Intermolecular Forces Intermolecular forces are attractive forces between

Intermolecular Forces Intermolecular forces are attractive forces between molecules. Intramolecular forces hold atoms together in a molecule. Intermolecular vs Intramolecular • 41 k. J to vaporize 1 mole of water (inter) • 930 k. J to break all O-H bonds in 1 mole of water (intra) “Measure” of intermolecular force Generally, intermolecular forces are much weaker than intramolecular forces. boiling point melting point DHvap DHfus DHsub 1
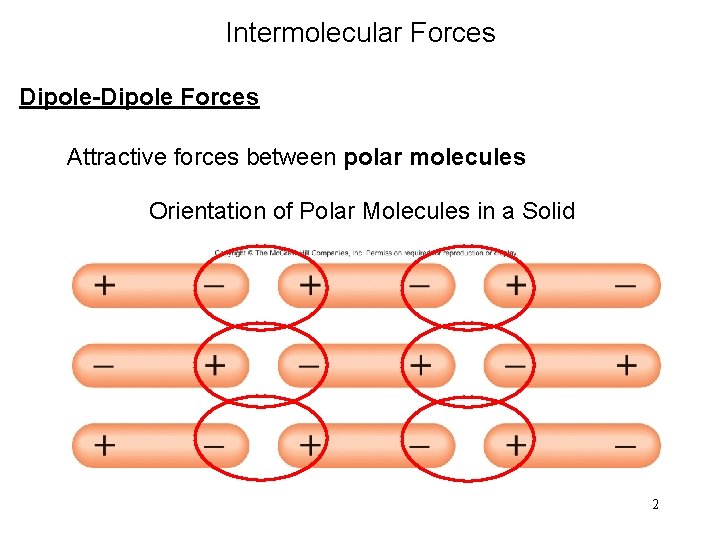
Intermolecular Forces Dipole-Dipole Forces Attractive forces between polar molecules Orientation of Polar Molecules in a Solid 2
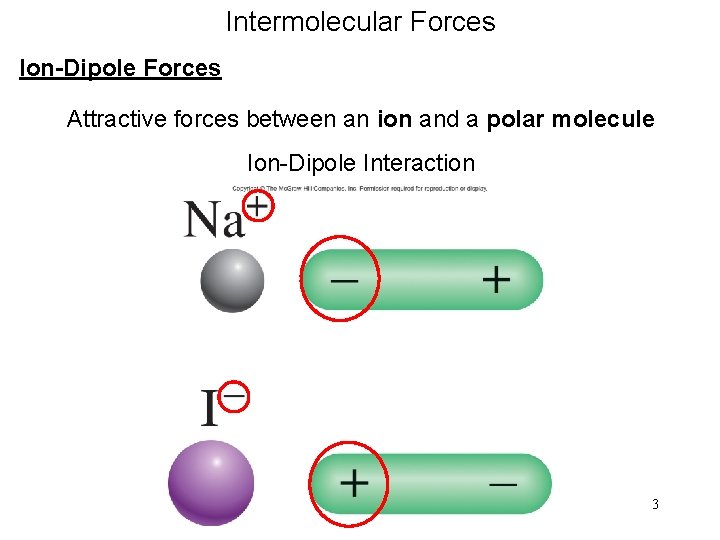
Intermolecular Forces Ion-Dipole Forces Attractive forces between an ion and a polar molecule Ion-Dipole Interaction 3
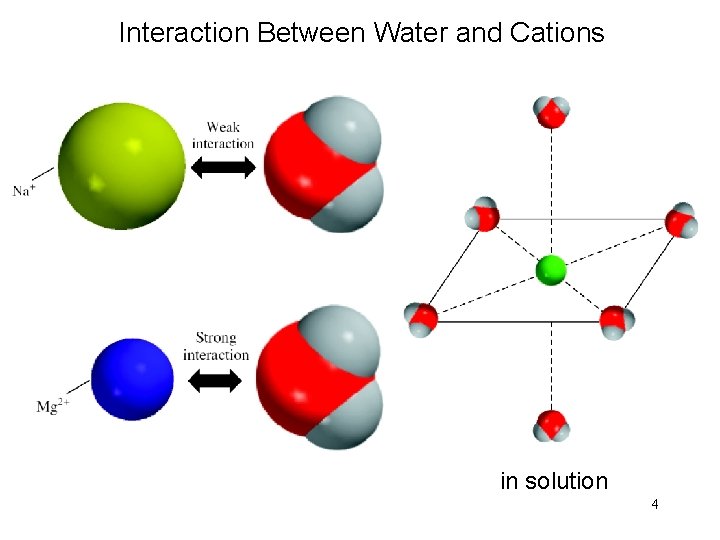
Interaction Between Water and Cations in solution 4
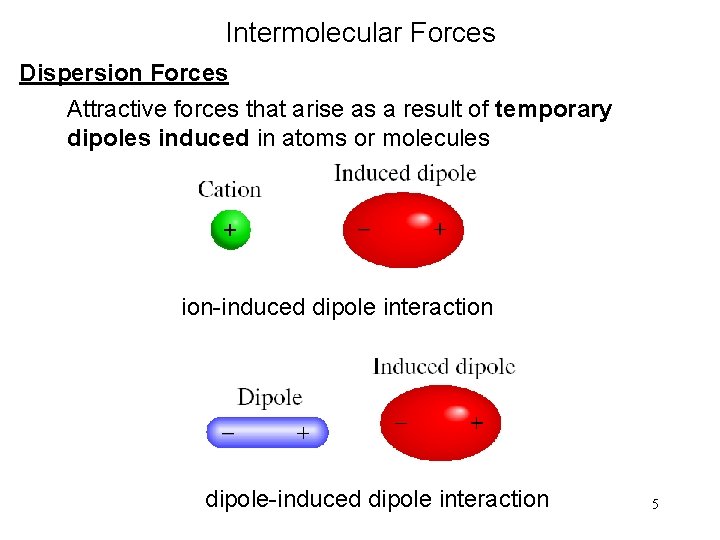
Intermolecular Forces Dispersion Forces Attractive forces that arise as a result of temporary dipoles induced in atoms or molecules ion-induced dipole interaction dipole-induced dipole interaction 5
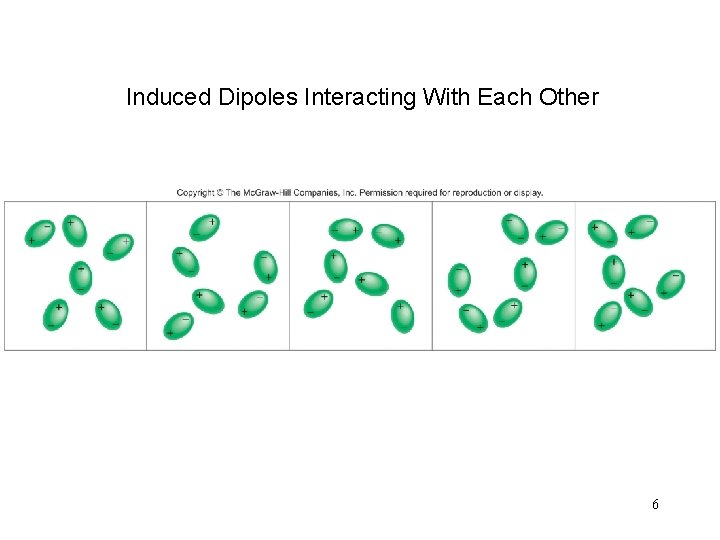
Induced Dipoles Interacting With Each Other 6
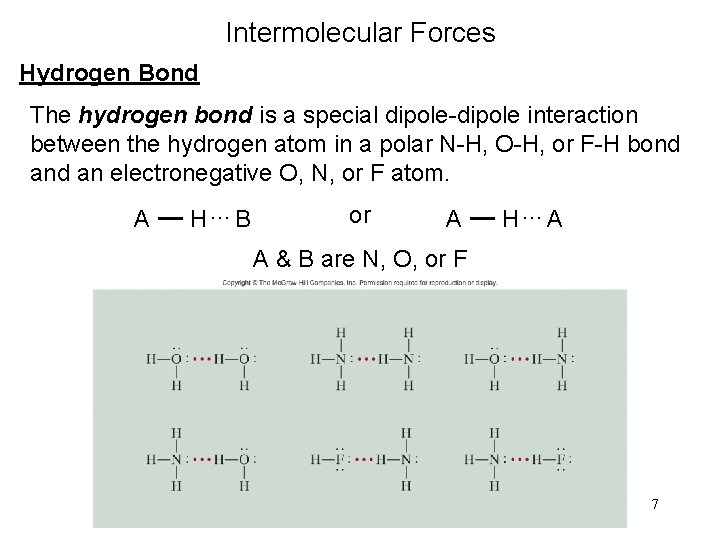
Intermolecular Forces Hydrogen Bond The hydrogen bond is a special dipole-dipole interaction between the hydrogen atom in a polar N-H, O-H, or F-H bond an electronegative O, N, or F atom. A H…B or A H…A A & B are N, O, or F 7

Example 11. 2 Which of the following can form hydrogen bonds with water? CH 3 OCH 3 CH 4 F− HCOOH Na+

Example 11. 1 What type(s) of intermolecular forces exist between the following pairs? (a) HBr and H 2 S (b) Cl 2 and CBr 4 (c) I 2 and (d) NH 3 and C 6 H 6
Total attraction pre • Hydrogen bond – Dipole-dipole – Dispersion V T t n re e f f i d s *Like l package e n n a ch • Dipole-dipole – Dispersion • Dispersion mi um ba sic 10

Intermolecular Forces Dispersion Forces Continued Polarizability is the ease with which the electron distribution in the atom or molecule can be distorted. Polarizability increases with: • greater number of electrons • more diffuse electron cloud Dispersion forces usually increase with molar mass. 11

Differences in Melting and Boiling point • Melting and boiling points increase as the number of electrons in a molecule increases • More electrons = larger molar mass 12
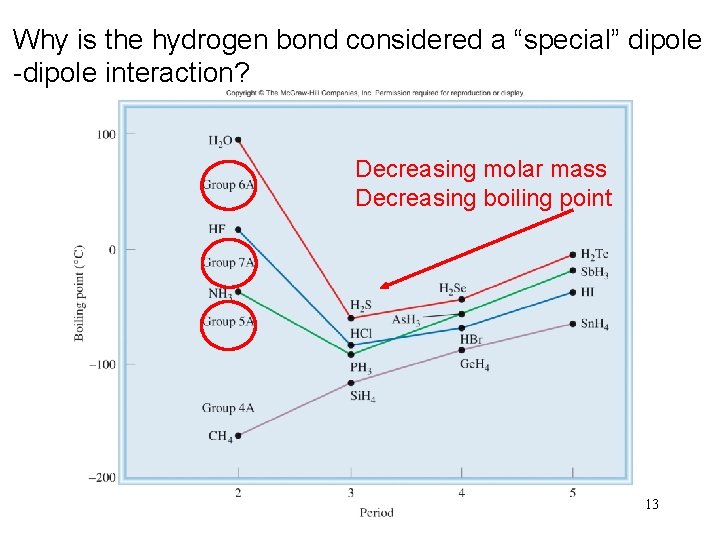
Why is the hydrogen bond considered a “special” dipole -dipole interaction? Decreasing molar mass Decreasing boiling point 13
- Slides: 13