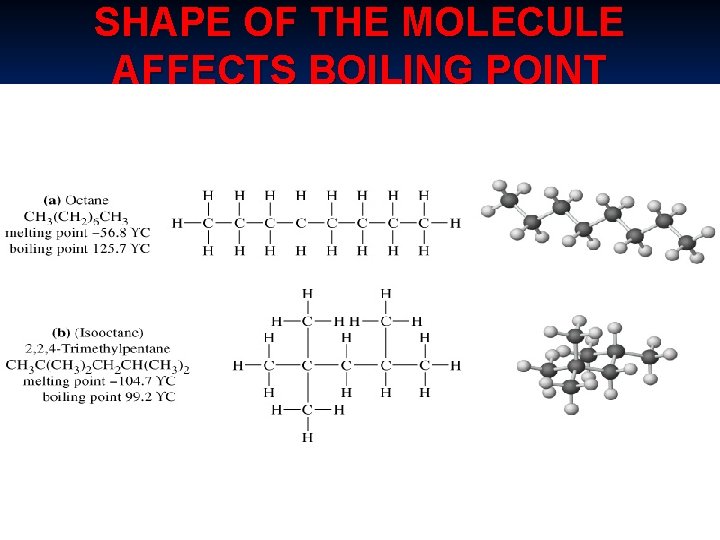
SHAPE OF THE MOLECULE AFFECTS BOILING POINT

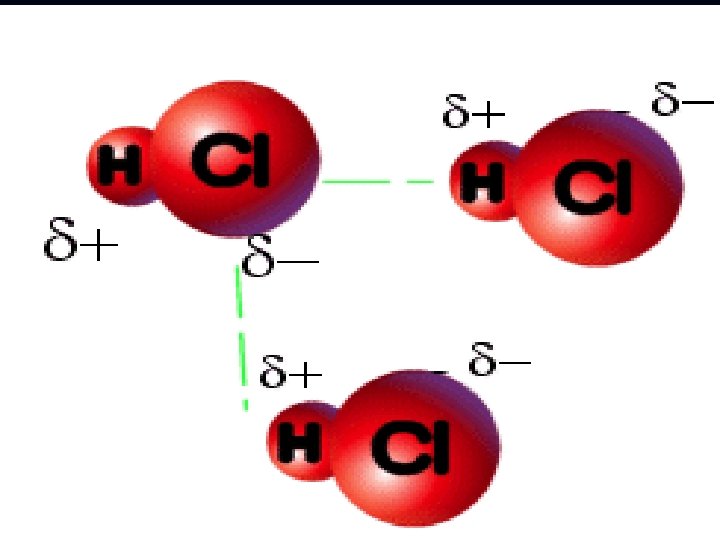
b. dipole-dipole interaction – present only in polar covalent molecules - caused by the dipole moment (differences in the electronegativy of the elements present)

Polarity A molecule, such as HF, that has a center of positive charge and a center of negative charge is said to be polar, or to have a dipole moment. H F + -
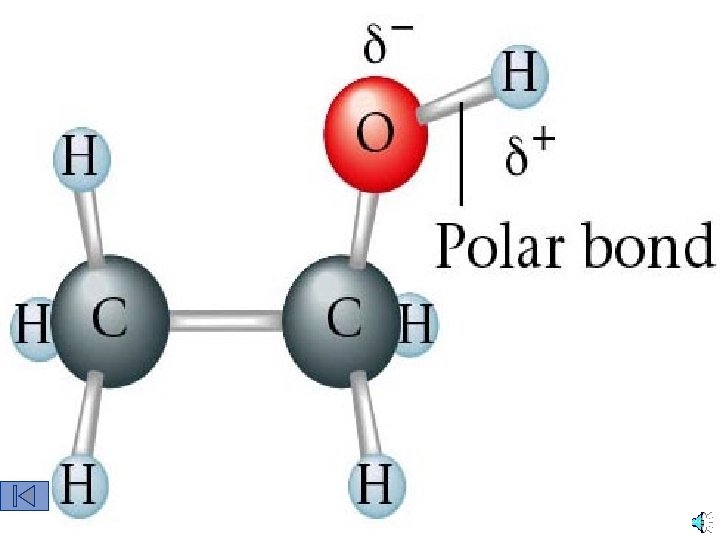
Ethanol is Polar

c. Hydrogen bonding - present in polar covalent compounds where hydrogen is directly attached to Fluorine, Oxygen and Nitrogen ( the three most electronegative elements) FON
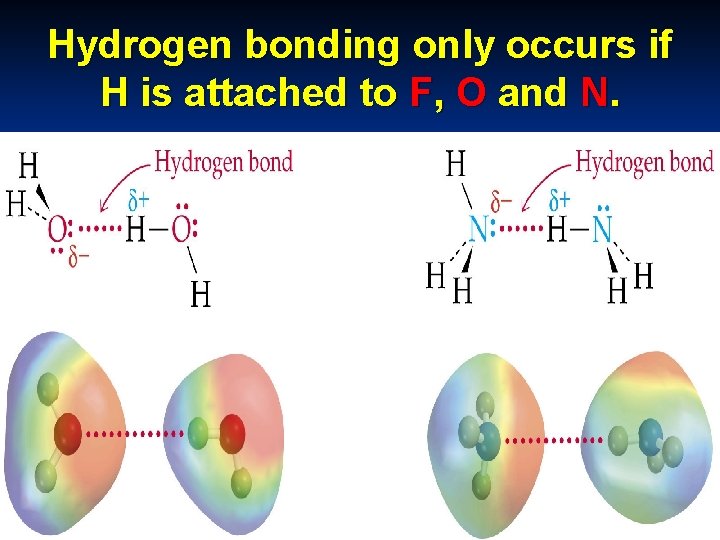
Hydrogen bonding only occurs if H is attached to F, O and N.
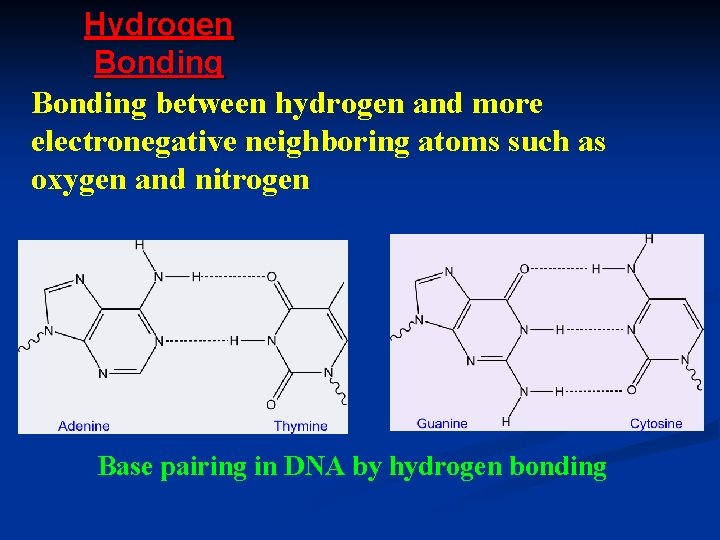
Hydrogen Bonding between hydrogen and more electronegative neighboring atoms such as oxygen and nitrogen Base pairing in DNA by hydrogen bonding
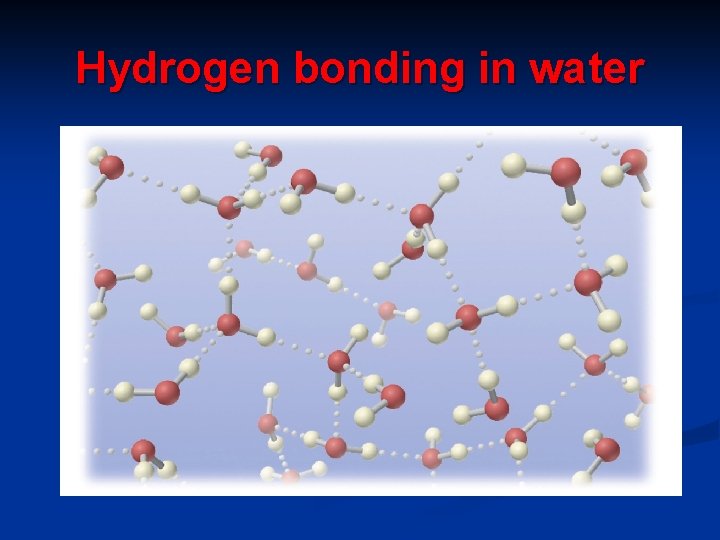
Hydrogen bonding in water
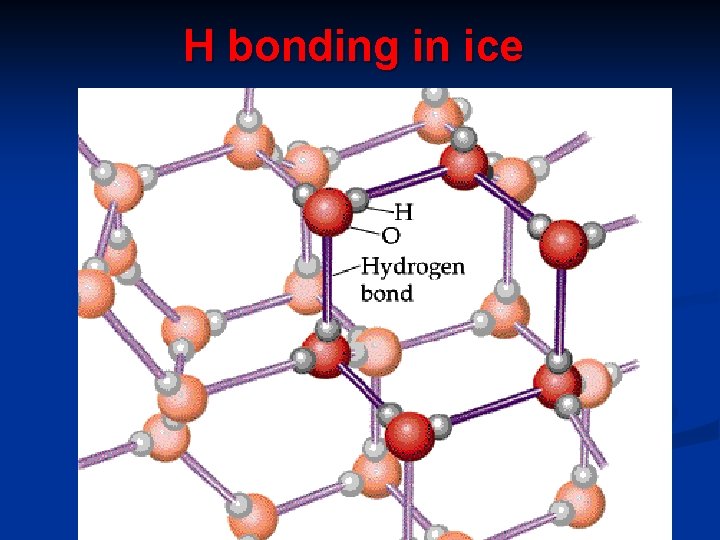
H bonding in ice

2. IONIC BONDING – - present only in ionic compounds. - Electrostatic force of attraction.
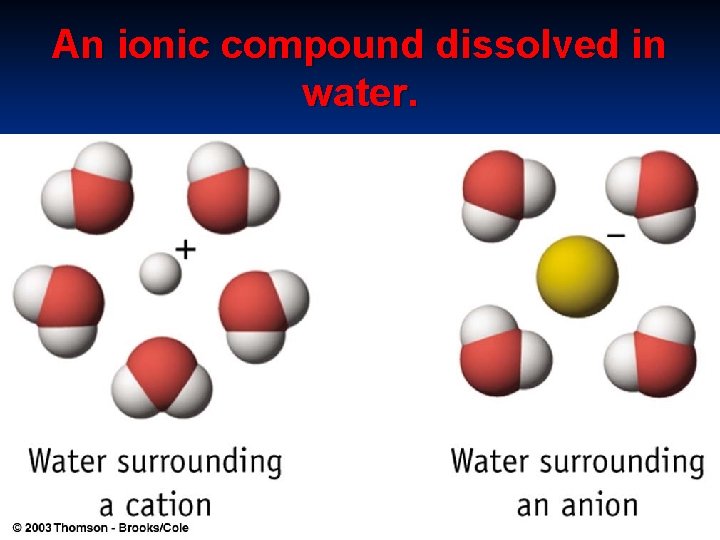
An ionic compound dissolved in water.

FORMULA TO DETERMINE THE NUMBER OF RINGS AND/OR DOUBLE AND TRIPLE BONDS ρ= 2 + 2 C + N – H – X 2 where C = number of carbon atoms N = number of nitrogen atoms H = number of hydrogen atoms X = number of halogen (Group 17) atoms
- Slides: 20