INTERMOLECULAR FORCES INVESTIGATING THE FORCES OF ATTRACTION BETWEEN

INTERMOLECULAR FORCES INVESTIGATING THE FORCES OF ATTRACTION BETWEEN MOLECULES, AND HOW THEY COMPARE TO IONS AND METALS

VOCAB • ______: the forces of attraction between molecules • ____: equal but opposite charges separated by a short distance • ______: the intermolecular force in which a hydrogen atom that is bonded to a highly electronegative atom is attracted to an unshared pair of electrons of an electronegative atom in a nearby molecule • _______: The intermolecular attraction resulting from the constant motion of electrons and the creation of instantaneous dipoles

BOILING • To understand the relative strength of intermolecular forces we must first understand how a ______ changes to a _____ • If you increase the _____ of a liquid it will reach a point where it begins to boil • The temperature of a substance is really just a measure of how ____ particles are moving

BOILING CONTINUED • In a liquid the particles are moving slow enough that their attractions cause them to _______ together. But they are still able to ______ past each other. • In a gas the particles are moving so fast that the attractions between ____ is no longer able to cause them to stick together • This means boiling is the point where the particles are moving so fast they can overcome the _____ holding them close together in liquids • Watch it in action

RELATIVE STRENGTH • The _____ is a good measure of the force of attraction between particles. • IMF vary in strength, but are generally ______ than the bonds joining atoms in molecule, ions in compounds, or metal atoms in solid metals • Section 6. 5: Intermolecular Forces

INTERMOLECULAR FORCES • In this section we will focus only on the attractive forces between separate _____ • There a variety of intermolecular forces, and each of them varies in _____

DIPOLE-DIPOLE FORCES • The ____ IMF exist between polar molecules • Polar molecules act as _______ because of their uneven charge distribution • The direction of a dipole is from the _____ pole to the _____ pole. Used a crossed arrow to indicate a dipole.

DIPOLES CONTINUED • The negative region in one polar molecule attracts the _______ region in an adjacent polar molecule • The forces of attraction between polar molecules are known as ______-______ forces • Effect can be seen by comparing boiling points of polar and _____ molecules

MOLECULAR POLARITY • The polarity of molecules with more than one bond depends on not just polarity of individual bonds, but also on _____ of each bond • This means we must always consider _____ when determining molecular polarity
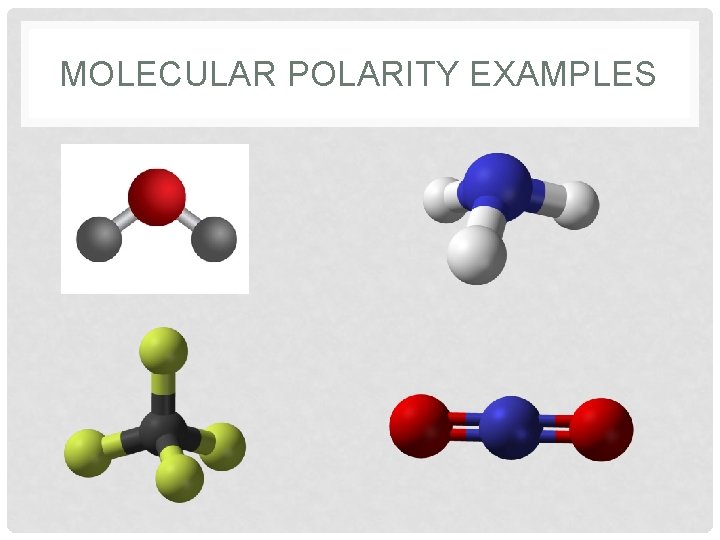
MOLECULAR POLARITY EXAMPLES

HYDROGEN BONDING • Some hydrogen containing compounds have unusually ____ boiling points (HF, H 2 O, NH 3) • This is caused by a strong type of dipole-dipole force called _________ • Only compounds made from ____, _____, and _____ with H can make hydrogen bonds
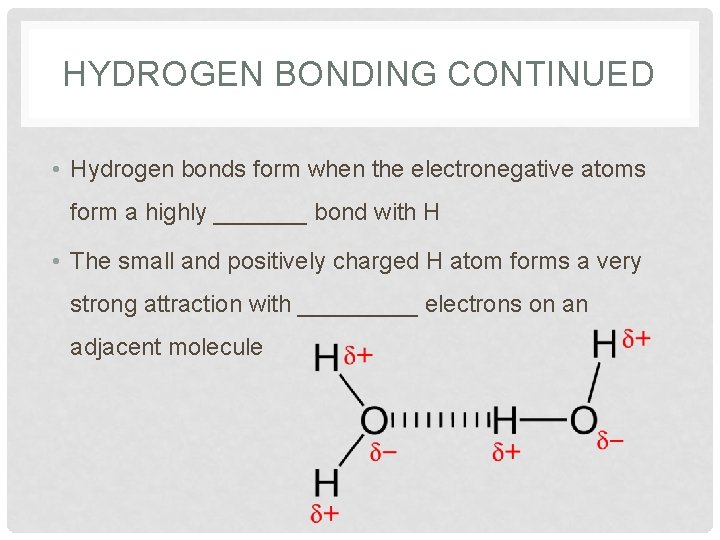
HYDROGEN BONDING CONTINUED • Hydrogen bonds form when the electronegative atoms form a highly _______ bond with H • The small and positively charged H atom forms a very strong attraction with _____ electrons on an adjacent molecule

STRENGTH COMPARISON OF HYDROGEN BONDING • Boiling point of phosphine (PH 3)_______ • Boiling point of ammonia (NH 3)____ • BP H 2 S ____ • BP H 2 O ____
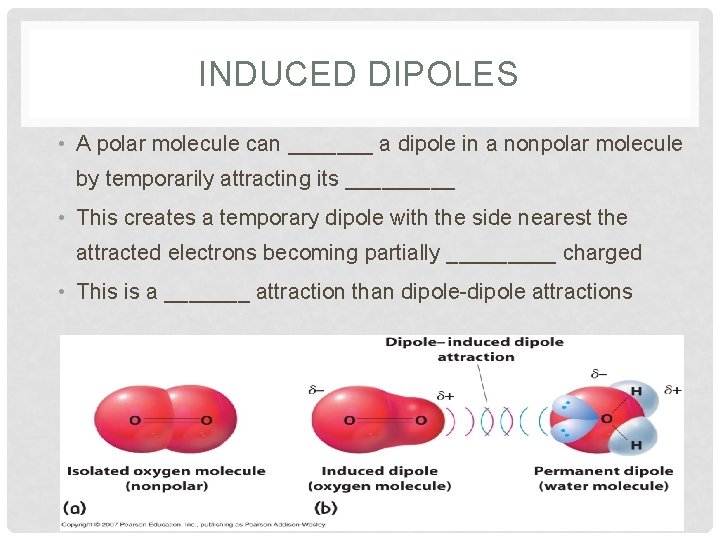
INDUCED DIPOLES • A polar molecule can _______ a dipole in a nonpolar molecule by temporarily attracting its _____ • This creates a temporary dipole with the side nearest the attracted electrons becoming partially _____ charged • This is a _______ attraction than dipole-dipole attractions
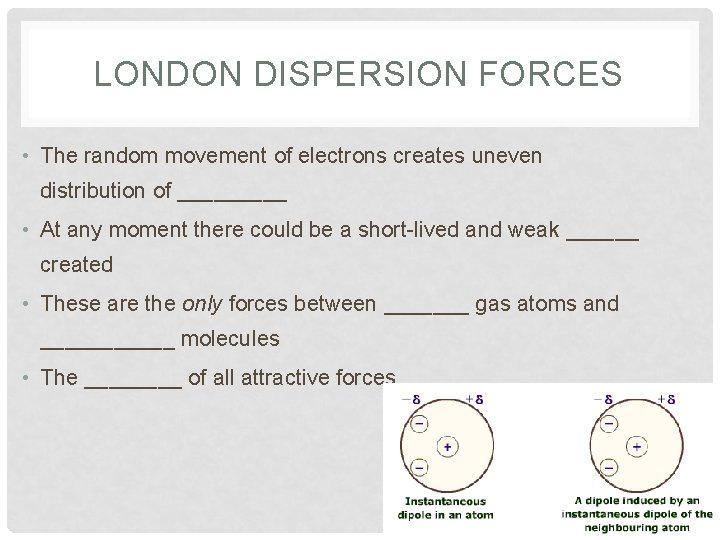
LONDON DISPERSION FORCES • The random movement of electrons creates uneven distribution of _____ • At any moment there could be a short-lived and weak ______ created • These are the only forces between _______ gas atoms and ______ molecules • The ____ of all attractive forces
- Slides: 15