Intermolecular Forces Intermolecular Forces This force is responsible

Intermolecular Forces

Intermolecular Forces • This force is responsible for many physical properties we observe in molecules, including the temperature at which they melt and boil.
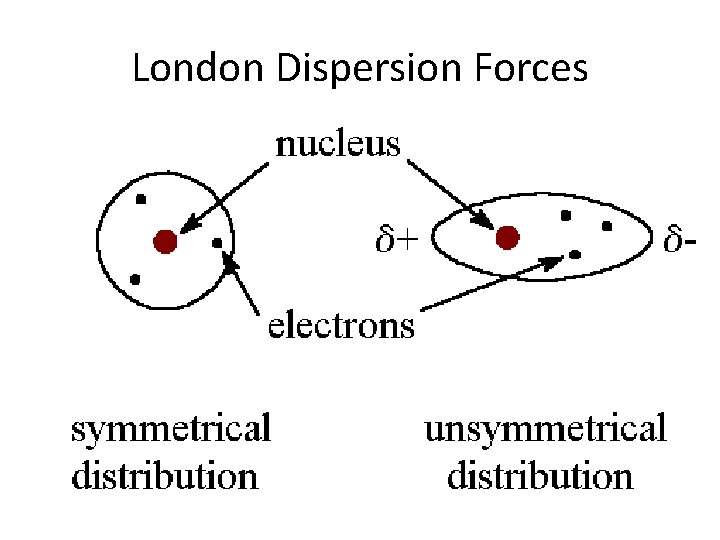
London Dispersion Forces

LDF • London dispersion force is the weakest intermolecular force • Temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles – This force is sometimes called an induced dipole attraction. • London forces are the attractive forces that cause nonpolar substances to condense to liquids and to freeze into solids when the temperature is lowered sufficiently.
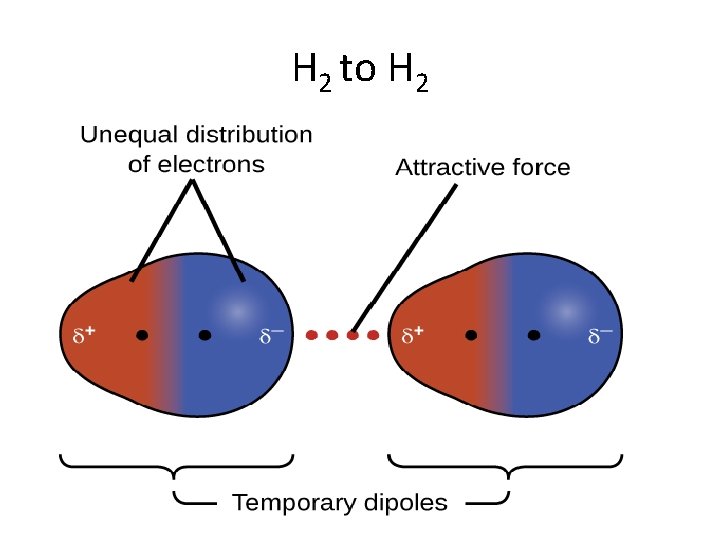
H 2 to H 2

Which molecule exhibits the strongest London Dispersion Force? • Propane C 3 H 8 • Hexane C 6 H 14 • Paraffin C 18 H 38

London (Dispersion) Forces • The weakest of intermolecular forces, these forces are proportional to the mass of the molecule • These are the only forces of attraction between completely nonpolar molecules • Large nonpolar molecules may have substantial dispersion forces, resulting in relatively high boiling points • Small nonpolar molecules have weak dispersion forces and exist almost exclusively as gases
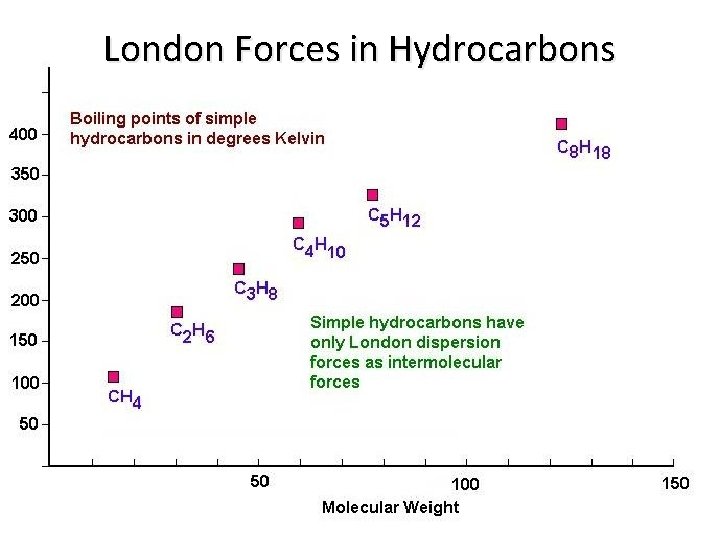
London Forces in Hydrocarbons

Polarity A molecule, such as HF, that has a center of positive charge and a center of negative charge is said to be polar, or to have a dipole moment. H + F -
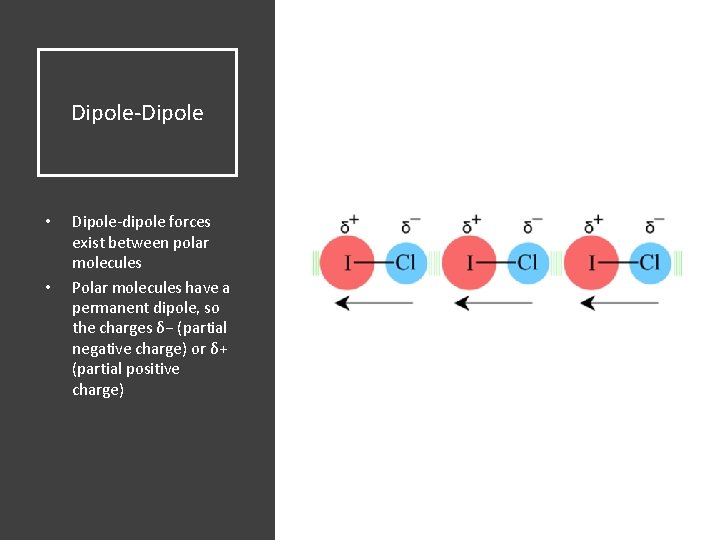
Dipole-Dipole • • Dipole-dipole forces exist between polar molecules Polar molecules have a permanent dipole, so the charges δ− (partial negative charge) or δ+ (partial positive charge)
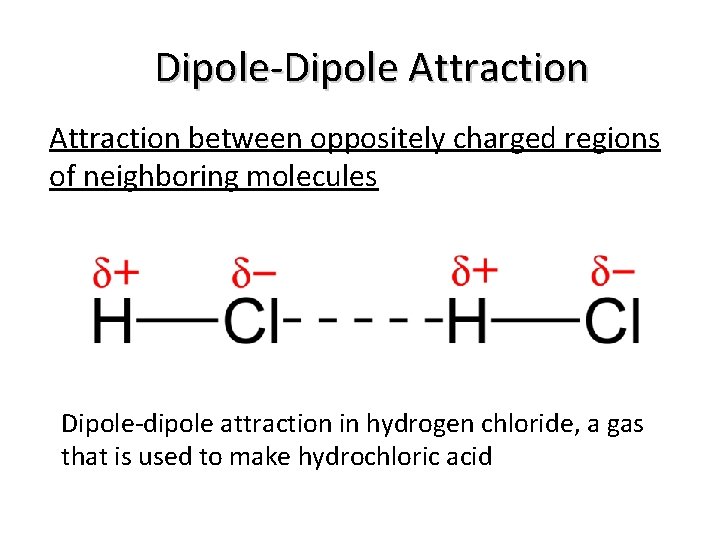
Dipole-Dipole Attraction between oppositely charged regions of neighboring molecules Dipole-dipole attraction in hydrogen chloride, a gas that is used to make hydrochloric acid
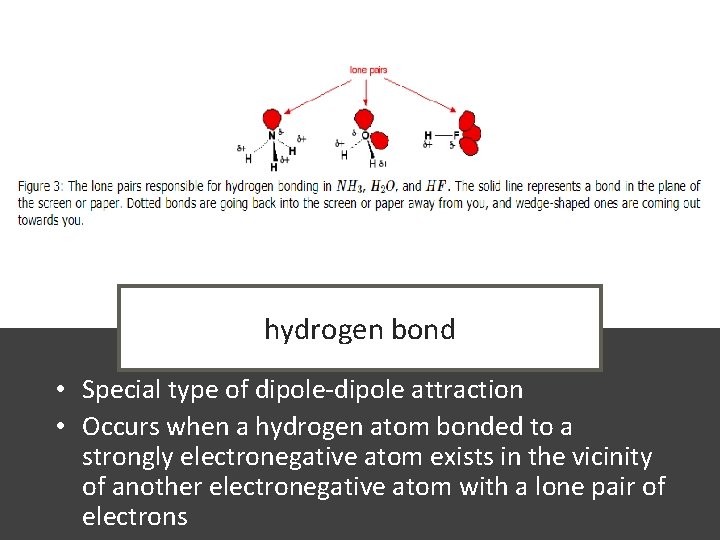
hydrogen bond • Special type of dipole-dipole attraction • Occurs when a hydrogen atom bonded to a strongly electronegative atom exists in the vicinity of another electronegative atom with a lone pair of electrons
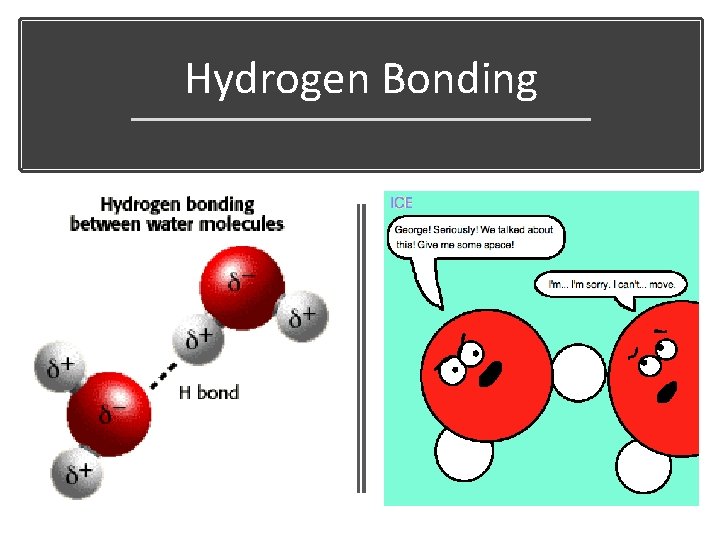
Hydrogen Bonding
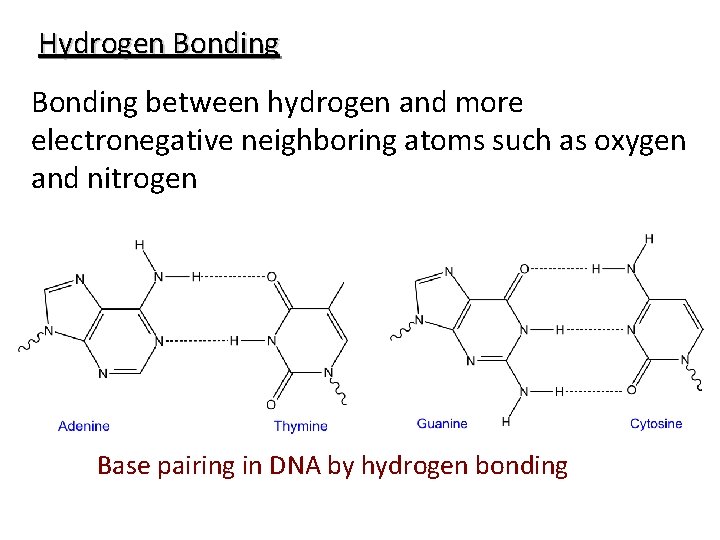
Hydrogen Bonding between hydrogen and more electronegative neighboring atoms such as oxygen and nitrogen Base pairing in DNA by hydrogen bonding
So What Do You Think is Happenning When We Boil Water?
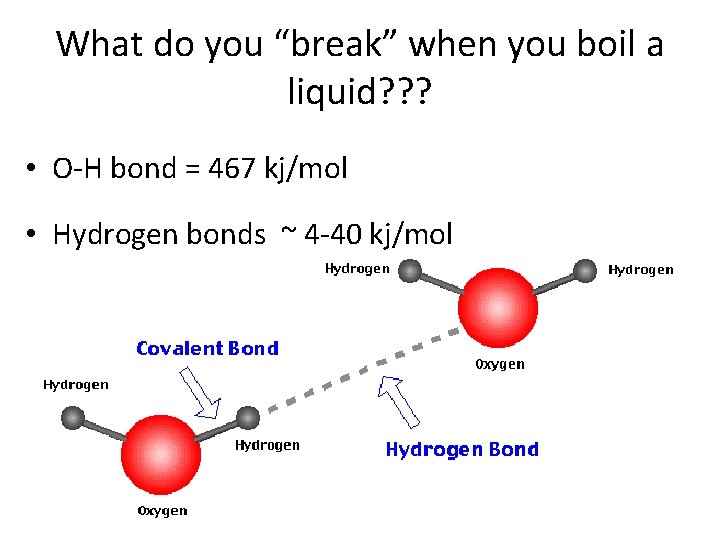
What do you “break” when you boil a liquid? ? ? • O-H bond = 467 kj/mol • Hydrogen bonds ~ 4 -40 kj/mol

Intermolecular Forces • London Dispersion Forces • Dipole-Dipole interactions – Hydrogen Bonding

Lab Reflection What parts of the lab involved hydrogen bonding?
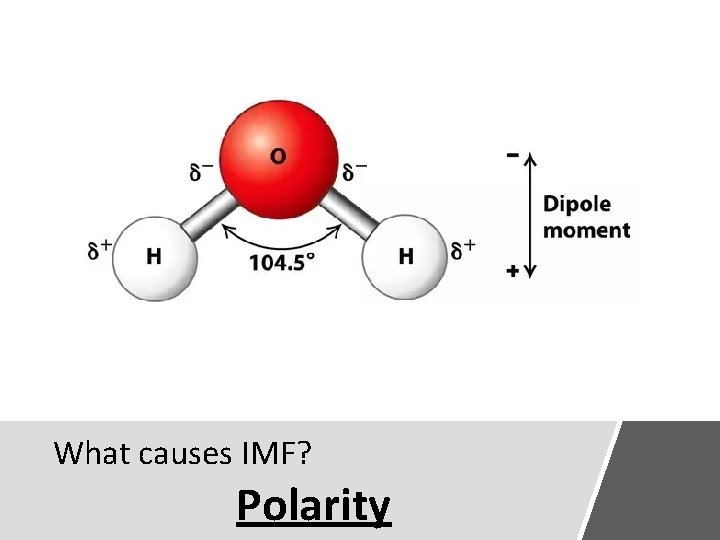
What causes IMF? Polarity

What caused the different penny results?
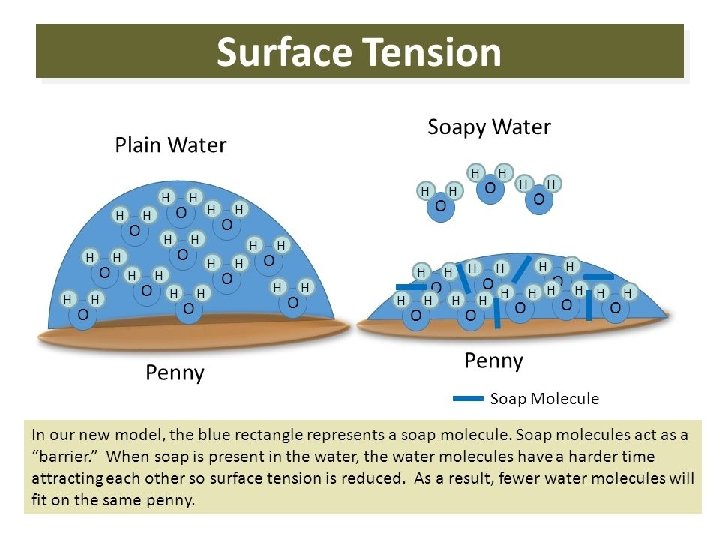

What caused the different penny results? • Non- polar substance messes with the bonding of a polar substance when mixes • Example, soap messing with water tension (caused by hydrogen bonding) • Like dissolves like • nonpolar substances dissolve best in nonpolar solvents, while polar substances dissolve best in polar solvents
- Slides: 22