Intermolecular Forces IMFs Forces not bondsBETWEEN molecules IMFs
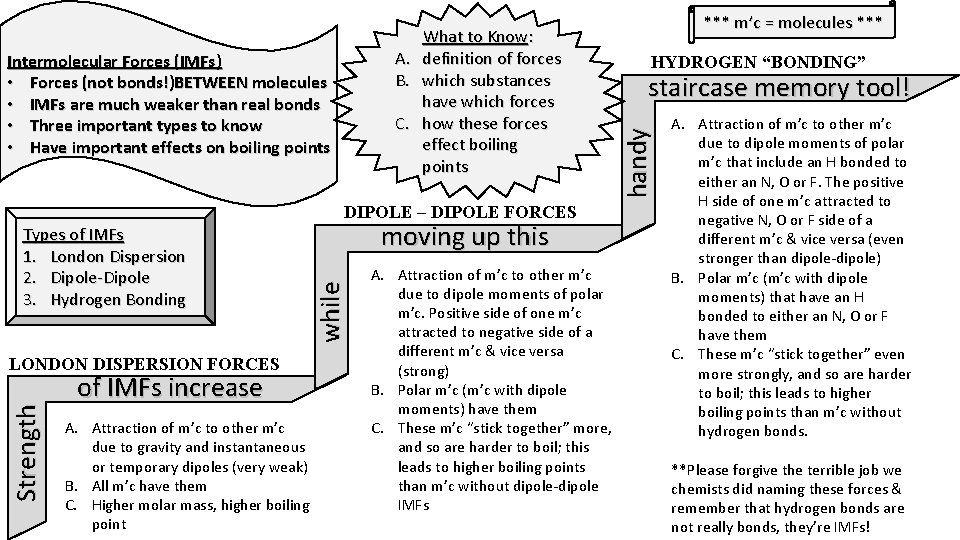
Intermolecular Forces (IMFs) • Forces (not bonds!)BETWEEN molecules • IMFs are much weaker than real bonds • Three important types to know • Have important effects on boiling points DIPOLE – DIPOLE FORCES Strength LONDON DISPERSION FORCES of IMFs increase A. Attraction of m’c to other m’c due to gravity and instantaneous or temporary dipoles (very weak) B. All m’c have them C. Higher molar mass, higher boiling point moving up this while Types of IMFs 1. London Dispersion 2. Dipole-Dipole 3. Hydrogen Bonding A. Attraction of m’c to other m’c due to dipole moments of polar m’c. Positive side of one m’c attracted to negative side of a different m’c & vice versa (strong) B. Polar m’c (m’c with dipole moments) have them C. These m’c “stick together” more, and so are harder to boil; this leads to higher boiling points than m’c without dipole-dipole IMFs *** m’c = molecules *** HYDROGEN “BONDING” staircase memory tool! handy What to Know: A. definition of forces B. which substances have which forces C. how these forces effect boiling points A. Attraction of m’c to other m’c due to dipole moments of polar m’c that include an H bonded to either an N, O or F. The positive H side of one m’c attracted to negative N, O or F side of a different m’c & vice versa (even stronger than dipole-dipole) B. Polar m’c (m’c with dipole moments) that have an H bonded to either an N, O or F have them C. These m’c “stick together” even more strongly, and so are harder to boil; this leads to higher boiling points than m’c without hydrogen bonds. **Please forgive the terrible job we chemists did naming these forces & remember that hydrogen bonds are not really bonds, they’re IMFs!
- Slides: 1