Intermolecular Forces Forces that occur BETWEEN molecules covalent

Intermolecular Forces
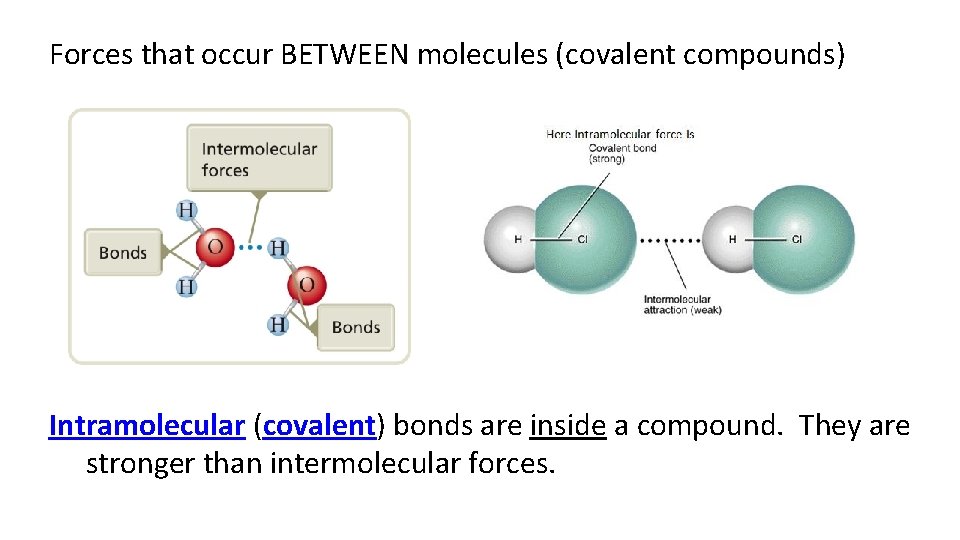
Forces that occur BETWEEN molecules (covalent compounds) Intramolecular (covalent) bonds are inside a compound. They are stronger than intermolecular forces.

Dipole - When there is a positive (δ+) and a negative (δ-) side to an atom or molecule Can be…… * temporary - forms when electrons momentarily shift to one side of an atom at random * induced - occurs when a neighboring molecule moves past one with a temporary dipole * permanent - the result of a polar bond in the molecular structure

There are three types of Intermolecular Forces (IMFs) all based on the strength of attractive forces
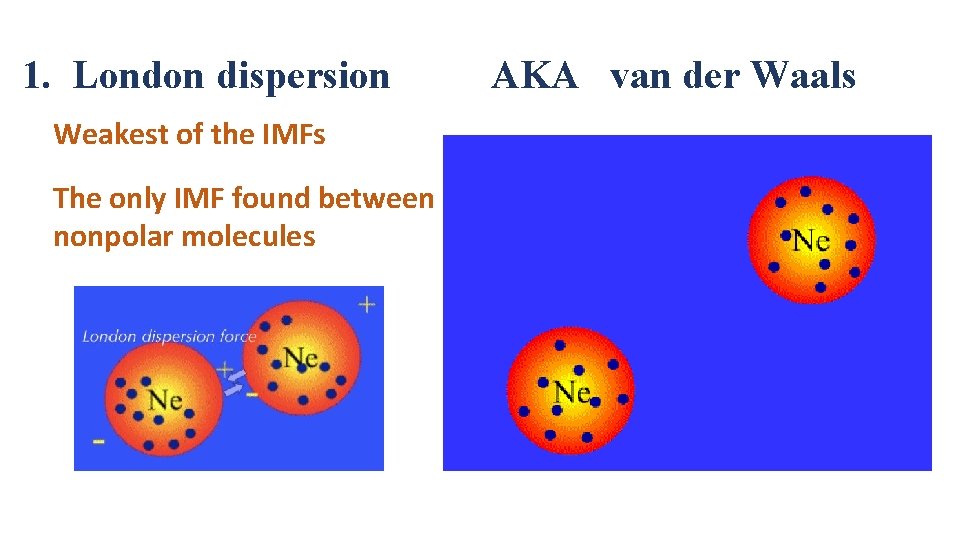
1. London dispersion Weakest of the IMFs The only IMF found between nonpolar molecules AKA van der Waals

While the electrons in helium would normally repel each other (and, therefore, tend to stay far away from each other), they do occasionally end up on the same side of the atom. At that instant, the helium atom is polar, with an excess of electrons on the left side and a shortage on the right side.

Another helium nearby, then, would have a dipole induced in it, as the electrons on the left side of helium atom 2 repel the electrons in the cloud on helium atom 1. London dispersion forces are attractions between an induced dipole and an induced dipole. These forces are present in ALL molecules
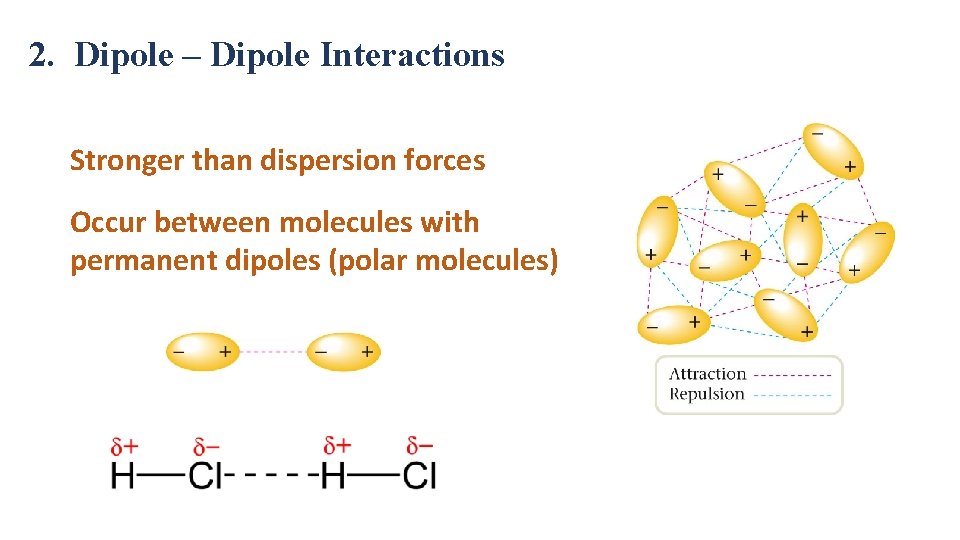
2. Dipole – Dipole Interactions Stronger than dispersion forces Occur between molecules with permanent dipoles (polar molecules)
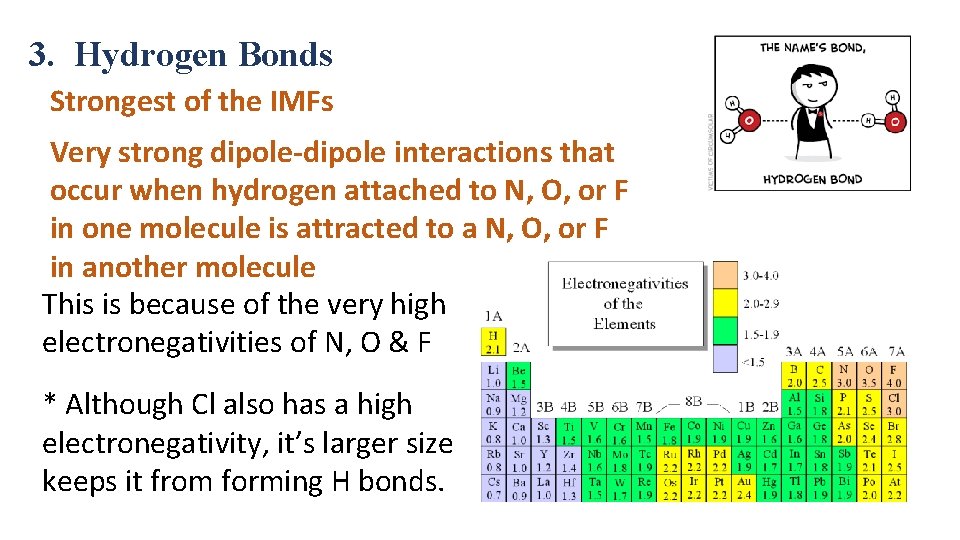
3. Hydrogen Bonds Strongest of the IMFs Very strong dipole-dipole interactions that occur when hydrogen attached to N, O, or F in one molecule is attracted to a N, O, or F in another molecule This is because of the very high electronegativities of N, O & F * Although Cl also has a high electronegativity, it’s larger size keeps it from forming H bonds.
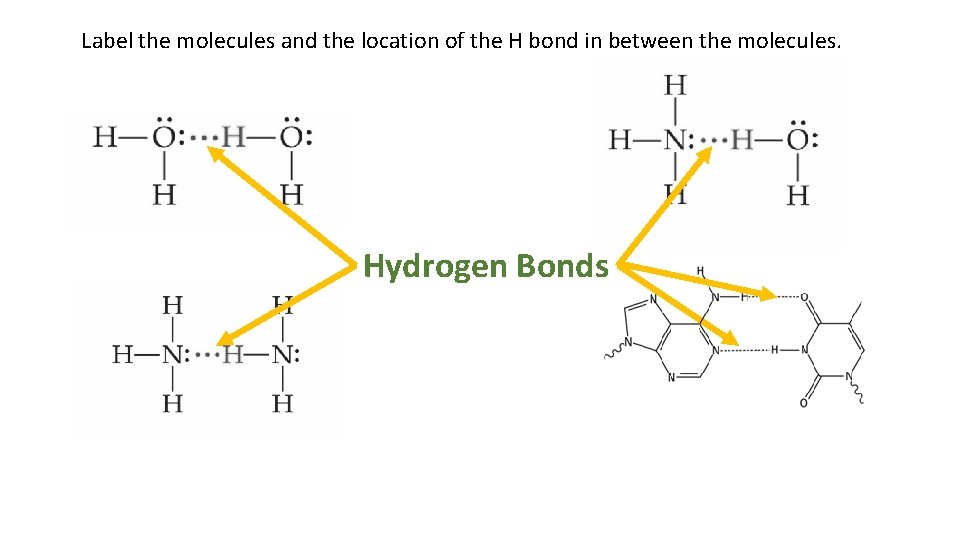
Label the molecules and the location of the H bond in between the molecules. Hydrogen Bonds

Hydrogen bonds are what make water have so many special properties

Intermolecular Forces Affect Many Physical Properties Viscosity – resistance of liquid to flow Increases with stronger intermolecular forces and decreases with higher temperature. Surface Tension - results from the inward force experienced by the molecules on the surface of a liquid Melting & Boiling Points – stronger IMFs equal higher melting & boiling points
- Slides: 12