Intermolecular Forces Intramolecular forces chemical bonds H 2
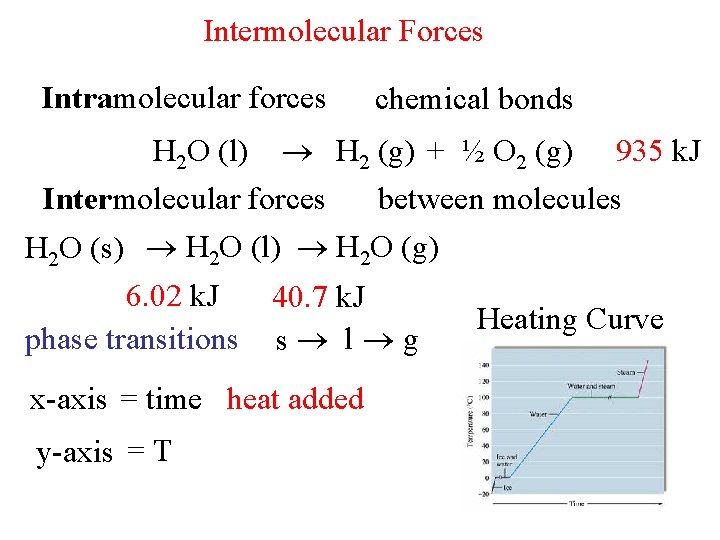
Intermolecular Forces Intramolecular forces chemical bonds H 2 O (l) H 2 (g) + ½ O 2 (g) 935 k. J Intermolecular forces between molecules H 2 O (s) H 2 O (l) H 2 O (g) 6. 02 k. J phase transitions 40. 7 k. J s l g x-axis = time heat added y-axis = T Heating Curve
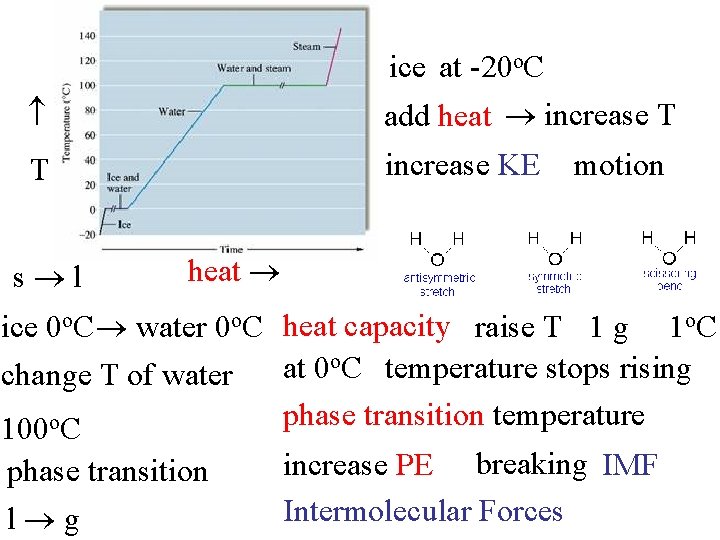
ice at -20 o. C add heat increase T T increase KE s l motion heat ice 0 o. C water 0 o. C heat capacity raise T 1 g 1 o. C temperature stops rising at 0 change T of water phase transition temperature 100 o. C phase transition l g increase PE breaking IMF Intermolecular Forces
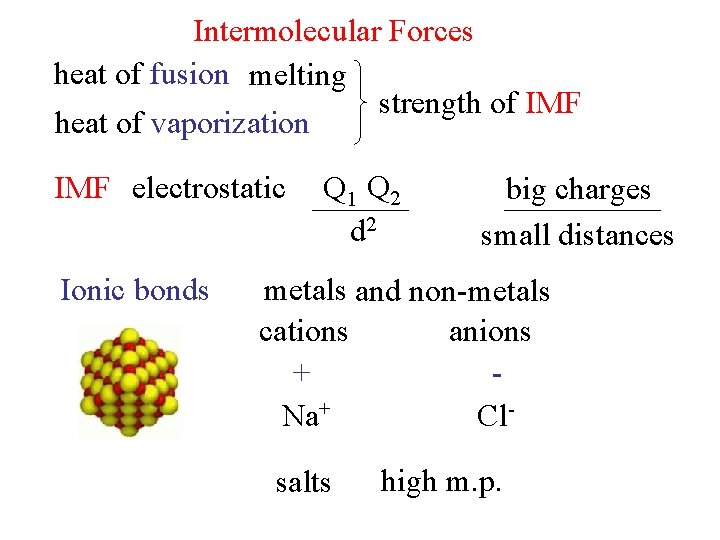
Intermolecular Forces heat of fusion melting strength of IMF heat of vaporization IMF electrostatic Ionic bonds Q 1 Q 2 d 2 big charges small distances metals and non-metals cations anions + Na+ Clsalts high m. p.
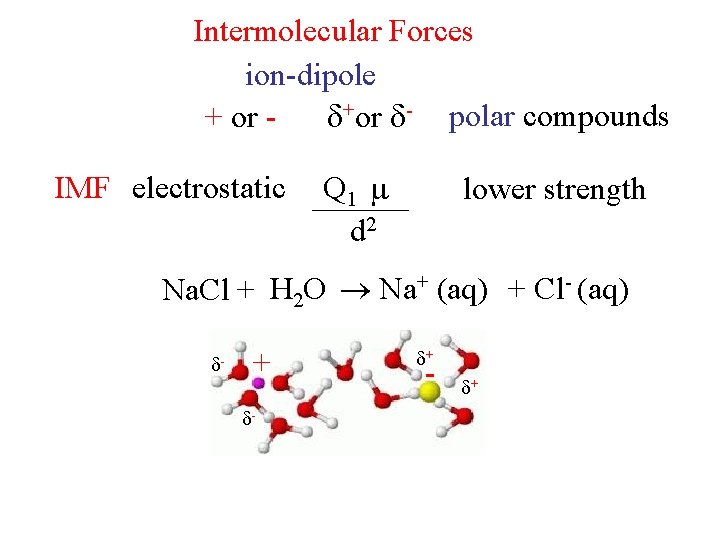
Intermolecular Forces ion-dipole + or +or - polar compounds IMF electrostatic Q 1 Q 2 d 2 lower strength Na. Cl + H 2 O Na+ (aq) + Cl- (aq) - + - +
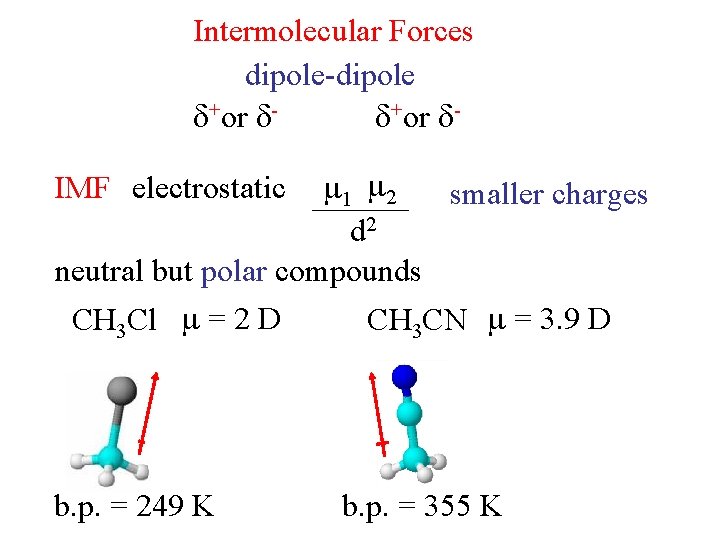
Intermolecular Forces dipole-dipole +or 1 2 smaller charges d 2 neutral but polar compounds CH 3 Cl = 2 D CH 3 CN = 3. 9 D IMF electrostatic b. p. = 249 K b. p. = 355 K
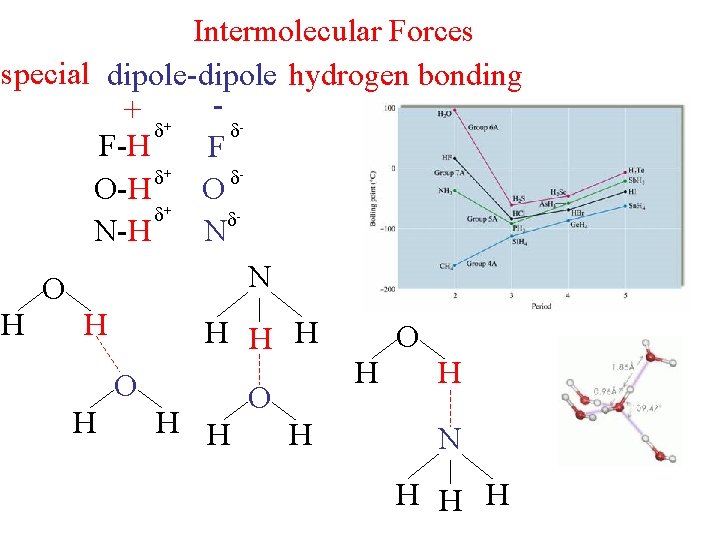
Intermolecular Forces special dipole-dipole hydrogen bonding + + - F-H F + O-H O + N-H N N O H H H O H H N H H H

Intermolecular Forces non-polar compounds London Dispersion Forces “instantaneous” dipole moments He He Ne Ar Kr Xe Rn e 2 10 18 36 54 86 b. p. -269 -246 -186 -152 -107 -62 + - small short lived short distance strength increases polarizibility e- further from nucleus size
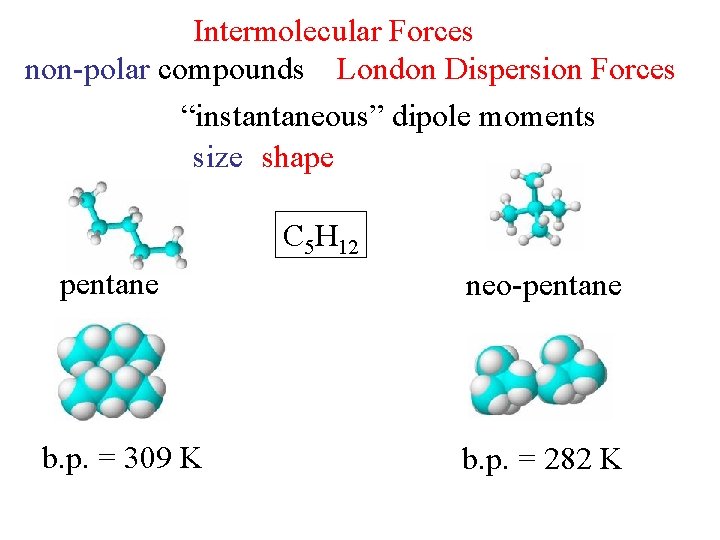
Intermolecular Forces non-polar compounds London Dispersion Forces “instantaneous” dipole moments size shape C 5 H 12 pentane b. p. = 309 K neo-pentane b. p. = 282 K
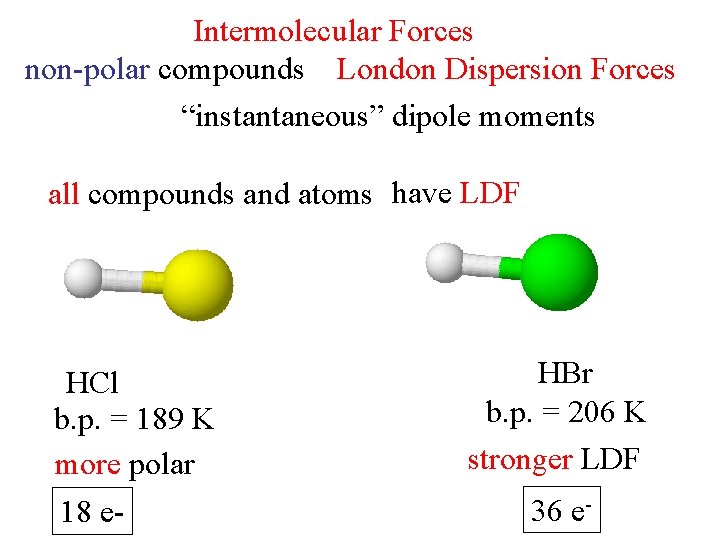
Intermolecular Forces non-polar compounds London Dispersion Forces “instantaneous” dipole moments all compounds and atoms have LDF HCl b. p. = 189 K more polar 18 e- HBr b. p. = 206 K stronger LDF 36 e-
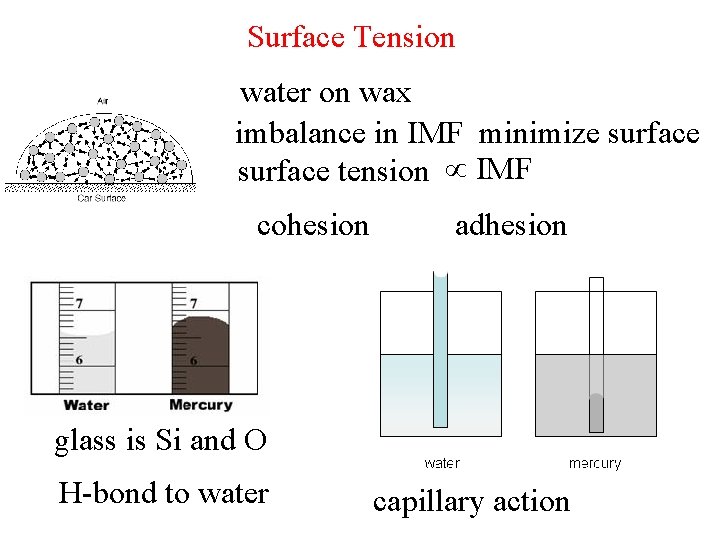
Surface Tension water on wax imbalance in IMF minimize surface tension IMF cohesion adhesion glass is Si and O H-bond to water capillary action
- Slides: 10